Abstract
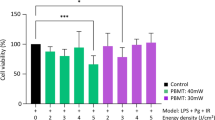
Erbium-doped yttrium aluminum garnet (Er:YAG) laser treatment has demonstrated favorable wound healing effect after periodontal therapy. One of the reasons may be the positive biological effect of the low-level laser on the irradiated tissues, although the mechanism remains unclear. The aim of this study was to investigate the effect of low-level Er:YAG laser irradiation on cell proliferation and laser-induced differential expression of proteins in human gingival fibroblasts (HGFs) by proteomic analysis. In the first experiment, HGFs were exposed to low-level Er:YAG laser irradiation and the laser-induced cell proliferation and damage were evaluated on day 3. In the second experiment, proteomic analysis was performed on day 1 after irradiation. The peptides prepared from HGFs were analyzed by a hybrid ion trap-Fourier transform mass spectrometer, Mascot search engine, and UniProtKB database. A significant increase in cell proliferation without cell damage after irradiation was observed. Among the total identified 377 proteins, 59 proteins, including galectin-7, which was associated with the process of wound healing, were upregulated and 15 proteins were downregulated in laser-treated HGFs. In the third experiment, the increase in messenger RNA (mRNA) and protein expression of galectin-7 in the irradiated HGFs was validated by various analytical techniques. In addition, the effect of recombinant human galectin-7 on the modulation of HGFs proliferation was confirmed. The results indicate that low-level Er:YAG laser irradiation can promote HGF proliferation and induce a significant change in protein expression and the upregulation of galectin-7 expression may partly contribute to the increase in cell proliferation.




Similar content being viewed by others
References
Pihlstrom BL, Michalowicz BS, Johnson NW (2005) Periodontal diseases. Lancet 366(9499):1809–1820. doi:10.1016/S0140-6736(05)67728-8
Oda S, Nitta H, Setoguchi T, Izumi Y, Ishikawa I (2004) Current concepts and advances in manual and power-driven instrumentation. Periodontol 2000 36:45–58. doi:10.1111/j.1600-0757.2004.03674.x
Schwarz F, Sculean A, Georg T, Reich E (2001) Periodontal treatment with an Er: YAG laser compared to scaling and root planing. A controlled clinical study. J Periodontol 72(3):361–367. doi:10.1902/jop.2001.72.3.361
Aoki A, Sasaki KM, Watanabe H, Ishikawa I (2004) Lasers in nonsurgical periodontal therapy. Periodontol 2000 36:59–97. doi:10.1111/j.1600-0757.2004.03679.x
Ishikawa I, Aoki A, Takasaki AA, Mizutani K, Sasaki KM, Izumi Y (2009) Application of lasers in periodontics: true innovation or myth? Periodontol 2000 50:90–126. doi:10.1111/j.1600-0757.2008.00283.x
Crespi R, Cappare P, Toscanelli I, Gherlone E, Romanos GE (2007) Effects of Er:YAG laser compared to ultrasonic scaler in periodontal treatment: a 2-year follow-up split-mouth clinical study. J Periodontol 78(7):1195–1200. doi:10.1902/jop.2007.060460
Sawabe M, Aoki A, Komaki M, Iwasaki K, Ogita M, Izumi Y (2013) Gingival tissue healing following Er:YAG laser ablation compared to electrosurgery in rats. Lasers Med Sci. doi:10.1007/s10103-013-1478-z
Mester E, Mester AF, Mester A (1985) The biomedical effects of laser application. Lasers Surg Med 5(1):31–39
Karu T (1989) Photobiology of low-power laser effects. Health Phys 56(5):691–704
Ohshiro T, Calderhead RG (1991) Development of low reactive-level laser therapy and its present status. J Clin Laser Med Surg 9(4):267–275
Pourzarandian A, Watanabe H, Ruwanpura SM, Aoki A, Ishikawa I (2005) Effect of low-level Er:YAG laser irradiation on cultured human gingival fibroblasts. J Periodontol 76(2):187–193. doi:10.1902/jop.2005.76.2.187
Aleksic V, Aoki A, Iwasaki K, Takasaki AA, Wang CY, Abiko Y, Ishikawa I, Izumi Y (2010) Low-level Er:YAG laser irradiation enhances osteoblast proliferation through activation of MAPK/ERK. Lasers Med Sci 25(4):559–569. doi:10.1007/S10103-010-0761-5
Peplow PV, Chung TY, Ryan B, Baxter GD (2011) Laser photobiomodulation of gene expression and release of growth factors and cytokines from cells in culture: a review of human and animal studies. Photomed Laser Surg 29(5):285–304. doi:10.1089/pho.2010.2846
Zhang Y, Song S, Fong CC, Tsang CH, Yang Z, Yang M (2003) cDNA microarray analysis of gene expression profiles in human fibroblast cells irradiated with red light. J Invest Dermatol 120(5):849–857. doi:10.1046/j.1523-1747.2003.12133.x
Schwanhausser B, Busse D, Li N, Dittmar G, Schuchhardt J, Wolf J, Chen W, Selbach M (2011) Global quantification of mammalian gene expression control. Nature 473(7347):337–342. doi:10.1038/nature10098
Tsuchida S, Satoh M, Umemura H, Sogawa K, Kawashima Y, Kado S, Sawai S, Nishimura M, Kodera Y, Matsushita K, Nomura F (2012) Proteomic analysis of gingival crevicular fluid for discovery of novel periodontal disease markers. Proteomics 12(13):2190–2202. doi:10.1002/pmic.201100655
Tsuchida S, Satoh M, Kawashima Y, Sogawa K, Kado S, Sawai S, Nishimura M, Ogita M, Takeuchi Y, Kobyashi H, Aoki A, Kodera Y, Matsushita K, Izumi Y, Nomura F (2013) Application of quantitative proteomic analysis using tandem mass tags for discovery and identification of novel biomarkers in periodontal disease. Proteomics 13(15):2339–2350. doi:10.1002/pmic.201200510
McKnight H, Kelsey WP, Hooper DA, Hart TC, Mariotti A (2014) Proteomic analyses of human gingival and periodontal ligament fibroblasts. J Periodontol 85(6):810–818. doi:10.1902/jop.2013.130161
Ara T, Kurata K, Hirai K, Uchihashi T, Uematsu T, Imamura Y, Furusawa K, Kurihara S, Wang PL (2009) Human gingival fibroblasts are critical in sustaining inflammation in periodontal disease. J Periodontal Res 44(1):21–27. doi:10.1111/j.1600-0765.2007.01041.x
Cao Z, Said N, Amin S, Wu HK, Bruce A, Garate M, Hsu DK, Kuwabara I, Liu FT, Panjwani N (2002) Galectins-3 and -7, but not galectin-1, play a role in re-epithelialization of wounds. J Biol Chem 277(44):42299–42305. doi:10.1074/jbc.M200981200
Cao Z, Said N, Wu HK, Kuwabara I, Liu FT, Panjwani N (2003) Galectin-7 as a potential mediator of corneal epithelial cell migration. Arch Ophthalmol 121(1):82–86
Gendronneau G, Sidhu SS, Delacour D, Dang T, Calonne C, Houzelstein D, Magnaldo T, Poirier F (2008) Galectin-7 in the control of epidermal homeostasis after injury. Mol Biol Cell 19(12):5541–5549. doi:10.1091/mbc.E08-02-0166
Rondanino C, Poland PA, Kinlough CL, Li H, Rbaibi Y, Myerburg MM, Al-bataineh MM, Kashlan OB, Pastor-Soler NM, Hallows KR, Weisz OA, Apodaca G, Hughey RP (2011) Galectin-7 modulates the length of the primary cilia and wound repair in polarized kidney epithelial cells. Am J Physiol Renal Physiol 301(3):F622–633. doi:10.1152/ajprenal.00134.2011
Cho SB, Kim JS, Zheng Z, Choi MJ, Choi IG, Oh HS, Ahn KJ (2013) Decreased tissue and serum expression of galectin-7 in patients with hypertrophic scars. Acta Derm Venereol 93(6):669–673. doi:10.2340/00015555-1583
Chan TA, Hermeking H, Lengauer C, Kinzler KW, Vogelstein B (1999) 14-3-3Sigma is required to prevent mitotic catastrophe after DNA damage. Nature 401(6753):616–620. doi:10.1038/44188
Hermeking H, Lengauer C, Polyak K, He TC, Zhang L, Thiagalingam S, Kinzler KW, Vogelstein B (1997) 14-3-3 sigma is a p53-regulated inhibitor of G2/M progression. Mol Cell 1(1):3–11
Fukuhara E, Goto T, Matayoshi T, Kobayashi S, Takahashi T (2006) Optimal low-energy laser irradiation causes temporal G2/M arrest on rat calvarial osteoblasts. Calcif Tissue Int 79(6):443–450. doi:10.1007/s00223-006-0072-9
Thompson HW, Malter JS, Steinemann TL, Beuerman RW (1991) Flow cytometry measurements of the DNA content of corneal epithelial cells during wound healing. Invest Ophthalmol Vis Sci 32(2):433–436
Kampfer H, Pfeilschifter J, Frank S (2003) Expression and activity of arginase isoenzymes during normal and diabetes-impaired skin repair. J Invest Dermatol 121(6):1544–1551. doi:10.1046/j.1523-1747.2003.12610.x
Witte MB, Barbul A, Schick MA, Vogt N, Becker HD (2002) Upregulation of arginase expression in wound-derived fibroblasts. J Surg Res 105(1):35–42. doi:10.1006/jsre.2002.6443
Campbell L, Saville CR, Murray PJ, Cruickshank SM, Hardman MJ (2013) Local arginase 1 activity is required for cutaneous wound healing. J Invest Dermatol 133(10):2461–2470. doi:10.1038/jid.2013.164
Abiko Y, Hiratsuka K, Kiyama-Kishikawa M, Tsushima K, Ohta M, Sasahara H (2004) Profiling of differentially expressed genes in human gingival epithelial cells and fibroblasts by DNA microarray. J Oral Sci 46(1):19–24
Abe D, Kubota T, Morozumi T, Shimizu T, Nakasone N, Itagaki M, Yoshie H (2011) Altered gene expression in leukocyte transendothelial migration and cell communication pathways in periodontitis-affected gingival tissues. J Periodontal Res 46(3):345–353. doi:10.1111/j.1600-0765.2011.01349.x
Chidgey M, Brakebusch C, Gustafsson E, Cruchley A, Hail C, Kirk S, Merritt A, North A, Tselepis C, Hewitt J, Byrne C, Fassler R, Garrod D (2001) Mice lacking desmocollin 1 show epidermal fragility accompanied by barrier defects and abnormal differentiation. J Cell Biol 155(5):821–832. doi:10.1083/jcb.200105009
Saussez S, Kiss R (2006) Galectin-7. Cell Mol Life Sci 63(6):686–697. doi:10.1007/s00018-005-5458-8
Bertoldi C, Bellei E, Pellacani C, Ferrari D, Lucchi A, Cuoghi A, Bergamini S, Cortellini P, Tomasi A, Zaffe D, Monari E (2013) Non-bacterial protein expression in periodontal pockets by proteome analysis. J Clin Periodontol 40(6):573–582. doi:10.1111/jcpe.12050
Brigstock DR (2003) The CCN family: a new stimulus package. J Endocrinol 178(2):169–175
Wilkinson B, Gilbert HF (2004) Protein disulfide isomerase. Biochim Biophys Acta 1699(1–2):35–44. doi:10.1016/j.bbapap.2004.02.017
Seifert GJ, Lawson D, Wiche G (1992) Immunolocalization of the intermediate filament-associated protein plectin at focal contacts and actin stress fibers. Eur J Cell Biol 59(1):138–147
Gillett GT, Fox MF, Rowe PS, Casimir CM, Povey S (1996) Mapping of human non-muscle type cofilin (CFL1) to chromosome 11q13 and muscle-type cofilin (CFL2) to chromosome 14. Ann Hum Genet 60(Pt 3):201–211
Berbee JF, Coomans CP, Westerterp M, Romijn JA, Havekes LM, Rensen PC (2010) Apolipoprotein CI enhances the biological response to LPS via the CD14/TLR4 pathway by LPS-binding elements in both its N- and C-terminal helix. J Lipid Res 51(7):1943–1952. doi:10.1194/jlr.M006809
Chiquet-Ehrismann R, Chiquet M (2003) Tenascins: regulation and putative functions during pathological stress. J Pathol 200(4):488–499. doi:10.1002/path.1415
Shefer G, Oron U, Irintchev A, Wernig A, Halevy O (2001) Skeletal muscle cell activation by low-energy laser irradiation: a role for the MAPK/ERK pathway. J Cell Physiol 187(1):73–80. doi:10.1002/1097-4652(2001)9999:9999<::aid-jcp1053>3.0.co;2-9
Ejiri K, Aoki A, Yamaguchi Y, Ohshima M, Izumi Y (2013) High-frequency low-level diode laser irradiation promotes proliferation and migration of primary cultured human gingival epithelial cells. Lasers Med Sci. doi:10.1007/s10103-013-1292-7
Fischer A, Baljuls A, Reinders J, Nekhoroshkova E, Sibilski C, Metz R, Albert S, Rajalingam K, Hekman M, Rapp UR (2009) Regulation of RAF activity by 14-3-3 proteins: RAF kinases associate functionally with both homo- and heterodimeric forms of 14-3-3 proteins. J Biol Chem 284(5):3183–3194. doi:10.1074/jbc.M804795200
Dong C, Li C, Wu G (2011) Regulation of alpha(2B)-adrenergic receptor-mediated extracellular signal-regulated kinase 1/2 (ERK1/2) activation by ADP-ribosylation factor 1. J Biol Chem 286(50):43361–43369. doi:10.1074/jbc.M111.267286
Yu W, Naim JO, McGowan M, Ippolito K, Lanzafame RJ (1997) Photomodulation of oxidative metabolism and electron chain enzymes in rat liver mitochondria. Photochem Photobiol 66(6):866–871
Morimoto Y, Arai T, Kikuchi M, Nakajima S, Nakamura H (1994) Effect of low-intensity argon laser irradiation on mitochondrial respiration. Lasers Surg Med 15(2):191–199
Lubart R, Eichler M, Lavi R, Friedman H, Shainberg A (2005) Low-energy laser irradiation promotes cellular redox activity. Photomed Laser Surg 23(1):3–9. doi:10.1089/pho.2005.23.3
Acknowledgments
This study was partially supported by the grant of the Global Center of Excellence Program of the International Research Center for Molecular Science in Tooth and Bone Diseases at TMDU funded by the Ministry of Education, Culture, Sports, Science and Technology of Japan and the grant of Grants-in-Aid for Scientific Research (C) (no. 22392308 and no. 23463212 for A.A.) from the Japan Society for the Promotion of Science. We would like to acknowledge Dr. Kengo Iwasaki and Dr. Yoichi Taniguchi at TMDU for their kind advices and supports.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table 1
LC-MS/MS-identified 377 proteins in HGFs with and/or without low-level Er:YAG laser irradiation (PDF 185 kb)
Rights and permissions
About this article
Cite this article
Ogita, M., Tsuchida, S., Aoki, A. et al. Increased cell proliferation and differential protein expression induced by low-level Er:YAG laser irradiation in human gingival fibroblasts: proteomic analysis. Lasers Med Sci 30, 1855–1866 (2015). https://doi.org/10.1007/s10103-014-1691-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-014-1691-4