Abstract
Hydrogen is one of the key components in renewable energy systems. Its storage and transport, however, are challenging. The Liquid Organic Hydrogen Carrier (LOHC) technology is a possible solution for this issue. With suitable organic components, hydrogen can be stored in a chemically bound form which is safer and has a higher energy density than other solutions. Furthermore, the storage and transport of the LOHC component with high hydrogen content can be provided under normal environmental conditions. The LOHC process cycle involves a catalytic hydrogenation and dehydrogenation step where the LOHC component can be recovered and recycled. The paper reviews a few relevant studies regarding the possible LOHC compound pairs, the potential catalyst systems, and the necessary equipment. Then, the paper discusses a simulation study of three LOHC systems, determining suitable operating conditions and estimating costs. The toluene-methylcyclohexane system was found to be the most promising for LOHC application. Two energy integration opportunities were also examined. The first case study was based on separate hydrogenation and dehydrogenation sites, while the second was based on one site utilization. The results show a lower energy demand in the case of a toluene system, with a reduction of 70% in heating and 45% in cooling.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hydrogen is one of the key components in the future of renewable energy. Still, the hydrogen economy offers promising solutions for recent challenges considering energy supply, climate changes, environmental problems, and a sustainable economy.
To address some of the above challenges, Hungarian Gas Storage Ltd. (HGS, a member of MVM Group) started the Aquamarine pilot project in February 2021. The main objective is to establish a hydrogen-based energy storage solution as a sector integrator and contributor to balance the electricity grid in underground gas storage (CEEnergyNews 2021). The installation: an approximately 2.5 MW PEM-electrolyser system, a diaphragm hydrogen compressor, storage tanks and corresponding hydrogen gas preparatory technology with mixing units have already been in progress. The PEM-electrolyser will produce 400 Nm3/h hydrogen; then hydrogen blended natural gas will be injected into the transmission system partly for consumers and partly be used in the compressors, gas engines and regenerators.
Altogether the 6 R&D subprograms demonstrate the suitability and feasibility of hydrogen technologies in existing natural gas infrastructure. One of these subprograms aims to solve the long-term pure hydrogen storage and transport by applying Liquified Organic Hydrogen Carrier (LOHC) technology that would allow diversifying the hydrogen storage portfolio.
Hydrogen had already gained high importance as a decisive element of circular economy and green energy systems in the first half of the twenty-first century. As a pure energy source, it is expected to get an even more outstanding role by the century's second half. The serious challenge is replacing fossil energy carriers primarily used today with less polluting and more sustainable energy sources. One of the promising solutions is employing hydrogen in the global energy supply chain. Nowadays, hydrogen is typically produced by steam reforming of methane, oxidation of hydrocarbons or electrolysis of water. In addition, there are novel methods, e.g. thermochemical decomposition of water using nuclear or solar energy or different biotechnological processes; however, they all require further research and development. Moreover, industrial-scale hydrogen production, storage and transport solutions are only applied in chemical and refinery plants (Bourane et al. 2016).
According to an estimate by Bank of America, the hydrogen industry is expected to produce 2.5 billion USD by 2050. This will induce the rapid development of related industries like hydrogen-fuelled vehicles and heating and power supply services, especially gas supply solutions.
Hydrogen production technologies are classified into four main categories (U.S. Office of Energy Efficiency and Renewable Energy 2021):
-
Thermochemical processes (natural gas reforming, coal or biomass gasification, solar thermochemical, reforming of biomass-derived liquid fuels)
-
Water electrolysis
-
Photochemical processes (electrochemical or biotechnological)
-
Bioprocesses (microbial biomass conversion, photo biotechnology)
While CO2-free hydrogen production is expensive today, it is expected to be much cheaper and replace traditional technologies by 2060. European Union plans to decrease hydrogen production from fossil sources and produce renewable, so-called green hydrogen (European Commission, Energy System Integration 2021). The carbon neutrality of the economy targeted by 2050 can only be reached by promoting green hydrogen production and application, which requires adequate storage solutions. Several hydrogen-related initiatives pursue similar objectives in the USA (U.S. Department of Energy 2021).
As generally accepted, hydrogen storage between production and consumption is decisive regarding using hydrogen as fuel. The main difficulty comes from its low gas density. Hence liquid phase storage and transportation, the pumping characteristics become very important. This aspect also involves LOHC material being easy to pump and having good thermal stability. The different storage solutions are reviewed and assessed by Morris et al. (2019). However, it cannot be decided which is the best since this depends on the application type.
It is clear that the key to the hydrogen economy, i.e. building the industrial and communal energy supply on hydrogen as an energy carrier, is solving the storage and transportation of hydrogen in a flexible, safe and economical way. A suitable and effective alternative to storing hydrogen under high pressure or in liquefied, cryogenic and metal hybrid forms can be the application of Liquid Organic Hydrogen Carriers. It is also important that the organic carrier can be hydrogenized at the gas and oil fields, significantly reducing CO2 emissions. Furthermore, the hydrogen carrier can easily be transported to the consumers by tankers, trucks or pipelines. The dehydrogenation step can be accomplished at separated hydrogen stations or onboard vehicles. This scheme offers less safety risk; the material is chemically stable and does not require cooling or heating during storage. The hydrogen loss can be low, too, since the transport and storage can be done under ambient temperature and pressure (Bourane et al. 2016).
The basic idea is a process cycle involving catalytic hydrogenation and dehydrogenation of the organic carrier. The application of benzene/cyclohexene had already been analyzed in detail 40 years ago, and it was stated that 98% theoretical efficiency could be reached by applying thermal recovery. Furthermore, based on the evaluations, the relatively low cost and the high hydrogen density (56 kg H2/m3 carrier) are considerable advantages. Therefore, it was concluded that the process could be feasible and economical either in daily, weekly or seasonal storage (Cacciola et al. 1984).
Two decades ago, Scherer et al. investigated the cost aspects of LOHC technologies, specifically in electric energy storage. In this case, they considered the toluene/methyl-cyclohexene carrier for storing electric energy produced at a relatively low cost during summer. An eight-stage heat exchanger and an adiabatic reactor were applied in the dehydrogenation step to improve energy efficiency. While the process was not economical regarding the recovery of electric energy; however, the approximately 80% lower CO2 emission and the strategic independence of the energy supply could compensate for that (Scherer et al. 1999).
Extensive research has been conducted studying several chemical compounds, including single and polycyclic cycloalkanes, heterocyclic compounds and even ionic liquids. Experimental works have mainly focused on developing efficient catalyst systems and suitable reactor constructions. At the same time, modelling efforts have targeted components allowing dehydrogenation with low energy demand. For example, preliminary studies showed that heterocyclic compounds are well-suited for onboard applications due to their low-energy dehydrogenation properties (Bourane et al. 2016).
Many authors consider methanol and liquid ammonia as liquid hydrogen carriers too. They have been produced and applied in industry for a long time but have not been directly applied as hydrogen carriers. Methanol can be produced from synthesis gas or recycled CO2 using different catalytic processes. At the same time, it can be directly applied as fuel, e.g. by using membrane fuel cells (Olah et al. 2018). Catalytic processes can also produce ammonia, and hydrogen can be recovered catalytically, or it can also serve as fuel, e.g. by employing PEM (Polymer Electrolyte Membrane) fuel cells. Ammonia has the highest volumetric hydrogen density (121 kg H2/m3) among liquid hydrogen carriers (Makepeace et al. 2019). However, these solutions are less flexible and less scalable; therefore, they cannot be suitable for balancing energy fluctuations (Bourane et al. 2016).
The REPowerEU strategy focuses on the clean and green energy transition, which has a key component: hydrogen (REPowerEU 2019). The production and utilization of green hydrogen is the primary goal of our Aquamarine project. One of the cornerstones of using hydrogen is storage and transportation, which can mostly be completed with the LOHC technologies, the main topic of our research.
Experiments in real-world systems, especially on a large scale, are often expensive and complicated, making it difficult to study the different technologies. Modelling can save time and money and support the decision and equipment design. In order to design the LOHC processes efficiently with minimal costs, it is essential to develop a practical model to investigate the limitations of the process equipment.
The primary contribution of our research is the simulation-based comparison of three promising LOHC-based technologies. Besides the CAPEX and OPEX calculation, energy integration scenarios are also included. Most of the detailed comparisons found in the literature are limited to conversion reactors, and do not use detailed kinetics or calculation of energy integration scenarios. This is the field where our work extends the current state of the art. The results can also support the pilot-scale development steps planned in the near future.
The first part of the manuscript is a detailed literature study about LOHC technology (sections "Liquid organic hydrogen carrier (LOHC) technology and Hydrogenation and dehydrogenation at the industrial scale"). Sections "Techno-economic evaluation of promising LOHC systems and Energy integration scenarios" focus on the techno-economic assessment of three different processes, while the energy integration scenarios are discussed in section "Discussion of EU and Hungarian policies and the contribution of the present work", followed by the Discussion of the EU and Hungarian policies and the Conclusion of our work (section "Conclusion").
Liquid organic hydrogen carrier (LOHC) technology
A recent historical review of organic hydrogen carriers reveals that the potential of using suitable organic compounds for hydrogen storage has been studied for more than 40 years. Different groups studied several hydrogen-rich/hydrogen-lean pairs, choosing suitable catalysts for the two steps of the circular process (Meille and Pitault 2021). In the following sections, the LOHC cycle, the applicable carriers and the catalyst are discussed before discussing the modelling methods and results.
The LOHC cycle
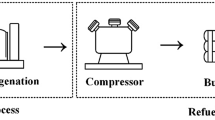
LOHC stands for organic compound pairs that allow storing hydrogen in the liquid phase in chemically bonded form and then recovering it (Rao and Yoo 2020). The basic concept of the LOHC technology was first evaluated by the benzene/cyclohexane system (Klvanaet al. 1988). Many carriers, including aliphatic (methanol, formic acid), single-ring aromatic (toluene), multi-ring aromatic (naphthalene, dibenzyl toluene), as well as nitrogen heterocycle aromatic (N-ethyl carbazole, phenazine) compounds have been studied so far (Cho et al. 2021). The reversible catalytic cycle is illustrated in Fig. 1.
The catalytic LOHC cycle [based on Makaryan and Sedov (2021)]
Hydrogenation is a very exothermic reaction, and it is generally conducted at medium temperatures (80–240 °C) and pressures (5–50 bar) (Rao and Yoo 2020). The dehydrogenation step is endothermic, requiring excess heating; simultaneously, it can be done at relatively low or even atmospheric pressure. The general arrangement of the technology is depicted in Fig. 2. The LOHC technologies can be part of processes taking gaseous hydrogen into the liquid phase (liquefying block). While catalytic hydrogenation is commonly used in the industry, large-scale catalytic dehydrogenation, especially aimed at recovering hydrogen, has not been elaborated so deeply (Rao et al. 2020).
A detailed study comparing four LOHC systems and discussing the design, economic and market variance aspects of a large-scale hydrogen delivery process (1000 Nm3/h) is presented by Brigljevic et al. (2020). They considered that the selected LOHC systems should be valid, i.e. confirmed experimentally and thermodynamically, recyclable, i.e. reused reversibly, and allow temperature stages in the dehydrogenation. The four LOHC systems were a eutectic biphenyl/diphenylmethane mixture, a pyridine-based system (MBP), N-phenyl carbazole and N-ethyl carbazole. The study used Aspen Plus simulations and cost estimations and assumed that the energy demand of the Process is covered by burning a part of the recovered hydrogen. The four LOHC systems were compared in themselves first. Then utilizing the temperature stages between the dehydrogenation reactions, a four-stage thermally coupled process was built. Since the flue gas exiting a higher temperature stage could be utilized in the lower temperature stage, they could reach close heat integration (Brigljevic et al. 2020).
Based on the cost estimations, economic assessment and market uncertainty analysis, it was stated that the MBP system had the best performance purely on an economic basis. However, considering market uncertainties, including LOHC, hydrogen price, and process parameter variations, the four-stage thermal cascade provided a much more robust solution.
LOHC systems/compounds
There are several critical components in the catalytic cycle:
-
LOHC carrier, the organic molecule pair that can be selectively hydrogenated—dehydrogenated, preferably with high recovery efficiency.
-
LOHC catalyst pair for hydrogenation and dehydrogenation.
-
Hydrogen as a reactant.
Several LOHC compounds have been tested in recent years, and the following are examined in detail in a comprehensive study (Niermann et al. 2019a):
-
Methanol.
-
Formic acid.
-
Toluene.
-
Dibenzyl toluene.
-
Naphthalene.
-
N-ethyl carbazole.
-
N-Propylcarbazole.
-
Phenazine.
-
N-ethylindole.
-
1,2-dihydro-1,2-azaborine.
Among the carrier compounds, low-carbon hydrocarbons are typically capable of taking up 1 mol of hydrogen (methanol, formic acid). The other compounds well suited for LOHC reactions are aromatic compounds, for example, toluene, which can bind 3 mol of hydrogen. The range of catalysts is also wide and, in many cases, specific. It can be said that the active elements of a catalyst are usually metals or metal oxides on some support.
The most widely used LOHC system is the toluene–methylcyclohexane system. In the case of toluene, during the hydrogenation process, the hydrogen molecule reacts chemically with the organic carrier, which is usually an aromatic hydrocarbon. As a result, the aromatic ring is saturated to yield the LOHC component, which has an increased energy density. Therefore, the resulting compound is stable in this state and is easy to transport. Before use, dehydrogenation is carried out on-site to recover the unsaturated aromatic carrier. The carrier can then be reused over several cycles (Niermann et al. 2019b).
Several market contributors are developing LOHC technologies using DBT as the carrier, for example, Hydrogeneous Technologies and Framatome. The toluene carrier is facilitated in the Chiyoda Corporation pilot plant. The reactions can be performed in fixed-bed reactors. However, in the case of dehydrogenation, to achieve proper heating, structured monolith, radial flow, or even spray pulsed reactors can be used (Modisha et al. 2019).
Heterogeneous catalysts are often used in industrial hydrogenation reactions (Makaryan and Sedov 2021). These are usually expensive, noble metal hydrogenation catalysts (nickel, palladium or platinum) fixed as a fine powder on cheap, porous support (e.g. alumina). Table 1 contains the properties of the most relevant hydrogenation catalysts in the case of different LOHC components. Dehydrogenation reactions typically use a Pt noble metal catalyst on various supports. Table 2 contains the properties of the most relevant dehydrogenation catalysts in the case of different LOHC components.
While reversible catalytic hydrogenation and dehydrogenation have well-established chemistry, suitable catalyst systems still require extensive research since they significantly influence the Process, especially considering the dehydrogenation rate. Makaryan and Sedov (2021) discuss these topics in their review paper, focusing on the catalyst aspects. Among the industrial initiatives, they mention the HySTOC project, which uses dibenzyl toluene based on the development of Hydrogenious Ltd. The choice of LOHC substrate, besides the more generic requirements (low freezing and high boiling point, nontoxic, safe storage, hydrogen density), is strongly based on energy aspects. Since hydrogenation enthalpy is below 62.7 kJ/mol it is expected that the temperature for dehydrogenation can be decreased. This involves that condensed polycyclic compounds are advantageous since the heat of the reaction decreases with the higher number of aromatic rings (Kustov and Kalenchuk 2002). For example, the dehydrogenation temperature can be lowered by lowering the pressure or applying separation techniques within the reactor (or between catalyst beds) (Müller et al. 2021). However, the applied pressure and conditions will affect the catalyst's performance and the conversion. Besides adding new unit operations, separators could excessively affect the CAPEX and OPEX costs. The choice of the catalyst system is a function of the selected LOHC substrate, and factors like cost, activity, selectivity, stability, and operation life must be considered. Besides the suitable mono or bimetallic components (mainly Pt and Pd), the catalyst support material, surface area, and particle size influence the catalytic properties. Al2O3, C, TiO2 and SiO2 are widely investigated as catalyst support materials. Since catalysts are losing their activity over time, another critical point can be the deactivation of the catalyst due to coke formation or poisoning, as well as the possible ways of their regeneration. The coke formation is often significant for LOHC dehydrogenation because the products generally contain aromatic rings (Ahn et al. 2022). Deactivation in the dehydrogenation phase can be slower at high pressure and feeding excess hydrogen. Regeneration can generally be reached by high-temperature oxidation with air (Makaryan and Sedov 2021). Recent studies have shown that less costly materials can replace expensive noble metal-based catalysts.
Hydrogenation and dehydrogenation at the industrial scale
First, hydrogenation and dehydrogenation processes are shortly examined here not purely from LOHC aspects but from organic chemistry. Then some aspects of their industrial-scale realization are discussed.
Hydrogenation
Catalytic hydrogenation of organic compounds in the presence of hydrogen is highly exothermic. Consequently, strict temperature control is necessary since increasing the temperature favours hydrogen release (reverse reaction) (Stoffels 2020).
The main factors influencing the reaction rate are the physical and chemical properties of the fluids, the catalyst characteristics and the operational conditions. The raw material is primarily liquid, while hydrogen enters gas into the reactor charged with a solid catalyst. The rate-limiting step is most often the hydrogen transport to the catalyst surface. Therefore excess gas feed is also a decisive parameter.
A wide variety of hydrogenation catalysts is available commercially. Usually, they are optimized for a given process. Active sites are formed by metals from the platinum or transitional group (Pt, Pd, Rh, Ru, Ni, Cu, Mo, Co). Depending on the required activity, selectivity, stability and lifecycle, different metal and metal oxide compositions are formed in the catalyst production (Sanfilippo and Rylander 2012).
Heterogeneous catalytic processes can be realized in fixed bed construction with an unstructured charge of 1–6 mm diameter catalyst pellets. Co- or countercurrent contact of the liquid and gas phases can be applied. To avoid temperature runaway, cold hydrogen is fed stagewise; cooling between the beds and external cooling of recycle streams can be applied. The contact between different phases is intensified by employing active or passive mixing elements. The other option is dispersing micro or nanoparticles in the liquid phase in fluidizing or suspending processes.
Hydrogen saturation of monocyclic compounds is a strongly exothermic reaction and can be conducted in the presence of a noble metal catalyst. The simplest system is the toluene/methylcyclohexane pair. This process can be accomplished, e.g. at 150–230 °C and 0.5–3 bar using Pt/Pd/Ru catalyst. Ni catalyst can be employed at 95–125 °C 20–40 bar. The catalyst support is usually Al2O3, although other supports like zeolites can also be used (e.g. ZSM-5, ZSM-22) (Thybaut 2002).
The hydrogen uptake of polycyclic compounds is similar to monocyclic ones; however, the required temperature is generally lower. E.g. from naphthalene, tetralin can be formed at 150–200 °C and 1 bar using (NiO/SiO2–Al2O3) catalyst and applying toluene as solvent. Increasing the reactor temperature allows the production of decalin (Kirumakki et al. 2006).
The hydrogenation potential of heterocyclic components is higher than that of the previous compounds. Although the relatively low temperature of dehydrogenation and the low vapour pressure are advantageous, there might be a need for separation due to the side products (Taube 1985).
Dehydrogenation (recovery of hydrogen)
Due to splitting the C-H bonds, the catalytic dehydrogenation of hydrocarbons is a highly endothermic reaction resulting in hydrogen production and frequent coking.
Industrial processes mainly focus on the dehydrogenation of alkanes at 500–700 °C using Pt/Sn or Cr2O3 on Al2O3 support as the catalyst. The Pt/Sn catalyst is usually applied on γ-Al2O3 support. The amount of Sn has an important effect on activity and selectivity, reducing the acidic character and, consequently, the coking rate (Armende 2016).
The following few industrial-scale dehydrogenation processes demonstrate how these can be realized (Maddah 2018).
The Catofin dehydrogenation process (Houndry-ABB Lummus) employs Cr/Al2O3 at 0.3–1 bar and 540–760 °C to produce propene. The reactors are operated in a four-stage cycle: vacuum, dehydrogenation, blowdown and decoking. The Oleflex dehydrogenation process (UOP) applies moving bed Pt/Sn/Al2O3 catalyst circulating several successive cascade units. After the last cascade element, the catalyst enters the regeneration unit (Nawaz 2015).
The fluid bed dehydrogenation process (FBD—Snamprogetti-Yarsintez) applies Cr/Al2O3 catalyst recycling in a fluidized twin reactor system at 500–700 °C. The heat released in the regenerator provides the heat required for dehydrogenation. The regeneration is accomplished using oxygen from the air. The novel processes (UOP, Axens) have already applied moving bed catalyst solution using more active (and coking more rapidly) catalysts. The regeneration cycle is part of the process scheme. It should be noted that considering the dehydrogenation (reforming) of different compounds, the dehydrogenation of cyclohexanes can be realized the most readily.
Techno-economic evaluation of promising LOHC systems
Based on the literature review, the most promising three processes were, applied toluene, dibenzyl-toluene and naphthalene. DBT and toluene were selected as they also have an existing technology for LOHC application, where naphthalene has the highest energy density according to the literature, which is why we chose naphthalene as a third one (Niermann et al. 2019a, b). A plant with a capacity of 50 Nm3 H2/h was simulated, a common pilot-level load for technologies (also applied by Chiyoda Corporation). The following boundary conditions were kept in mind during the simulations for comparability. The conditions of the inlet hydrogen stream were at 30 bar and 25 °C for the hydrogenation technologies.
In contrast, the dehydrogenation technologies were calculated so that the conditions of the hydrogen stream leaving them were also at 30 bar and 25 °C. The thermodynamic estimation method used in the simulations of naphthalene—decalin and toluene—ethylcyclohexane systems was the Peng-Robinson method, while in the case of dibenzyltoluene,—perhydro-dibenzyltoluene system was the UNIQUAC method because of the large size of molecules (Anderson et al. 1978a, b). The missing parameters for heavy LOHC materials were estimated by the UNIFAC model (Naseem et al. 2021).
Aslam et al. (2016) measured the hydrogen solubility in potential LOHC systems. They found the hydrogen solubility in hydrogenated forms of the LOHCs to be higher than in the dehydrogenated forms. For all three dehydrogenation technologies, ideal separation of hydrogen from the LOHC components was only considered. This study did not address the separation of LOHC components from each other due to their complexity. The technologies thus represent the same design level, allowing their comparison.
For the simulation of the technologies, plug flow reactors were used using reaction kinetics models available in the literature. The size of the reactors was adapted to the required residence times, the optimisation of which is not the aim of this study and will not be dealt with in detail in the presentation of the simulators.
The reactions were modelled as LHHW reactions, with the following general formula used in the process simulator:
Table 3 shows the \({\text{Kinetic factor}}\) data for each LOHC system. In all cases, the kinetic data refer to the use of a plug flow reactor and a Pt-Al2O3 catalyst. Details of the kinetics can be found in the literature referred.
The results are shown for every process: short process description, operation parameters, main operations, process flowsheet, and the CAPEX and OPEX values were calculated using Aspen Plus V.11 software (Aspen Technology 2019). The detailed material streams are presented as Supplementary material.
Naphthalene—decalin system
To determine the kinetics of the hydrogenation reaction, we have used the work of Lu et al. and Pt/Al2O3 (1 wt% Pt content) as the catalyst of interest since it has the lowest activation energy and several sources confirm the stability of this catalyst. Since naphthalene is solid under ambient conditions, the generally saturated hydrocarbon solvent is applied. The solvent used is here cyclohexane. The hydrogenation reaction can be divided into two steps. naphthalene is hydrogenated to tetralin in the first step and then further hydrogenated to decalin. The reaction rate equation can be written separately for the two steps (Lu et al. 2000).
To determine the kinetics of the dehydrogenation reaction, we used the work of Wang et al. who applied a 0.8 wt% Pt-0.3 wt% Sn/γ-Al2O3 catalyst. The catalyst results in high decalin conversion and naphthalene selectivity. Furthermore, if hydrogen is added to the feed stream, the catalyst lifetime can be increased; that is why we use hydrogen recirculation in dehydrogenation technology.
The reaction kinetics model used in this article is of the Hougen–Watson type, and the dehydrogenation reaction is broken down into several steps (Wang et al. 2008). Figure 3 shows the PFD of the hydrogenation technology of naphthalene. The operating pressure is 35 bar, while the reaction temperature is around 250 °C. The technology also contains a high-pressure separator and a distillation column. The excess hydrogen and the product can be recirculated if necessary. Stream S1 is the input hydrogen, while stream S7 is the recirculated hydrogen. Stream S14 is the input naphthalene, while streams S10 and S11 contain the recirculated solvent (cyclohexane). Stream S13 contains the product decalin. Table 4 shows the CAPEX and OPEX of the hydrogenation of naphthalene.
Figure 4 shows the flow diagram of the dehydrogenation technology of decalin. The reactor, in this case, operates at atmospheric pressure and 320 °C temperature. Stream S1 contains decalin, while stream S7 is the recirculated hydrogen to increase the catalyst's lifetime in the reactor. Stream S5 contains the product naphthalene and unconverted decalin, while S16 is the hydrogen stream.
Table 5 shows the various costs of the technology, i.e. installed and equipment cost, utility usage and costs per hour, in addition to the total capital and operating cost.
Toluene—methylcyclohexane system
The simulator was built by applying reaction kinetics models for LOHC technology using a toluene carrier available in the literature (Usman and Aslam 2014). For both hydrogenation and dehydrogenation, kinetics represented a Pt/Al2O3 catalyst. The Langmuir–Hinshelwood–Hougen–Watson Equation was used to describe the hydrogenation reaction of toluene. The catalyst and reaction conditions used for the dehydrogenation reaction were based on literature data (Usman and Aslam 2014).
Figure 5 shows the flow diagram of the hydrogenation technology of toluene. The reactor operates at a lower temperature and pressure than the naphthalene (20 bar, 175 °C). Stream S1 contains the input hydrogen, while stream S2 contains the input toluene. The product methylcyclohexane, contained in stream S8, can be separated by applying a flash drum at a lower temperature (20 °C). The excess hydrogen can be recirculated (S10). Stream S11 is a flush because stream S9 can be contained some impurities. Table 6 shows the CAPEX and OPEX of the hydrogenation of toluene.
Figure 6 shows the flow diagram of the dehydrogenation technology of methylcyclohexane (MCH). The reactor for the MCH dehydrogenation operates at 300 °C and 4 bar (required because of the catalyst). The obtained toluene and hydrogen can be separated using a flash drum at a lower temperature (25 °C). Stream S1 contains the input methylcyclohexane; stream S6 contains the product toluene and the unconverted MCH, while stream S11 contains the product hydrogen. Table 7 shows the CAPEX and OPEX of the dehydrogenation of methylcyclohexane.
Dibenzyltoluene—perhydro-dibenzyltoluene system
The simulator was built using reaction kinetics models available in the literature. For both hydrogenation and dehydrogenation, kinetics represented a 5% Pt/Al2O3 catalyst.
The scheme of hydrogenation and dehydrogenation of dibenzyltoluene (DBT) and perhydro-dibenzyltoluene (PDBT) is suggested by Roland (2019). The reaction kinetics model is of the Arrhenius type for hydrogenation and dehydrogenation.
Figure 7 shows the PFD of the hydrogenation technology of DBT. The process operates at mild temperatures and higher pressures than the toluene system (175 °C, 30 bar). The products can be separated using only a flash drum, which is favourable. Stream S1 contains the input hydrogen, while stream S2 contains the input DBT. Stream S12 contains the product PDBT and stream S10 contains the recirculated hydrogen. Stream S11 is a flush because stream S8 may contain some impurities. The impurities could come from the long-term application of LOHC material (the counterpart of the carrier can be a higher concentration). In this study, no impurities were considered since these calculations only serve design purposes.
Table 8 contains the various costs of the technology, i.e. installed and equipment cost, utility usage and costs per hour, in addition to the total capital and operating cost.
Figure 8 shows the flow diagram of the dehydrogenation technology of PDBT. The reactor for the PDBT dehydrogenation operates at 300 °C and atmospheric pressure. The obtained DBT and hydrogen can be separated using a flash drum at a lower temperature (35 °C). Stream S1 contains the input PDBT; stream S5 contains the product DBT and unconverted PDBT, while stream S14 contains the product hydrogen.
Table 9 contains the various costs of the technology, i.e. installed and equipment cost, utility usage and costs per hour, in addition to the total capital and operating cost.
Cost comparison of the three LOHC systems
The estimates for capital expenditures are summarised in Table 10. Finally, the operating expenditures are summarised in Table 11. In the tables, the toluene/methylcyclohexane carrier-based technology is highlighted in green, as this technology gives the best economic performance at this design level. In addition, the naphthalene/decalin system is a significant outlier in operating costs. This is due to the increased equipment size from the large amount of solvent required and the high energy demand of the separation system for solvent recovery. On this basis, it can be concluded that engaging in a technology that requires solvents is not economically viable.
Energy integration scenarios
In this section, two energy integration opportunities were also examined. The first case study (Case 1) was based on separate hydrogenation and dehydrogenation sites, while the second (Case 2) was based on one site utilization. The pinch method was used, which is the mainly used method in chemical engineering for the energy integration of plants. It is based on defining a minimal temperature approach and calculating the possible heat exchange between the hot and cold streams (Advanced Thermodynamics for Engineers (Second Edition) 2015).
The following approximations were used:
-
10 °C minimal temperature approach was used.
-
All the streams were preheated before the reaction.
-
The column in the case of naphthalene technology was not included in the integration.
-
The compressor heat exchangers were included in the integration.
-
The reaction heat was not included.
The background behind the first case result is the higher energy supply on one site and higher hydrogen supply on the other. The hydrogen can be transported between the sites with petroleum pipelines or ships. Scenario two focused on localized hydrogen and energy storage. The excess hydrogen can be stored in the LOHC and removed when necessary. In this case, the hydrogenation and dehydrogenation steps can be calculated together.
Results
The toluene hydrogenation process only contains one heating (if we consider the preheating, and one cooling, so there is no need to calculate energy integration. On the other hand, the decalin dehydrogenation technology was not integrated due to violating the pinch method rules. Table 12 shows the comparison of the different cases.
Comparing the initial cases, we can see that the highest energy demand process is the naphthalene system, primarily due to the necessary application of the solvent. A significant reduction can be reached in Case 1 in all LOHC carriers. Further reduction only happened in the case of the toluene system, which we also show in Fig. 9.
The best heating reduction percentage can be seen in naphthalene, while the best cooling reduction percentage can be seen in toluene as we can see in Fig 9a. In case of the naphthalene the majority of the reduction can be explained by the usage of the solvent. The mass-flow are higher in this case as well as the corresponding enthalpies, so there are more opportunity for energy integration. However, if we look at Fig. 9b, it can be seen that the energy demand of the integrated toluene–MCH system is the best of the three cases. The toluene-MCH system has the lowest base-case energy usage from the three discussed scenarios, which can be lowered further with energy integration.
Discussion of EU and Hungarian policies and the contribution of the present work
As stated before, the Aquamarine project was organized according to the REPowerEU strategy. Therefore, in this section, we collected the main points of the REPowerEU and Hungarian Hydrogen strategy (kormany.hu, 2023) and the present work's contributions.
The REPowerEU strategy has three main actions, clean energy, clean industry and saving energy. One of the main cornerstones of the clean transition is the photovoltaic energy-based green hydrogen production, which could serve as an alternate fuel for the transportation and chemical industries.
The Hungarian Hydrogen Strategy aims to introduce clean hydrogen and hydrogen technologies and establish the background infrastructure for the hydrogen industry. One of the primary sources of hydrogen will be electrolysis (240 MW electrolyzer capacity), and LOHC is identified as one of the fields with the most innovation potential.
Our study is performed within the framework of the Aquamarine project. Based on our findings, the next step will be an industrial implementation, contributing to the clean energy and clean industry goal by utilizing electrolysis. Besides, the manuscript also discusses energy integration opportunities, contributing to the energy-saving action.
Conclusion
Hydrogen can be one of the most important compounds in the future, and it is the flagship of the European Union Green Initiative. So the research and development of hydrogen-related technologies are crucial to ensure our survival. One of the most important issues related to hydrogen is the storage and transportation of hydrogen. This study investigated the application of three different systems as liquid organic hydrogen carrier (LOHC) systems. After a comprehensive literature study of carriers, processes and catalysts, the selected systems were compared using specific and detailed flowsheeting simulations using kinetic data available in the literature. The main focus was to calculate CAPEX and OPEX, and based on the results, the toluene-methylcyclohexane systems were found to be the most promising for LOHC application. In addition, two energy integration scenarios were examined for the three LOHC systems. The results show a lower energy demand compared to the initial (without energy integration) systems, in the case of a toluene system with a reduction by 70% of heating and 45% of cooling.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ahn C-I, Kwak Y, Kim A-R, Jang M, Badakhsh A, Cha J, Kim Y, Jo YS, Jeong H, Choi SH, Nam SW, Yoon CW, Sohn H (2022) Dehydrogenation of homocyclic liquid organic hydrogen carriers (LOHCs) over Pt supported on an ordered pore structure of 3-D cubic mesoporous KIT-6 silica. Appl Catal B 307(2022):121169
Asif F, Hamayun MH, Hussain M, Hussain A, Maafa IM, Park Y (2021) Performance analysis of the perhydro-dibenzyl-toluene dehydrogenation system — a simulation study. Sustainability 13(11):6490. https://doi.org/10.3390/su13116490
Aspen Technology (2019) Aspen plus getting started guide, version 11.1, Cambridge
Aslam R, Müller K, Müller M, Koch M, Wasserscheid P, Arlt W (2016) (2015) Measurement of hydrogen solubility in potential Liquid Organic Hydrogen Carriers. Chem Eng Data 61(1):643–649
Bourane A, Elanany M, Pham TV, Katikaneni SP (2016) An overview of organic liquid phase hydrogen carriers. Int J Hydrog Energy 41(48):23075–23091
Brückner N, Obesser K, Bösmann A, Teichmann D, Arlt W, Dungs J, Wasserscheid P (2014) Evaluation of industrially applied heat-transfer fluids as liquid organic hydrogen carrier systems. Chemsuschem 7(1):229–235. https://doi.org/10.1002/cssc.201300426
Bulgarin A, Jorschick H, Preuster P, Bösmann A, Wasserscheid P (2020) Purity of hydrogen released from the Liquid Organic Hydrogen Carrier compound perhydro dibenzyltoluene by catalytic dehydrogenation. Int J Hyd En 45:712–720
Cacciola G, Giordano N, Restuccia G (1984) Cyclohexane as a liquid phase carrier in hydrogen storage and transport. Int J Hydrog Energy 9(5):411–419
Castano P, Arandes JM, Pawelec B, Fierro JLG, Gutierrez A, Bilbao J (2007) (2007) Kinetic model discrimination for toluene hydrogenation over noble-metal-supported catalysts. Ind Eng Chem Res 46:7417–7425
CEEnergyNews (2021) Hungarian Gas Storage's Aquamarine Project to play an active role in the decarbonization process, March 24, 2021
Cho J-Y, Kim H, O J-E, Park BY (2021) Recent advances in homogeneous/heterogeneous catalytic hydrogenation and dehydrogenation for potential liquid organic hydrogen carrier (LOHC) systems. Catalysts 2021(11):1497
Frauwallner M-L, López-Linares F, Lara-Romero J, Scott CE, Ali C, Hernandez E, Pereira-Almao P (2011) Toluene hydrogenation at low temperature using a molybdenum carbide catalyst. Appl Catal A 394(1–2):62–70. https://doi.org/10.1016/j.apcata.2010.12.024
Hodoshima S, Takaiwa S, Shono A, Satoh K, Saito Y (2005) (2005) Hydrogen storage by decalin/naphthalene pair and hydrogen supply to fuel cells by use of superheated liquid-film-type catalysis. App Cat a: Gen 283:235–242
Huang T-C, Kang B-C (1995) Naphthalene hydrogenation over Pt/Al2O3 catalyst in a trickle bed reactor. Ind Eng Chem Res 34:2349–2357
Kim K, Oh J, Kim TW, Park JH, Han JW, Suh Y-W (2017) (2017) Different catalytic behavior of Pd and Pt metals in decalin dehydrogenation to naphthalene. Catal Sci Tech 7:3728–3735
Kirumakki SR, Shpeizer BG, Sagar GV, Chary KVR, Clearfield A (2006) (2006) Hydrogenation of Naphthalene over NiO/SiO2-Al2O3 catalysts: structure-activity correlation. J Catal 242:319–331
Klvana D, Chaouki J, Kusohorsky D, Chavarie C (1988) Catalytic storage of hydrogen: hydrogenation of toluene over a nickel/silica aerogel catalyst in integral flow conditions. Appl Catal 42(1988):121–130
https://kormany.hu/dokumentumtar/magyarorszag-nemzeti-hidrogenstrategiaja. Accessed Jan 2023
Kustov LM, Kalenchuk AN (2002) (2022) The role of the metal in the catalytic reactions of hydrogenation-dehydrogenation of polycyclic hydrocarbons for hydrogen storage. Metals 2022:12
Lu CM, Lin YM, Wang I (2000) Naphthalene hydrogenation over Pt/TiO2–ZrO2 and the behavior of strong metal–support interaction (SMSI). Appl Catal A 198(1–2):223–234
Maddah HA (2018) A comparative study between propane dehydrogenation (PDH) technologies and plants in Saudi Arabia. American Scientific Research Journal for Engineering, Technology and Sciences
Makaryan IA, Sedov IV (2021) Hydrogenation/dehydrogenation catalysts for hydrogen storage systems based on liquid organic carriers (a review). Pet Chem 61(9):977–988
Makepeace JW, He T, Weidenthaler C, Jensen TR, Chang F, Vegge T et al (2019) Reversible ammonia-based and liquid organic hydrogen carriers for high-density hydrogen storage: recent progress. Int J Hydrog Energy 44(15):7746–7767
Meille V, Pitault I (2021) Liquid organic hydrogen carriers or organic liquid hydrides: 40 years of history. Reactions 2(2):94–101
Modisha PM, Ouma CNM, Garidzirai R, Wasswescheid P, Bessarabov D (2019) The prospect of hydrogen storage using liquid organic hydrogen carriers. Energy Fuels 33(4):2778–2796
Morris L et al (2019) A manganese hydride molecular sieve for practical hydrogen storage under ambient conditions. Energy Environ Sci 12:1580–1591
Müller K, Skeledzic T, Wasserscheid P (2021) Strategies for low-temperature liquid organic hydrogen carrier dehydrogenation. Energy Fuels 35:10929–10936
Naseem M, Usman M, Lee S (2021) A parametric study of dehydrogenation of various Liquid Organic Hydrogen Carrier (LOHC) materials and its application to methanation process. Int J Hydrog En 46:4100–4115
Nawaz Z (2015) Light alkane dehydrogenation to light olefin technologies: a comprehensive review. Rev Chem Eng 31:413–436
Niermann M, Beckendorff A, Kaltschmitt M, Bonhoff K (2019a) Liquid Organic Hydrogen Carrier (LOHC) Assessment based on chemical and economic properties. Int J Hydrog Energy 44(13):6631–6654. https://doi.org/10.1016/j.ijhydene.2019.01.199
Niermann M, Drünert S, Kaltschmitt M, Bonhoff K (2019b) Liquid organic hydrogen carriers (LOHCs)–techno-economic analysis of LOHCs in a defined process chain. Energy Environ Sci 12(1):290–307
Olah GA, Goeppert A, Prakash GS (2018) Beyond oil and gas: the methanol economy. Wiley
Park S, Naseem M, Lee S (2021) Experimental assessment of perhydro-dibenzyltoluene dehydrogenation reaction kinetics in a continuous flow system for stable hydrogen supply. Materials 14:7613
Rao PC, Yoo M (2020) Potential liquid-organic hydrogen carrier (LOHC) systems: a review on recent progress. Energies 13:6040
Rautanen PA, Lylykangas MS, Aittamaa JR, Krause AOI (2002) Liquid-phase hydrogenation of naphthalene and tetralin on Ni/Al2O3: kinetic modeling. Ind Eng Chem Res 41:5966–5975
REPowerEU Strategy (2019) https://commission.europa.eu/strategy-and-policy/priorities-2019-2024/european-green-deal/repowereu-affordable-secure-and-sustainable-energy-europe_en. Accessed 04 Jan 2023
Sanfilippo D, Rylander PN (2012) Hydrogenation and dehydrogenation. In: Ullmann's encyclopedia of industrial chemistry. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, p 452
Scherer GWH, Newson E, Wokaun A (1999) Economic analysis of the seasonal storage of electricity with liquid organic hydrides. Int J Hydrog Energy 24(12):1157–1169
Sebastián D, Bordejé EG, Calvillo L, Lázaro MJ, Moliner R (2008) Hydrogen storage by decalin dehydrogenation/naphthalene hydrogenation pair over platinum catalysts supported on activated carbon. Int J Hydrog Energy 33:1329–1334
Shi L, Qi S, Qu J, Che T, Yi C, Yang B (2019) Integration of hydrogenation and dehydrogenation based on dibenzyltoluene as liquid organic hydrogen energy carrier. Int J Hydrogen Energy 44(11):5345–5354
Stoffels MA (2020) Technology trends of catalysts in hydrogenation reactions: a patent landscape analysis. Adv Synth Catal 362:1258–1274
Taube M (1985) A prototype truck powered by hydrogen from organic liquid hydrides. Int J Hydrog Energy 10(9):595–599
Thybaut W-J (2002) Hydrogenation kinetics of toluene on Pt/ZSM-22. Chem Eng J 90(1–2):117–129
U.S. Department of Energy (2021) Office of energy efficiency and renewable energy. https://www.energy.gov/eere/fuelcells/hydrogen-storage. Accessed 21 Jun 2021
Usman MR, Aslam R (2014) Dehydrogenation of methylcyclohexane for on-board hydrogen use: initial rate kinetics over 1.0 wt% Pt/γ-Al2O3. Arab J Sci Eng 39(2):615–620
Usman MR, Aslam R, Alotaibi F (2011) Hydrogen storage in a recyclable organic hydride: kinetic modeling of methylcyclohexane dehydrogenation over 1.0 wt% Pt/θ-Al2O3. Energy Sour, Part a: Recov, Util, Environ Eff 33(24):2264–2271. https://doi.org/10.1080/15567036.2011.585388
Usman MR, Aslam R, Alotaibi F (2015) Dehydrogenation–hydrogenation of methylcyclohexane-toluene system on 1.0 wt% Pt/zeolite beta catalyst. Progr React Kinet Mech 40(4):353–366. https://doi.org/10.3184/146867815X14413752286029
Wang B, Goodman DW, Froment GF (2008) Kinetic modeling of pure hydrogen production from decalin. J Catal 253(2):229–238
Wunsch A, Berg T, Pfeifer P (2020) Hydrogen production from the LOHC perhydrodibenzyl-toluene and purification using a 5 μm PdAg-membrane in a coupled microstructured system. Materials. https://doi.org/10.3390/ma13020277
Acknowledgements
This work has been supported by the project Aquamarine—Hydrogen-based energy storage solution at Hungarian Gas Storage Ltd. funded by the Ministry of Technology and Industry under Grant Agreement No. 2020-3.1.2-ZFR-KVG-2020-00001. The authors acknowledge the cooperation of András Bartha, János György Kiss, Richárd Varga.
Funding
Open access funding provided by University of Pannonia. The funding was provided by Ministry of Technology and Industry (Grant Nos. 2020-3.1.2-ZFR-KVG-2020-00001.
Author information
Authors and Affiliations
Contributions
Ágnes Bárkányi organisation of the first draft, simulation, preparation of sections "Liquid organic hydrogen carrier (LOHC) technology, Hydrogenation and dehydrogenation at the industrial scale, Techno-economic evaluation of promising LOHC systems and Energy integration scenarios", Bálint Levente Tarcsay introduction, simulation László Lovas energy integration, simulation Tamás Mérő manuscript final review, funding acquisition Tibor Chován introduction, manuscript final review, simulation Attila Egedy energy integration, policy section, preparation of sections "Discussion of eu and hungarian policies and the contribution of the present work and Conclusion" All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bárkányi, Á., Tarcsay, B.L., Lovas, L. et al. Future of hydrogen economy: simulation-based comparison of LOHC systems. Clean Techn Environ Policy 26, 1521–1536 (2024). https://doi.org/10.1007/s10098-023-02528-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-023-02528-w