Abstract
The bioethanol produced using fermentation is obtained at a very low concentration. To be used as fuel in gasoline blends, a higher than 99% weight purity is required. Therefore, highly demanding energy separation and purification processes are used. Given this fact, this work presents, via simulation, an economic and environmental comparison of alternative methods for ethanol dehydration based on ethylene oxide/propylene oxide hydration and azeotropic distillation with benzene and cyclohexane. These reactive methods consist of conventional reaction separation processes and intensified processes such as reactive distillation (RD), both coupled with organic Rankine cycles, offering an additional value-added product (ethylene glycol or propylene glycol), electric power generation and the capacity to reduce the global process steps in anhydrous ethanol production. The results indicate that reactive dehydration, specifically the reactor–separator process using propylene oxide at a low ethanol concentration (dilution effect of the fermenter output), is the best economic and environmental option among all the processes studied for anhydrous ethanol production. Likewise, reactive dehydration yields a higher net profit, ethanol yield and ethanol purity and lower CO2 emissions than azeotropic distillation.
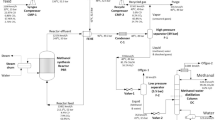
Graphic abstract





Similar content being viewed by others
Abbreviations
- Np:
-
Net profit
- As:
-
Annual sales
- Rm:
-
Raw material
- Tc:
-
Technology cost
- Mfo:
-
Mass flow output
- Sp:
-
Sales price
- Mfe:
-
Mass flow input
- Rmp:
-
Raw material price
- Uc:
-
Utility cost
- Ec:
-
Equipment cost
- Amp:
-
Azeotropic mixture price
- Emfe:
-
Ethanol mass fraction equivalent
- Aep:
-
Anhydrous ethanol price
- Te:
-
Total CO2 emissions
- Ce:
-
CO2 emissions
- RDEO2080:
-
Reactive distillation using ethylene oxide at 20% mol water and 80% mol ethanol
- RDEO9604:
-
Reactive distillation using ethylene oxide at 96% mol water and 4% mol ethanol
- RDPO2080:
-
Reactive distillation using propylene oxide at 20% mol water and 80% mol ethanol
- RDPO9604:
-
Reactive distillation using propylene oxide at 96% mol water and 4% mol ethanol
- RSEO2080:
-
Reactor–separator process using ethylene oxide at 20% mol water and 80% mol ethanol
- RSEO9604:
-
Reactor–separator process using ethylene oxide at 96% mol water and 4% mol ethanol
- RSPO2080:
-
Reactor–separator process using propylene oxide at 20% mol water and 80% mol ethanol
- RSPO9604:
-
Reactor–separator process using propylene oxide at 96% mol water and 4% mol ethanol
- AZBE2080:
-
Azeotropic distillation using benzene at 20% mol water and 80% mol ethanol
- AZBE9604:
-
Azeotropic distillation using benzene at 96% mol water and 4% mol ethanol
- AZCH2080:
-
Azeotropic distillation using cyclohexane at 20% mol water and 80% mol ethanol
- AZCH9604:
-
Azeotropic distillation using cyclohexane at 96% mol water and 4% mol ethanol
- R245FA:
-
1,1,1,3,3-Pentafluoropropane
- ORC:
-
Organic Rankine cycle
- H :
-
Working hours (8000 h/year)
- i :
-
Interest rate (0.1%)
- k f :
-
Factor to annualize the investment
- U :
-
Utilities
- m:
-
Utilities
- q:
-
Equipment
- j:
-
Raw material stream
- p:
-
Product stream
- n :
-
Life period of each technology (20 years)
References
AFDC (2018) Gobal ethanol production. Retrieved from https://afdc.energy.gov/data/. Accessed 15 Dec 2018
Aguilar-Sánchez P, Navarro-Pineda FS, Sacramento-Rivero JC, Barahona-Pérez LF (2018) Life-cycle assessment of bioethanol production from sweet sorghum stalks cultivated in the state of Yucatan, Mexico. Clean Technol Environ Policy 20(7):1685–1696. https://doi.org/10.1007/s10098-017-1480-4
Álvarez del Castillo-Romo A, Morales-Rodriguez R, Román-Martínez A (2018) Multiobjective optimization for the socio-eco-efficient conversion of lignocellulosic biomass to biofuels and bioproducts. Clean Technol Environ Policy 20(3):603–620. https://doi.org/10.1007/s10098-018-1490-x
An W, Lin Z, Chen J, Zhu J (2014) Simulation and analysis of a reactive distillation column for removal of water from ethanol–water mixtures. Ind Eng Chem Res 53:6056–6064. https://doi.org/10.1021/ie403906z
Avilés-Martínez A, Medina-Herrera N, Jiménez-Gutiérrez A, Serna-González M, Castro-Montoya AJ (2015) Risk analysis applied to bioethanol dehydration processes: azeotropic distillation versus extractive distillation. Comput Aided Chem Eng. https://doi.org/10.1016/B978-0-444-63577-8.50151-0
Baeyens J, Kang Q, Appels L, Dewil R, Lv Y, Tan T (2015) Challenges and opportunities in improving the production of bio-ethanol. Prog Energy Combust Sci 47:60–88. https://doi.org/10.1016/j.pecs.2014.10.003
Cardona CA, Sánches OJ, Gutiérrez LF (2010) Process synthesis for fuel ethanol production. Focus Catal. https://doi.org/10.1016/S1351-4180(10)70424-4
Delgado JA, Águeda VI, Uguina MA, Sotelo JL, García A, Brea P, García-Sanz A (2013) Separation of ethanol–water liquid mixtures by adsorption on a polymeric resin Sepabeads 207®. Chem Eng J 220:89–97. https://doi.org/10.1016/j.cej.2013.01.057
Dias MOS, Junqueira TL, Rossell CEV, MacIel Filho R, Bonomi A (2013) Evaluation of process configurations for second generation integrated with first generation bioethanol production from sugarcane. Fuel Process Technol 109:84–89. https://doi.org/10.1016/j.fuproc.2012.09.041
Esquivel-Patiño GG, Serna-González M, Nápoles-Rivera F (2017) Thermal integration of natural gas combined cycle power plants with CO2 capture systems and organic Rankine cycles. Energy Convers Manag 151(August):334–342. https://doi.org/10.1016/j.enconman.2017.09.003
Gangadwala J, Radulescu G, Kienle A, Steyer F, Sundmacher K (2008) New processes for recovery of acetic acid from waste water. Clean Technol Environ Policy 10(3):245–254. https://doi.org/10.1007/s10098-007-0101-z
Huang K, Nakaiwa M (2006) Reactive distillation design with considerations of heats of reaction. AIChE J 52:2518–2534. https://doi.org/10.1002/aic.10885
Kiss AA, Suszwalak DJPC (2012) Enhanced bioethanol dehydration by extractive and azeotropic distillation in dividing-wall columns. Sep Purif Technol 86:70–78. https://doi.org/10.1016/j.seppur.2011.10.022
Kozlovsky IA, Kozlovsky RA, Koustov AV, Makarov MG, Suchkov JP, Shvets VF (2002) Kinetics and products distribution of selective catalytic hydration of ethylene- and propylene oxides in concentrated aqueous solutions. Org Process Res Dev 6(5):660–664. https://doi.org/10.1021/op010099+
Lai H, Lin Y, Tu C (2013) Isobaric vapour-liquid equilibria for the ternary system of (ethanol + water + 1,3-propanediol) and three constituent binary systems at P = 101.3 kPa. J Chem Thermodyn. https://doi.org/10.1016/j.jct.2013.08.020
Martinez-Gomez J, Nápoles-Rivera F, Ponce-Ortega JM, El-Halwagi MM (2017) Optimization of the production of syngas from shale gas with economic and safety considerations. Appl Therm Eng 110:678–685. https://doi.org/10.1016/j.applthermaleng.2016.08.201
Orosz MS, Mueller A, Quolin S, Hemond H (2010) Small scale solar orc system for distributed power. In: Conference SolarPaces 2009, Berlin, Germany, vol 1, no 3
Oseguera-Villaseñor I, Martínez-Rodríguez G, Barroso-Muñoz FO, Segovia-Hernández JG, Hernández S (2018) Multiplicities in dividing wall distillation columns in the purification of bioethanol: energy considerations. Clean Technol Environ Policy 20(7):1631–1637. https://doi.org/10.1007/s10098-017-1415-0
Palomino A, Parientes DA, Gomez H, Paucar P (2013) Modelling ethanol-water pressure swing distillation in an structured packed bed column, Peruvian J Chem Eng 16(1):75–84
Press AIN, Balat M, Balat H, Öz C (2008) Progress in bioethanol processing. Prog Energy Combust Sci 34:551–573. https://doi.org/10.1038/ismej.2010.175
Santarelli L, Saxe M, Gross C, Surget A, Dulawa S, Weisstaub N, Hen R (2014) The path forward for biofuels. Science 301(5634):805–809. https://doi.org/10.1007/s13398-014-0173-7.2
Schefflan R (2011) Teach yourself the basics of Aspen Plus. Wiley, New York. https://doi.org/10.1002/9780470910061
Singh N, Prasad R (2011) Fuel grade ethanol by diffusion distillation: an experimental study. J Chem Technol Biotechnol 86(5):724–730. https://doi.org/10.1002/jctb.2579
Singh A, Rangaiah GP (2017) Review of technological advances in bioethanol recovery and dehydration. Ind Eng Chem Res 56(18):5147–5163. https://doi.org/10.1021/acs.iecr.7b00273
Tavan Y, Hosseini SH (2013) A novel integrated process to break the ethanol/water azeotrope using reactive distillation—part I: parametric study. Sep Purif Technol 118:455–462. https://doi.org/10.1016/j.seppur.2013.07.036
Tchanche BF, Lambrinos G, Frangoudakis A, Papadakis G (2011) Low-grade heat conversion into power using organic Rankine cycles—a review of various applications. Renew Sustain Energy Rev 15(8):3963–3979. https://doi.org/10.1016/j.rser.2011.07.024
Wang XD, Zhao L, Wang JL, Zhang WZ, Zhao XZ, Wu W (2010) Performance evaluation of a low-temperature solar Rankine cycle system utilizing R245fa. Sol Energy 84(3):353–364. https://doi.org/10.1016/j.solener.2009.11.004
Wang X, Levy EK, Pan C, Romero CE, Banerjee A, Rubio-Maya C, Pan L (2019) Working fluid selection for organic Rankine cycle power generation using hot produced supercritical CO2 from a geothermal reservoir. Appl Therm Eng 149(December 2018):1287–1304. https://doi.org/10.1016/j.applthermaleng.2018.12.112
Acknowledgements
The authors acknowledge the financial support from the Mexican Council for Science and Technology (CONACyT) and the Scientific Research Council of the Universidad Michoacana de San Nicolás de Hidalgo.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Guzmán-Martínez, C.E., Castro-Montoya, A.J. & Nápoles-Rivera, F. Economic and environmental comparison of bioethanol dehydration processes via simulation: reactive distillation, reactor–separator process and azeotropic distillation. Clean Techn Environ Policy 21, 2061–2071 (2019). https://doi.org/10.1007/s10098-019-01762-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-019-01762-5