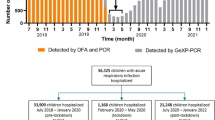
Abstract
Purpose
We aimed to determine the changes in the frequency of respiratory pathogens and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) during containment of the 2019 coronavirus disease pandemic and elucidate the epidemiological interference that may have occurred after lifting pandemic measures.
Methods
A total of 4,770 Nasopharyngeal swab samples were collected from children with ARTIs from the First People’s Hospital of Yunnan Province between January 2022 and December 2023 and subjected to nucleic acid testing for 13 types of respiratory pathogens and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
Results
The frequency of pathogens among children from 2020 to 2022 was in the following order: HRV > Mp > HADV > H3N2 > HMPV and HRV > HRSV > HPIV > H1N1 > H3N2. In weeks 1 to 3 of 2023, the frequency of pathogens significantly declined, and then H1N1 rebounded significantly in 2023. HRV, HRSV, and H3N2 showed a shift in the season of high frequency. Patterns of multi-pathogen infections were more complex in 2023 than in 2022, with HRV having a higher frequency and co-infection rate than other pathogens. These changes may have been associated with interference caused by the resurgence of SARS-CoV-2 prevalence, in addition to being influenced by changes in pandemic containment and lifting measures.
Conclusions
The frequency rate of common respiratory pathogens among children was not significantly different and remained high. The study findings help elucidate the aforementioned unique historical period and effectively control respiratory tract infections to reduce the harm to pediatric health caused by respiratory pathogens.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute respiratory infections (ARIs) are infectious diseases with high incidence and mortality rates worldwide [1, 2]. Respiratory pathogens are the main cause of ARTIs in children, with 90% of respiratory tract infections (RTIs) in infants and young children caused by respiratory viruses [3]. In China, viral infection rates of up to 46.9% have been reported in children aged ≤ 5 years [2]. Innate and adaptive immunity plays important roles in restricting respiratory infections [4, 5]. Children are vulnerable to different types of infections, especially ARIs, because their immune system are not fully developed, leading to attenuated resistance and increased susceptibility to infections [6,7,8]. Respiratory viruses have high transmission abilities and frequency rates among children [9,10,11]. Additionally, co-infections with multiple pathogens are very common and prone to misdiagnoses and missed diagnoses by clinicians [12].
Following the official designation of coronavirus disease (COVID-19) as a global pandemic by the World Health Organization on March 11, 2020, nonpharmaceutical interventions, such as face mask-wearing, hand hygiene, social distancing, quarantine, travel restrictions, and school closures, were implemented by countries worldwide to control the pandemic [13]. These policies have evolved over the course of the COVID-19 pandemic, with regional and national differences in their ongoing use. The Chinese government established a prevention and control policy involving restricting the flow of people since the outbreak in Hubei, China, in January 2020 [14, 15]. From the closure of Wuhan in 2020 to the dynamic zero COVID policy, precise prevention and control has prevented the spread of new coronavirus strains [16, 17]. With the weakening of the pathogenicity of COVID-19, the popularization of vaccination, and the accumulation of experience in prevention and control, the transmission of respiratory pathogens among children has to a certain extent been curbed [13]. On December 7, 2022, China adjusted and optimized its pandemic prevention and control strategies [15]. Afterwards, Omicron rapidly spread throughout China, and cases have peaked. The complete lifting of pandemic containment and mitigation measures has led to a sharp decline in the use of face masks, resumption of public events, and reopening of public places and schools. Children have since faced the transition from a period of low-pathogen exposure to a restriction-free period. With the increase in opportunistic infections, whether the frequency of respiratory pathogens has undergone a rapid resurgence is unclear.
In the present study, the city of Kunming, China, was selected as the study area for the investigation of changes in the frequency of common respiratory pathogens among children in the post-pandemic era. Kunming is on the southwestern border of China and has a resident population of over 8.6 million. It serves as China’s key gateway to Southeast Asia and South Asia and possesses unique climatic and population distribution characteristics. However, its healthcare infrastructure is relatively underdeveloped, resulting in heavier disease burdens in the region. To our knowledge, relevant studies have not been conducted in this region. Therefore, the objectives of this study were to determine changes in the circulation of respiratory pathogens and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) during and after the containment of the 2019 coronavirus disease pandemic and to assess epidemiological interference that may have occurred after pandemic measures were lifted.
Materials and methods
Study population
The study population included individuals under the age of 18 recruited between January 2022 and December 2023 from the First People’s Hospital of Yunnan Province in China. The inclusion criteria were as follows: (1) age < 18 years; (2) no clinical treatment administered before sample collection; and (3) a diagnosis of ARIs. ARIs were defined as illnesses with (1) onset within 7 d; (2) at least two respiratory symptoms from the following list: nasal discharge, nasal obstruction, sneezing, sore throat, cough, malaise, chills, and headache; and (3) a Jackson score of ≥ 2 [18]. The exclusion criteria were as follows: (1) concomitant major diseases; (2) incomplete clinical data; or (3) presence of immunodeficiency.
Before commencement, the study was approved by the institutional review board of the First People’s Hospital of Yunnan Province (Approval Number: KHLL2023-KY055), and informed consent was obtained from the parents or guardians of all participants. The parents or guardians were informed of the laboratory results of pathogen detection.
Sample collection
For each participant, one nasopharyngeal swab sample was taken on the day of the clinic visit or the day after and stored at 4 °C in a sterile sample tube containing cell preservation solution (HEALTH Gene Technologies Co., Ltd., Ningbo, China). All testing procedures were completed within 24 h of sample collection.
Nucleic acid extraction
Nucleic acid was extracted from clinical samples using a Nucleic Acid Extraction or Purification Kit (HEALTH Gene Technologies Co., Ltd., Ningbo, China), and the extracted nucleic acid was stored at − 80 °C. An automated nucleic acid extraction system (Smart LabAssist-32; Taiwan Advanced Nanotech Inc, Taiwan) was used.
Pathogen detection
Detection of respiratory pathogens was performed using a Multiple Detection Kit for Thirteen Respiratory Pathogens (13× kit, HEALTH Gene Technologies Co., Ltd.). Instruments used to detect respiratory pathogens included a PCR thermal cycler A300 (Hangzhou LongGene Scientific Instruments Co, Ltd, China) and Applied Biosystems 3500 Dx analyzer (Thermo Fisher Scientific, USA). The detected pathogens included the following: Influenza virus types A (InfA), InfA-H1N1pdm09, InfA-H3N2, influenza virus types B (InfB), human respiratory syncytial virus (HRSV), human parainfluenza virus (HPIV), human coronavirus (HCOV), human rhinovirus (HRV), human metapneumovirus (HMPV), bocavirus (Boca), human adenovirus (HADV), chlamydia (Ch: including Chlamydia trachomatis and Chlamydia pneumoniae), and Mycoplasma pneumoniae (Mp). The SARS-CoV-2 detection kits used in this study (50× kit) were purchased from Shanghai BioGerm Medical Biotechnology Co., Ltd. (Shanghai, China).
Statistical analysis
The results were comparatively analyzed based on the period and age group. The cases selected for the study were divided into 2022 and 2023 groups based on the time point of sample collection (before or after the lifting of pandemic containment and mitigation measures), and the two groups were further divided into five subgroups based on age ranges: 2022: 0–28 d, 1–12 months, 1–3 years), 4–6 years, and > 7 years; 2023: 0–28 d, 1–12 months, 1–3 years, 4–6 years, and > 7 years. A descriptive analysis was performed using absolute and relative frequencies (percentages) of the variables studied. Statistical analysis was performed using SPSS statistical software (version 26.0, SPSS, Inc., Chicago, USA) [19, 20]. Plotting was performed using OriginPro 2021. Count data are expressed as the number of cases and percentages, and the χ2 test was used to compare groups. P < 0.05 was considered a statistically significant difference, with a test level α = 0.05 (bilateral).
Results
Of the 4,770 cases that met the required criteria, 1,669 (959 males and 710 females) were included in the study in 2022, and 3,101 (1,786 males and 1,315 females) in the study in 2023. The cases studied in 2022 were distributed as follows according to age groups: 0–28 d (20.00 ± 2.94; 32 cases), 1–12 months (5.60 ± 3.15; 257 cases), 1–3 years (2.10 ± 0.85; 426 cases), 4–6 years (4.79 ± 0.78; 351 cases), and > 7 years (10.39 ± 2.81; 603 cases); 2023: 0–28 d (16.14 ± 4.39; 27 cases), 1–12 months (6.03 ± 3.22; 531 cases), 1–3 years (2.19 ± 0.85; 867 cases), 4–6 years (4.73 ± 0.81; 570 cases), and > 7 years (10.98 ± 3.36; 1,106 cases) (Table S3).
In 2022, the frequency of positivity was 52.67%, and 2023, 52.34%. The difference in the frequency rate between the 2 years was not significant (P > 0.05), nor were the differences in single-pathogen infections and multi-pathogen infections before and after the lifting of pandemic containment and mitigation measures (P > 0.05). In 2022, the following pathogens had the highest frequency rates (in descending order): HRV > Mp > HADV > InfA-H3N2 > HMPV, whereas InfA-H1N1pdm09 had the lowest frequency rate. In 2023, the pathogens with the highest frequency rates were HRV > HRSV > HPIV > InfA-H1N1pdm09 > InfA-H3N2, whereas Ch had the lowest frequency rate. The difference in frequency rates between the 2 years was significant for HRV, HPIV, HRSV, InfA-H1N1pdm09, HADV, InfB, and Mp (P < 0.05). Among all age groups, the group aged 1–3 years had the highest infection rates, followed by the group aged > 7 years, whereas patients aged 0–28 d had the lowest infection rates. Boys had higher frequency rates than girls (Table 1; Fig. 1).
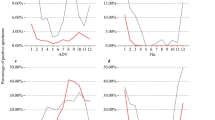
HRV had the highest frequency rate among all pathogens. In January 2023, all pathogens exhibited an interruption in frequency except for HRSV, HMPV, and Mp, which had low frequency rates. The frequency of InfA-H1N1pdm09 experienced an interruption in 2022 but rebounded from March to June 2023. Compared to 2022, the frequency rates of HADV and Mp in 2023 were reduced, with significant differences in January, March, April, June, and July (P > 0.05). The season of high frequency of HRV shifted from late spring to early summer in 2022 to late autumn to early winter in 2023. For HRSV and InfA-H3N2, a shift occurred from late winter to early spring to late summer to early winter. The changes in the season of high frequency of all other pathogens were not significant (Fig. 2).
Frequency trends and changes in the seasons of high frequency in 2022 and 2023. The length of the colored bars indicates pathogen frequency. *P < 0.05, where two groups are significantly different. HRV had the highest frequency rate among all pathogens. Compared to those in 2022, the frequency rates of HRV in 2023 were reduced in January, March, April, May, June, July, and August (P > 0.05). In January 2023, all pathogens exhibited an interruption in frequency except for HRSV, HMPV, and Mp. The frequency of InfA-H1N1 rebounded significantly in 2023, and RSV increased in February to October (P > 0.05). Conversely, the frequency rates of HADV and Mp in 2023 were reduced, with significant differences in January, March, April, June, and July (P > 0.05)
Most pathogens were prevalent in children aged 1–3 years and exhibited obvious umbrella-shaped distribution characteristics with non-significant differences in frequency rates before and after the lifting of pandemic containment and mitigation measures. The InfA-H1N1pdm09 infection rate increased with an increase in patient age after the rebound of InfA-H1N1pdm09 frequency in 2023, and the Mp infection rate was also highest in older children. HRSV was the most common respiratory virus among children aged ≤ 2 years. Except for Mp and InfA-H1N1pdm09, all other pathogens exhibited a significant decrease in the frequency rates in older children (P > 0.05) (Fig. 3).
Age distribution of pathogens in 2022 and 2023. The frequency of most pathogens was highest in children aged 1–3 years and lowest in those aged 0–28 days. Obvious umbrella-shaped distribution characteristics were observed and with non-significant differences in overall frequency from 2022 to 2023 (P > 0.05). HRSV was most common in children under 2 years of age, and Mp was predominant in the > 7 years group. However, except for Mp and InfA-H1N1, all other pathogens decreased in frequency in the > 7 years group in 2023 (P > 0.05)
SARS-CoV-2 was undetected from week 1 to week 49 in 2022. SARS-CoV-2 was detected from week 50 of 2022 until the last week of 2023, with the highest frequency rate being 43.75% in week 52 of 2022. The second highest frequency was 42.11% in week 16 of 2023. The frequency sharply declined from week 44 (Fig. 4 and Table S4).
Weekly frequency of 13 respiratory pathogens and SARS-COV-2 in 2022 and 2023. The frequency of COVID-19 sharply increased from week 50 of 2022 and continued to be an epidemic. Conversely, common respiratory pathogens significantly declined in weeks 1 to 3 of 2023 and reached their lowest level. Negative correlations were evident between the frequency of COVID-19 and that of other pathogens
The differences in co-infection rates before and after the lifting of pandemic containment and mitigation measures were not significant (P > 0.05). However, the patterns of multi-pathogen infections were more complex in 2023, as evidenced by the higher rate of triple-pathogen infections and the detection of a case of quadruple-pathogen infection (HRV-Boca-HPIV-HADV). The most common co-infection combination patterns were HRV-Mp, HRV-HPIV, and HRV-HADV in 2022, and HRV-Mp, HRV-HPIV and HRV-HRSV in 2023. HRV had the highest co-infection rate among all pathogens. During the study screening, only six cases of co-infection with SARS-CoV-2 and other viruses were detected (Fig. 5 and Table S5).
Co-infection pairwise calculation of composition ratio in different years (upper is 2022, lower is 2023). The numerator was the number of patients co-infected with ‘X’ and ‘Y,’ and the denominator was the total number of infections in patients who were tested for ‘X’ and ‘Y.’ Larger circles indicate higher co-infection rates between two pathogens
Discussion
Respiratory viruses are key pathogens that induce ARTIs and are the main cause of hospital admissions and deaths among children in low-income countries [21,22,23]. The rapid development of molecular diagnostic techniques has allowed for the widespread application of multiplex respiratory virus nucleic acid testing methods in clinical practice.
Epidemiological interference among respiratory viruses can affect virus frequency at the host and population levels and interfere with the subsequent frequency and duration of a certain virus or type of virus [24,25,26,27]. Mitigation measures were gradually eased in countries worldwide with the progression of the COVID-19 pandemic over time. Whether the revival of global travel, relaxation of protective measures, and reopening of public places and schools have led to a resurgence in respiratory viruses in children, or whether the lifting of pandemic containment and mitigation measures and the frequency of COVID-19 have interfered with the transmission of other respiratory pathogens are unclear.
The study was based at the First People’s Hospital of Yunnan Province, which has over 2,000 hospital beds and receives more than 2.5 million patients annually. Our results revealed that the overall frequency rate of common respiratory pathogens among children was not significantly affected by the pandemic containment and mitigation measures and remained high throughout the study period. However, the frequency rate determined in this study was considerably higher than the reported rates of 32.1% in Australia [28] and 36% in Xuzhou, China [29], indicating significant regional differences in respiratory pathogen frequency in children. Our results also indicated higher frequency rates in males than in females in 2023, which is inconsistent with findings reported in another study conducted in Guangzhou, China [30].
The most prevalent pathogens differed before and after the lifting of pandemic containment and mitigation measures. The frequency rates of Mp, HADV, and HMPV significantly decreased in 2023, whereas those of influenza-like viruses significantly increased in 2023 compared to 2022, with influenza virus A InfA-H1N1pdm09 and HPIV showing the most notable rebound. The pathogens with the highest frequency rates differed from those reported in other studies conducted in China, which may be attributed to differences in geographical region, study population, and sample size [3, 28]. HRV had the highest frequency rate during the entire study period, which is in agreement with previously reported findings [27]. HRSV frequency negatively correlated with InfA frequency, which is consistent with the results of a study conducted in Germany [31]. At 4 to 6 weeks after the lifting of pandemic containment and mitigation measures, most pathogens exhibited an interruption in frequency. This suggests that the resurgence in SARS-CoV-2 frequency generated a certain degree of interference at the host level, thereby causing a temporary decrease in the susceptibility of the host to viral infections and inducing an immune refractory period toward other pathogens. The specific mechanisms of interference may be related to the sustained upregulation of interferon-stimulated genes, chemokines, and cytokines caused by the frequency of COVID-19 [32].
Children aged 1–3 years had the highest frequency rates, which is consistent with previous findings [3]. Most pathogens exhibited obvious umbrella-shaped distribution characteristics and non-significant differences in frequency before and after the lifting of pandemic containment and mitigation measures. This suggests that the immature immune system of young children may have been insufficient to generate immunity after infection, resulting in a higher tendency for recurrent infections and cross-infections of respiratory pathogens. Interestingly, infants had the lowest infection rates, and most pathogens were undetected in this age group. Further research is necessary to determine whether the unique characteristics of respiratory pathogen infections in infants are related to the immune refractory period caused by immaturity of the immune system. RSV was most common among children aged ≤ 2 years, which is consistent with a previous study [33]. In contrast, the Mp infection rate gradually increased with an increase in age. Our results also indicated the occurrence of a shift in the season of high frequency of HRV, HRSV, and influenza virus A InfA-H3N2. However, further tracking and monitoring will be required to determine whether such shifts in the season of high frequency are temporary or ongoing changes owing to the short duration of the study.
Before December 2022, China had adopted a different response strategy from other countries. SARS-CoV-2 was undetected from week 1 to week 49 in 2022; precise prevention and control prevented the spread of the new coronavirus. Since the lifting of pandemic containment and mitigation measures in December 2022, the frequency of COVID-19 sharply increased and continued to be an epidemic; however, in weeks 1 to 3 of 2023, common respiratory pathogens significantly declined and reached their lowest level. This finding demonstrates the existence of an obvious negative correlation between the frequency of COVID-19 and other pathogens, which is consistent with the finding of a previous study (Fig. 4) [24].
Co-infections with pathogens were mainly dominated by double-pathogen infections; however, the patterns of co-infection became more complex in 2023. It remains unclear whether such changes in co-infection patterns are related to re-entry into the period of high exposure to bacteria and viruses. However, such a phenomenon should receive adequate attention from clinical fields, communities, and epidemic prevention and control authorities. The co-infection rate of HRV was considerably higher than that of all other pathogens and the elucidation of the mechanisms of co-infections is currently a focal topic in research [28]. Screening for SARS-CoV-2 revealed the presence of only six cases of co-infection with SARS-CoV-2 and other pathogens, which again demonstrates the existence of viral interference between COVID-19 frequency and other pathogens.
Despite our promising results, this study also had certain limitations. (1) The study was confined to Kunming, China, which could limit the generalizability of the findings to other regions with different climatic and population characteristics. Clinical and epidemiologic features of some other diseases may be linked to the geographic regions associated with the subjects [34, 35]. (2) Only a limited range of respiratory pathogens were included in the study, which may not encompass all bacteria and viruses that cause respiratory tract symptoms in children and those that are detected using other diagnostic tools, e.g., culture, antigen testing, and sequencing [36,37,38,39,40,41]. (3) Samples were collected from a single hospital over a short period, so studies with a longer study period and wider sample range will be conducted in the future to fully capture long-term trends and epidemiological shifts.
The present study, for the first time, presents valuable insights into the changes in the types and frequency patterns of common respiratory pathogens in children with ARTIs in Kunming during the post-COVID-19 pandemic period under the influence of changes in pandemic prevention and control measures and interference caused by the COVID-19 pandemic. These findings enhance our understanding of the interactions among respiratory pathogens, serve as valuable data for this unique historical period, and are of great significance for the effective control of RTIs and the reduction of the harm caused to pediatric health by respiratory pathogens.
Data availability
The datasets analyzed to support the findings of the current study are available from the corresponding author upon reasonable request.
References
Liu BM, Beck EM, Fisher MA (2021) The brief case: ventilator-associated Corynebacterium accolens pneumonia in a patient with respiratory failure due to COVID-19. J Clin Microbiol 59(9):e0013721. https://doi.org/10.1128/JCM.00137-21
Li ZJ, Zhang HY, Ren LL et al (2021) Etiological and epidemiological features of acute respiratory infections in China. Nat Commun 12:5026. https://doi.org/10.1038/s41467-021-25120-6
Jartti T, Jartti L, Ruuskanen O et al (2012) New respiratory viral infections. Curr Opin Pulm Med 18:271–278. https://doi.org/10.1097/MCP.0b013e328351f8d4
Liu BM, Li NL, Wang R et al (2024) Key roles for phosphorylation and the coiled-coil domain in TRIM56-mediated positive regulation of TLR3-TRIF-dependent innate immunity. J Biol Chem 300(5):107249. https://doi.org/10.1016/j.jbc.2024.107249
Pieren D, Boer MC, de Wit J (2022) The adaptive immune system in early life: the shift makes it count. Front Immunol 13:1031924. https://doi.org/10.3389/fimmu.2022.1031924
Brealey JC, Sly PD, Young PR et al (2015) Viral bacterial co-infection of the respiratory tract during early childhood. FEMS Microbiol Lett 362(10):fnv062. https://doi.org/10.1093/femsle/fnv062
Liu BM, Rakhmanina NY, Yang Z et al (2024) Mpox (monkeypox) virus and its co-infection with hiv, sexually transmitted infections, or bacterial superinfections: double whammy or a new prime. Culprit? Viruses 16(5):784. https://doi.org/10.3390/v16050784
Brodin P (2023) Immune responses to SARS-CoV-2 infection and vaccination in children. Semin IImmunol 69:101794. https://doi.org/10.1016/j.smim.2023.101794
Doenhardt M, Gano C, Sorg A et al (2022) Burden of pediatric SARS-CoV-2 hospitalizations during the Omicron wave in Germany. Viruses 14(10):2102. https://doi.org/10.3390/v14102102
Zhu L, Luo T, Yuan Y et al (2023) Epidemiological characteristics of respiratory viruses in hospitalized children during the COVID-19 pandemic in southwestern China. Front Cell Infect Microbiol 13:1142199. https://doi.org/10.3389/fcimb.2023.1142199
O’Driscoll M, Ribeiro DSG, Wang L et al (2021) Age-specific mortality and immunity patterns of SARS-CoV-2. Nature 590(7844):140–145. https://doi.org/10.1038/s41586-020-2918-0
Mandelia Y, Procop GW, Richter SS et al (2021) Dynamics and predisposition of respiratory viral co-infections in children and adults. Clin Microbiol Infect 27. https://doi.org/10.1016/j.cmi.2020.05.042. :631.e1–636.e6
Chow EJ, Uyeki TM, Chu HY (2023) The effects of the COVID-19 pandemic on community respiratory virus activity. Nat Rev Microbiol 21:195–210. https://doi.org/10.1038/s41579-022-00807-9
Wang ST, Wu YP, Li L et al (2023) Forecast for peak infections in the second wave of the Omicron after the adjustment of zero-COVID policy in the mainland of China. Infect Dis Model 8(2):562–573. https://doi.org/10.1016/j.idm.2023.05.007
Chen YC, Wang X, Teng YS et al (2023) Otolaryngology-related symptoms of COVID-19 in children in the post-epidemic era: a cross-sectional web-based survey study. Front Pediatr 11:1190734. https://doi.org/10.3389/fped.2023.1190734
Su Z, Cheshmehzangi A, McDonnell D et al (2022) The advantages of the zero-COVID-19 strategy. Int J Environ Res Public Health 19(14):8767. https://doi.org/10.3390/ijerph19148767
Lao X, Luo L, Lei Z et al (2021) The epidemiological characteristics and effectiveness of countermeasures to contain coronavirus disease 2019 in Ningbo City, Zhejiang Province, China. Sci Rep 11(1):9545. https://doi.org/10.1038/s41598-021-88473-4
Jackson GG, Dowling HF, Muldoon RL (1962) Acute respiratory diseases of viral etiology. VII. Present concepts of the common cold. Am J Public Health Nations Health 52:940–945. https://doi.org/10.2105/ajph.52.6.940
Karunarathne L, Amarasiri W, Fernando A (2023) Respiratory function in healthy long-term meditators: a cross-sectional comparative study. Heliyon 9(8):e18585. https://doi.org/10.1016/j.heliyon.2023.e18585
Peng Y, Liu B, Hou J et al (2015) Naturally occurring deletions/insertions in HBV core promoter tend to decrease in hepatitis B e antigen-positive chronic hepatitis B patients during antiviral therapy. Antivir Ther 20(6):623–632. https://doi.org/10.3851/IMP2955
GBD 2017 Influenza Collaborators (2019) Mortality, morbidity, and hospitalisations due to influenza lower respiratory tract infections, 2017: an analysis for the global burden of Disease Study 2017. Lancet Respir Med 7:69–89. https://doi.org/10.1016/S2213-2600(18)30496-X
Yen CY, Wu WT, Chang CY et al (2019) Viral etiologies of acute respiratory tract infections among hospitalized children - a comparison between single and multiple viral infections. J Microbiol Immunol Infect 52:902–910. https://doi.org/10.1016/j.jmii.2019.08.013
Di Mattia G, Nenna R, Mancino E et al (2021) During the COVID-19 pandemic where has respiratory syncytial virus gone? Pediatr Pulmonol 56:3106–3109. https://doi.org/10.1002/ppul.25582
Zheng X, Song Z, Li Y et al (2017) Possible interference between seasonal epidemics of influenza and other respiratory viruses in Hong Kong, 2014–2017. BMC Infect Dis 17:772. https://doi.org/10.1186/s12879-017-2888-5
Nickbakhsh S, Mair C, Matthews L et al (2019) Virus-virus interactions impact the population dynamics of influenza and the common cold. Proc Natl Acad Sci U S A 116:27142–27150. https://doi.org/10.1073/pnas.1911083116
Wu A, Mihaylova VT, Landry ML et al (2020) Interference between rhinovirus and influenza a virus: a clinical data analysis and experimental infection study. Lancet Microbe 1:e254–e262. https://doi.org/10.1016/s2666-5247(20)30114-2
Takashita E, Kawakami C, Momoki T et al (2021) Increased risk of rhinovirus infection in children during the coronavirus disease-19 pandemic. Influenza Other Respir Viruses 15:488–494. https://doi.org/10.1111/irv.12854
Price OH, Sullivan SG, Sutterby C et al (2019) Using routine testing data to understand circulation patterns of influenza A, respiratory syncytial virus and other respiratory viruses in Victoria, Australia. Epidemiol Infect 147:e221. https://doi.org/10.1017/S0950268819001055
Cao R, Du Y, Tong J et al (2023) Influence of COVID-19 pandemic on the virus spectrum in children with respiratory infection in Xuzhou, China: a long-term active surveillance study from 2015 to 2021. BMC Infect Dis 23:467. https://doi.org/10.1186/s12879-023-08247-3
Liao X, Hu Z, Liu W et al (2015) New epidemiological and clinical signatures of 18 pathogens from respiratory tract infections based on a 5-year study. PLoS ONE 10:e138684. https://doi.org/10.1371/journal.pone.0138684
Buchholz U, Lehfeld AS, Tolksdorf K et al (2022) Respiratory infections in children and adolescents in Germany during the COVID-19 pandemic. J Health Monit, 2023,8(2):20–38. https://doi.org/10.25646/11437
Ong HH, Andiappan AK, Duan K et al (2022) Transcriptomics of rhinovirus persistence reveals sustained expression of RIG-I and interferon-stimulated genes in nasal epithelial cells in vitro. Allergy 77:2778–2793. https://doi.org/10.1111/all.15280
Yu J, Xie Z, Zhang T et al (2018) Comparison of the prevalence of respiratory viruses in patients with acute respiratory infections at different hospital settings in North China, 2012–2015. BMC Infect Dis 18:72. https://doi.org/10.1186/s12879-018-2982-3
Liu B, Yang JX, Yan L, Zhuang H, Li T (2018) Novel HBV recombinants between genotypes B and C in 3’-terminal reverse transcriptase (RT) sequences are associated with enhanced viral DNA load, higher RT point mutation rates and place of birth among Chinese patients. Infect Genet Evol 57:26–35. https://doi.org/10.1016/j.meegid.2017.10.023
Serrano-Lomelin J, Nielsen CC, Hicks A et al (2020) Geographic inequalities of respiratory health services utilization during childhood in Edmonton and Calgary, Canada: a tale of two cities. Int J Environ Res Public Health 17(23):8973. https://doi.org/10.3390/ijerph17238973
Liu BM, Carlisle CP, Fisher MA, Shakir SM (2021) The brief case: Capnocytophaga sputigena bacteremia in a 94-year-old male with type 2 diabetes mellitus, pancytopenia, and bronchopneumonia. J Clin Microbiol 59(7):e0247220. https://doi.org/10.1128/JCM.02472-20
Wu X, Chen Q, Li J et al (2022) Diagnostic techniques for COVID-19: a mini-review. J Virol Methods 301:114437. https://doi.org/10.1016/j.jviromet.2021.114437
Liu B (2017) Universal PCR primers are critical for direct sequencing-based enterovirus genotyping. J Clin Microbiol 55(1):339–340. https://doi.org/10.1128/JCM.01801-16
Riccobono E, Bussini L, Giannella M et al (2022) Rapid diagnostic tests in the management of pneumonia. Expert Rev Mol Diagn 22(1):49–60. https://doi.org/10.1080/14737159.2022.2018302
Liu B, Forman M, Valsamakis A (2019) Optimization and evaluation of a novel real-time RT-PCR test for detection of parechovirus in cerebrospinal fluid. J Virol Methods 272:113690. https://doi.org/10.1016/j.jviromet.2019.113690
Hou N, Wang K, Zhang H et al (2020) Comparison of detection rate of 16 sampling methods for respiratory viruses: a bayesian network meta-analysis of clinical data and systematic review. BMJ Glob Health 5(11):e003053. https://doi.org/10.1136/bmjgh-2020-003053
Acknowledgements
The authors thank all the staff of the First People’s Hospital of Yunnan Province who participated in the sample collection and all the participants for their contribution to the study.
Funding
This work was supported by National Natural Science Foundation of China (grant number 82260395); Yunnan Provincial Department of Science and Technology-Kunming Medical University Applied Fundamental Research Joint Special Fund (grant number 202201AY070001-245) and Yunnan Provincial Key Laboratory of Clinical Virology (grant number 2023A4010403-05).
Author information
Authors and Affiliations
Contributions
Guiqian Zhang (Conceptualization, Formal analysis, Investigation, Writing–original draft, Writing–review & editing), Kaimei Wang (Data curation, Investigation, Formal analysis, Writing–original draft), Limei Ba (Investigation), Shumei Dong (Investigation), Jianmei Gao (Supervision, Methodology, Project administration, Writing – review & editing).
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the institutional review board of the First People’s Hospital of Yunnan Province (Approval Number: KHLL2023-KY055).
Consent to participate
Informed consent was obtained from the parents or guardians of all participants. The parents or guardians were informed of the laboratory results of pathogen detection.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, G., Wang, K., Ba, L. et al. Dynamic changes in the circulation of respiratory pathogens in children during and after the containment of the 2019 coronavirus disease pandemic in Kunming, China. Eur J Clin Microbiol Infect Dis (2024). https://doi.org/10.1007/s10096-024-04945-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10096-024-04945-1