Abstract
Purpose
The aim of this study was to to compare the antimicrobial resistance rate and its relationship with the antibiotic consumption in two separate Intensive Care Units (ICUs) of the same hospital, one with and other without selective decontamination of the digestive tract (SDD).
Methods
We performed a retrospective study in the two ICUs of the Araba University Hospital. Trauma and neurosurgical patients are admitted to the SDD-ICU, and general digestive surgery patients go to the no SDD-ICU. From 2014 to 2018 we analyzed the number of isolates, and the bacterial resistance trends of 47 antimicrobial-microorganism combinations. Additionally, antimicrobial consumption was estimated in both ICUs. Resistance rates were also compared with those reported in ENVIN-HELICS Spanish national registry.
Results
In the ICU with SDD protocol, there was a significant decrease in the resistance of E. coli to amoxicillin/clavulanic acid and in the resistance of E. faecalis to high concentration of gentamycin and high concentration of streptomycin. A significant increase of resistance of Staphylococcus coagulasa negative (CoNS) to linezolid in the no SDD-ICU was also detected. Overall, the level of resistance in the SDD-ICU was lower or of the same order than in the ICU without SDD and that reported in the Spanish national registry.
Conclusions
SDD had neither a clinically relevant impact on emergence and spread of resistance, nor in the overall systemic antimicrobial use. The patient type rather than the SDD protocol showed to condition the ecology and therefore, the resistance rate in the ICUs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Selective decontamination of the digestive tract (SDD), proposed almost 40 years ago, is a measure to prevent infection in intensive care unit (ICU) patients [1]. It involves the application of topical, nonabsorbable antimicrobial agents (usually colistin, tobramycin, and nystatin) that selectively spare the anaerobic flora. Apart from the topical prophylaxis applied to the patients, intravenous antibiotics, usually a second- or third-generation cephalosporin (without activity against the anaerobic Gram-negative microbiota) is added to treat any incubating infection caused by the commensal flora of the respiratory tract at the time of hospital admission [2]. The control of gut overgrowth is one mechanism proposed to explain how SDD regimens might prevent ICU-acquired infection; therefore the aim of SDD is to eradicate colonization with aerobic Gram-negative bacteria, Staphylococcus aureus and yeasts, while leaving the anaerobic flora intact (and thereby improve patient outcomes) [3]. Patients with an expected ICU stay of at least two or three days, and receiving mechanical ventilation are the targeted population, and immediately upon ICU admission the preferred moment to start the SDD protocol [4, 5]. This intervention has been extensively studied and has been proven to be effective in infection prevention and mortality reduction, particularly in settings with a low prevalence of multidrug-resistant bacteria [6,7,8].
In spite of the benefits of SDD, its implementation in the ICUs is still a matter of debate [9,10,11] mainly due to concerns that it may promote the development of antimicrobial-resistant pathogens [12,13,14]. In settings with relatively low prevalence of multidrug-resistant pathogens, such as ICUs in the Netherlands, Australia, and New Zealand, evidence that the implementation of SDD has a negative impact in resistance ecology is scarce [2, 15]. In a recent systematic review and meta-analysis in which 32 randomized clinical trials including 24,389 participants were analyzed, the use of SDD in ICU adults treated with mechanical ventilation was associated with lower hospital mortality in comparison with standard care or placebo, but evidence regarding the effect of SDD on antimicrobial resistance was of very low certainty [16]. For settings with high prevalence of multidrug-resistant microorganisms, large-scale studies are still needed to evaluate the effect of SDD on resistance rates.
In a previous study carried out by our group [17], we did not detect relevant differences in the overall susceptibility rate before and after the implementation of an SDD protocol in an ICU of a tertiary hospital of the north of Spain, a geographical area with moderate to high rate of antibiotic resistance. In the present investigation, our aim was to compare the antimicrobial resistance rate and its relationship with the antibiotic consumption in two separate ICUs of the same hospital, one with SDD and other without SDD.
Materials and methods
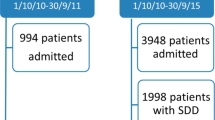
The study was carried out in the two ICUs of the Araba University Hospital (Vitoria-Gasteiz, Spain), an 800 beds tertiary care teaching facility. Both ICUs are located in two different buildings, and had different staff. Mainly trauma and neurosurgical patients are admitted to the ICU with SDD, a 13 bed ICU where SDD was implemented in 2002. Mainly general digestive surgery patients go to the ICU without SDD (no SDD-ICU), with 18 beds. In the SDD-ICU, SDD was administered to patients expected to require mechanical ventilation for more than 48 h. A 2% mixture gentamycin (replaced by tobramycin in 2017), colistin and amphotericin B was applied on the buccal mucosa, and a suspension of the same drugs (respective doses of 80, 100 and 500 mg) was provided in the gastrointestinal tract at 6-h intervals. For the first 3 days, systemic ceftriaxone (2 g IV a day) was administered to all SDD patients. The SDD of the digestive tract started on the day of tracheal intubation and continued until the patients were weaned from mechanical ventilation.
The mean age of the patients (63 vs. 66 years-old in the SDD-ICU and no SDD-ICU, respectively), the stay length (5 vs. 4 days in the SDD-ICU and no SDD-ICU, respectively) and the number of admissions/year (880 vs. 1058 in the SDD-ICU and no SDD-ICU, respectively) were similar in the two ICUs.
Except the SDD, the protocols for infection control were the same in the two ICUs; among others International Standard ISO 9001:2000 guidance, the ‘Bacteremia Zero’ program, the ‘Zero-VAP’ (where VAP stands for ventilator-associated pneumonia) bundle, and Zero Resistance program.
This study met the exemption criteria of the ethics committee of clinical research because the data analyzed were collected in the routine practice and did not allow the identification of patients.
Bacterial isolates and antimicrobial resistance
We retrospectively collected the number of isolates, resistance rates and bacterial resistance trends in 47 antimicrobial-microorganism combinations in both ICUs from 2014 to 2018 (Table S1 supplementary material). Quarterly data of isolates and resistance rates were used (in the SDD-ICU, data collection started from the first quarter of 2014, and in the no SDD-ICU, data collection started from the last quarter of 2014). For enterococci, we also tested the high-level aminoglycoside resistance (isolates with gentamycin MIC ≥ 500 mg/L or streptomycin MIC ≥ 1000 mg/L) since it suppresses the synergic effect with cell wall synthesis inhibitors, which compromises the treatment in case of enterococcal endocarditis and meningitis. For resistance rate calculations, we considered only the first isolate from the patients admitted to the ICU, according to the CLSI guidelines [18, 19] and the National Guideline [20]. The resistance data, derived from microbiological surveillance and clinical care were re-interpreted using interpretive criteria current at the time of analysis and based on the Clinical and Laboratory Standards Institute (CLSI) [19]. Data. Surveillance samples from the nasal, throat and rectum (swabs), were obtained twice weekly. Blood, urine and another diagnostic samples were taken on clinical indication only. The laboratory data from the Microbiology department were managed with Whonet [21], a Windows-based database software package for the management of microbiology laboratory data and the analysis of antimicrobial susceptibility test results. It is a free software developed by the WHO Collaborating Centre for Surveillance of Antimicrobial Resistance for laboratory-based surveillance of infectious diseases and antimicrobial resistance.
We also obtained data of antimicrobial resistance from the ENVIN-HELICS national registry [22] (Spanish Surveillance Study of Nosocomial Infection in the ICU, which includes data from all patients admitted to the participating ICUs for more than 24 h). According to the ENVIN-HELICS protocol, for each pathogen, only the first isolate is included, even if there are several infections or colonizations. The source that assumes the greatest severity (for example, blood over abdominal drainage) is chosen. For this work, data from the 2018 report were used (corresponding to resistance data from April 1 to June 30, 2018), which includes a total of 27.514 patients and 219 ICUs. Less than 5% of these ICUs have implanted the SDD; therefore, these data were used as reference resistance values in absence SDD.
Antibiotic use
From 2014 to 2018, quarterly quantities of the antimicrobial drugs consumed in each ICU were retrospectively collected from the pharmacy stock management system (in both ICUs, data collection started in the first quarter of 2014). Similarly, numbers of occupied bed-days (OBD) per quarter were obtained from the hospital´s admission department. Antibacterial consumption, expressed as DDDs/100 patient-days, was calculated quarterly in each ICU, according to the 2020 version of the ATC/DDD classification [23]. Prophylactic and therapeutic medication were not distinguished. For every antibiotic, the amount prescribed was also expressed as the percentage of the total antibiotics prescribed.
Statistical analysis
Trends in the rate of resistance and in the antimicrobial consumption were analyzed with linear correlation. A p value of < 0.05 was considered statistically significant. Statistical analyses were performed with the IBM® SPSS® software, (Statistics for Windows, Version 27). When resistance was always 0, the resistance trends was not evaluated.
Results
Figure 1 features the evolution of the number of Gram-positive and Gram-negative isolates in the two ICUs from 2014 to 2018. The more frequent Gram-negative microorganisms were P. aeruginosa and E. coli, and no relevant differences were found between the two UCIs. Overall, the number of Gram-positive isolates were higher in the SDD-ICU than in the no SDD-ICU.
In both ICUs, the most prescribed antibiotics (apart from the antibiotics included in the SDD protocol in the SDD-ICU) were piperacillin/tazobactam, amoxicillin/clavulanic acid, cloxacillin, ceftriaxone, meropenem, levofloxacin, and linezolid (Supplementary Fig. 1). Regarding the consumption rate of each antibiotic with respect to the total antibiotics prescribed in each ICU (Supplementary Table 2), the greater differences between the two ICUs were for cloxacillin (9.51% in the SDD-ICU vs. 3.62% in the no SDD-ICU), cefepime (5.28% in the SDD-ICU vs. 2.50% in the no SDD-ICU), piperacillin/tazobactam (8.81% in the SDD-ICU vs. 19.20% in the no SDD-ICU), and levofloxacin (6.68% in the SDD-ICU vs. 11.08% in the no SDD-ICU).
Table 1 shows the antimicrobial consumption trends in the two ICUs from 2014 to 2018 (only significant trends are included). In the SDD-ICU, the consumption of amikacin, gentamycin and piperacillin/tazobactam significantly decreased, whereas the consumption of ceftazidime, tobramycin, and all aminoglycosides significantly increased. In the no SDD-ICU, the use of aztreonam, daptomycin, linezolid, meropenem, all beta-lactams, and all antimicrobials significantly increased.
Table 2 shows significant antibiotic resistance trends from 2014 to 2018 in the two ICUs. In the SDD-ICU, there was a significant decrease in the resistance of E. coli to amoxicillin/clavulanic acid and in the resistance of E. faecalis to high concentration of gentamycin and high concentration of streptomycin. In the no SDD-ICU, we detected a significant increase of the resistance of K. pneumoniae to ceftazidime and cefotaxime, and the resistance of CoNS to linezolid.
Table 3 presents a comparison of the resistance rate of the most frequent microorganisms in the SDD-ICU and no SDD-ICU in 2018 (annual data) and those published in the ENVIN-HELICS report for 2018. Except for ceftazidime and piperacillin/tazobactam, resistance rate of E. coli in the no SDD-ICU were comparable to that from the ENVIN-HELICS study, and higher than those in the SDD-ICU. Regarding P. aeruginosa, the higher difference was detected for amikacin, ceftazidime, cefepime and piperacillin/tazobactam (higher in ENVIN-HELICS), and for colistin and imipenem, (higher in our two ICUs). Resistance rate of S. aureus to oxacillin was lower in the SDD-ICU than in the no SDD-ICU, and comparable to the ENVIN-HELICS report. In both ICUs and in the national survey, resistance of CoNS to linezolid was of the same order.
Discussion
The effect of SDD on ICU-level antimicrobial resistance rates is largely underexplored and existing studies of selective decontamination have not answered the question of how selective decontamination affects ICU-level antimicrobial resistance rates over time [2]. One of the difficulties to evaluate the effect of SDD on the emergence of resistance rate is the controversy due to variable study design including control groups that may not receive SDD protocol. In this observational and retrospective study, we have compared the resistance rate in two ICUs belonging to the same institution, one with SDD protocol and the other without it. According to admission criteria, trauma and neurosurgical patients with ventriculoperitoneal shunts are admitted to SDD-ICU. It is important to note that trauma patients at the ICU often require prolonged mechanical ventilation, and therefore, they may benefit from SDD. On the contrary, general digestive surgery patients are admitted to the no SDD-ICU, where no SDD has been implemented.
The application or not of SDD protocols may conditions the circulating microorganisms at the ICU [1]. In fact, the antimicrobial spectrum of the classic SDD regimen lacks coverage of most Gram-positive bacteria, and increased rates of colonization and infection with enterococci and methicillin-resistant S. aureus (MRSA) can be expected as it has been reported [24, 25]. In line with this, we detected a higher number of E. faecalis and S. aureus isolates in the ICU with the SDD protocol, and a decrease in the number of E. faecium isolates. These results are in agreement with those found by van der Bij et al. [26] in Dutch ICUs with and without SDD, who reported that the introduction of SOD (selective oropharyngeal decontamination)/SDD was associated with increased rates of S. aureus and E. faecalis isolates in respiratory tract specimens; moreover, they observed an increase in E. faecium isolates in the absence of SOD/SDD.
The differences in the prescription of some antimicrobials between the two ICUs may be justified in part by the patient and infection type, and in part by the application of the SDD protocol. Directly related to the SDD protocol, the consumption of gentamycin, tobramycin and colistin was much higher in the SDD-ICU than in the no SDD-ICU, where these antibiotics are hardly prescribed. The change in the SDD protocol in 2017 (gentamycin was replaced by tobramycin) explains the significant increase of tobramycin consumption and the significant decrease of gentamycin consumption in the SDD-ICU. Our SDD protocol included ceftriaxone since resistance rate of Enterobacterales is below 10%. In other countries with moderate-to-high-prevalence of antibiotic resistant bacteria, including third-generation cephalosporin-resistant Enterobacterales, intravenous cephalosporin is absent in the SDD protocols [5] since such prophylaxis was considered unappropriated.
Regarding the kind of patient and infection, and according to the admission criteria, gastrointestinal surgery patients go to no SDD-ICU, and since these patients are at high risk of abdominal infection, the consumption of antibiotics indicated for this kind of infections are higher in this ICU, such us piperacillin/tazobactam and levofloxacin. On the contrary, infections by Gram-positive microorganisms are more frequent in SDD-ICU, who admit the trauma and neurosurgical patients, and therefore, antibiotics such as cloxacillin and cephalosporins, were more prescribed in this ICU.
The high meropenem prescription in our ICUs may be related to the high prevalence of multiresistent P. aeruginosa in our region (north of Spain), as demonstrated in a large-scale Spanish nationwide survey of P. aeruginosa infections [27]. This pathogen has remarkable capacity for acquiring new resistance mechanisms under selective antibiotic pressure [28, 29]; in fact, carbapenem-resistant P. aeruginosa has become common worldwide, and it is known that resistance is associated with carbapenems use.
The increase along the period of study of linezolid and daptomycin consumption, both antibiotics indicated for the treatment of Gram-positive microorganism infections, more frequent in this ICU, may be due to the introduction of the generic drug products in the therapeutic guide of the hospital, linezolid in 2015 and daptomycin in 2018. The increase of prescription of these antibiotics after generic approval is also reported in other studies [30]. In a recent study [6], overall total daily defined doses for each antibiotic class were not significantly different between SDD and standard of care. In our study, we detected a significant increase of all beta-lactams and all antibiotics use over time in the ICU without SDD, while in the ICU with SDD, the consumption of both, all beta-lactams and all antibiotics remained constant. These results are in agreement with Daneman [31], which postulates that the use of prophylactic selective decontamination could even lead to reductions in the need for therapeutic antimicrobials.
We found reduction in the resistance rate of E. coli to amoxicillin/clavulanic acid in the SDD-ICU, which was not detected in the no SDD-ICU. In a previous study [32], it was shown that previous exposition to piperacillin/tazobactam is a predisposing factor for amoxicillin/clavulanic acid-resistant E. coli isolates. In this sense, the reduction of the resistance of E. coli to amoxicillin/clavulanic acid in the SDD-ICU could be related to the significant reduction of the piperacillin/tazobactam consumption in this ICU. Additionally, significant decrease of E. faecalis resistant to high concentration of gentamycin and to high concentration of streptomycin was also found, which is probably related to the change of gentamycin by tobramycin the SDD protocol. K. pneumoniae is a major pathogen implicated in nosocomial infections that is known to spread easily, and it is frequently associated with resistance to the highest-priority critically important antimicrobial agents [33]. We found a significant increase of K. pneumoniae to cefotaxime and ceftazidime in the no SDD-ICU, not observed in the SDD-ICU. This result may be explained, in part, by the higher use of fluoroquinolones (mainly levofloxacin) in this ICU, since the consumption of these antibiotics is associated with the development of extended-spectrum-beta-lactamases (ESBL) [34], one of the most common resistance mechanisms of K. pneumoniae [35, 36]. This resistance pattern may be also explained by the decontamination protocol, since SDD has demonstrated to be efficacious in controlling colonization with ESBL-producing bacteria [37]. However, considering the low number of isolates, this result must be interpreted with caution.
Resistance of CoNS to linezolid increased in the ICU without SDD protocol, where a significant increase of this antibiotic use was also found. CoNS are the main pathogens in health care-associated ventriculitis and meningitis (HCAVM) [38]. Although linezolid is not registered for the treatment of CoNS infections, it is widely used off-label for the treatment of meningitis, ventriculitis, osteomyelitis and prosthetic-joint infections caused by CoNS promoting emergence of resistance [39, 40].
Our results in the ICU with SDD are in line with a previous case-control study of 5034 mechanically ventilated critically ill patients over 5-year period, which revealed that ESBL-producing K. penumoniae was more common in the ICU patients not treated with SOD [41]. However, contrary to this study, we did not detected increase of vancomycin-resistant E. faecium in any of the two ICUs.
In order to better evaluate if the SDD could have an impact on the antimicrobial resistance, we compared the resistance rates of the most frequent microorganisms in the two ICUs of our hospital with those reported in the ENVIN-HELICS national registry [22], which includes data from 219 Spanish ICUs (SDD represents less than 5%). We have confirmed the high resistance level of P. aeruginosa against meropenem in our ICUs, similar to the media of Spanish ICUs, which may be related to the high prevalence in our geographical area of the sequence type (ST) 175 [27], as mentioned above. Resistance of P. aeruginosa against piperacillin/tazobactam in our ICUs was much lower than that reported in the national survey. In the SDD-ICU, MRSA rate was similar to that reported in the national registry, being lower than in the ICU without SDD; this result may be explained, at least in part, by the contribution of SDD to control the resistance of MRSA. The worrying resistance of CoNS to linezolid observed in our ICUs and in the national registry may be related to the ability of these bacteria to develop resistance quite easily following linezolid exposure [42]. Actually, the rate of linezolid-resistant CoNS is increasing worldwide [43].
Our study presents some limitations. First, since the admission to one or the other ICU is conditioned by the kind of patient, the ICU without SDD protocol cannot strictly be considered as a control. Therefore, in the ICU with SDD, pneumonias are predominantly expected, while abdominal infections may dominate in the no SDD ICU, and accordingly, different pathogens are to be expected and therefore different antibiotics are used. Second, this is a retrospective study including a single hospital, with low number of isolates. However, this study provides unique information on the trends in antibiotic resistance in two ICUs with and without SDD located in a geographical area with medium-high rates of resistance. Third, antibiotic consumption and resistance rate are from pre-Covid 19 period; it has been reported that the occurrence of bacterial pneumonia, mostly ventilator-associated pneumonia (VAP), is a frequent complication in critically ill COVID-19 patients with a higher incidence than in non-COVID-19 patients [44]; moreover, and similarly to other patients admitted to the ICU, in COVID-19 patients, the development of secondary infections prolongs the length of mechanical ventilation and hospitalization, increasing the mortality risk [45]. In a previous study [46], the use of SDD for VAP prevention did not led to changes in the incidence of multidrug-resistant bacteria in COVID-19 patients; therefore, the conclusions obtained in the 2014–2018 period could be applied to the post-Covid 19 age.
In conclusion, by analyzing the resistance rates of the isolates in the two ICUs of our hospital, we have confirmed that SDD had not a clinically relevant impact on emergence and spread of resistance. Additionally, SDD has not been found to increase overall systemic antibiotic use. The patient type rather than the SDD protocol showed to condition the ecology and therefore, the resistance rate at the ICUs.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Wittekamp BHJ, Oostdijk EAN, Cuthbertson BH et al (2020) Selective decontamination of the digestive tract (SDD) in critically ill patients: a narrative review. Intensive Care Med 46:343–349. https://doi.org/10.1007/s00134-019-05883-9
Bonten M (2022) Selective decontamination of the Digestive Tract: an answer at last? JAMA 328:2310–2311. https://doi.org/10.1001/jama.2022.18623
van Hout D, Plantinga NL, Bruijning-Verhagen PC et al (2019) Cost-effectiveness of selective digestive decontamination (SDD) versus selective oropharyngeal decontamination (SOD) in intensive care units with low levels of antimicrobial resistance: an individual patient data meta-analysis. BMJ Open 9:e028876. https://doi.org/10.1136/bmjopen-2018-028876
de Smet AM, Kluytmans JA, Cooper BS et al (2009) Decontamination of the digestive tract and oropharynx in ICU patients. N Engl J Med 360:20–31. https://doi.org/10.1056/NEJMoa0800394
Wittekamp BH, Plantinga NL, Cooper BS et al (2018) Decontamination strategies and bloodstream infections with antibiotic-resistant microorganisms in ventilated patients: a Randomized Clinical Trial. JAMA 320:2087–2098. https://doi.org/10.1001/jama.2018.13765
SuDDICU Investigators for the Australian and New Zealand Intensive Care Society Clinical Trials Group, Myburgh JA, Seppelt IM, Goodman F et al (2022) Effect of selective decontamination of the Digestive Tract on Hospital Mortality in critically ill patients receiving mechanical ventilation: a Randomized Clinical Trial. JAMA 328:1911–1921. https://doi.org/10.1001/jama.2022.17927
Oostdijk EAN, Kesecioglu J, Schultz MJ et al (2014) Effects of decontamination of the oropharynx and intestinal tract on antibiotic resistance in ICUs: a randomized clinical trial. JAMA 312:1429–1437. https://doi.org/10.1001/jama.2014.7247
Minozzi S, Pifferi S, Brazzi L, et a (2021) l Topical antibiotic prophylaxis to reduce respiratory tract infections and mortality in adults receiving mechanical ventilation. Cochrane Database Syst Rev 1:CD000022. https://doi.org/10.1002/14651858.CD000022.pub4
De Waele JJ, Leroux-Roels I, Depuydt P (2023) Selective digestive decontamination - pro. Intensive Care Med 49:979–981. https://doi.org/10.1007/s00134-023-07100-0
Hurley JC (2023) Establishing the safety of selective digestive decontamination within the ICU population: a bridge too far? Trials 2023;24:337. https://doi.org/10.1186/s13063-023-07356-3
Wiersinga WJ (2023) Selective digestive decontamination- not sure. Intensive Care Med 49:984–986. https://doi.org/10.1007/s00134-023-07115-7
Halaby T, Al Naiemi N, Kluytmans J et al (2013) Emergence of colistin resistance in Enterobacteriaceae after the introduction of selective digestive tract decontamination in an intensive care unit. Antimicrob Agents Chemother 57:3224–3229. https://doi.org/10.1128/AAC.02634-12
Brink AJ, Coetzee J, Corcoran C et al (2013) Emergence of OXA-48 and OXA-181 carbapenemases among Enterobacteriaceae in South Africa and evidence of in vivo selection of colistin resistance as a consequence of selective decontamination of the gastrointestinal tract. J Clin Microbiol 51:369–372. https://doi.org/10.1128/JCM.02234-12
Buitinck SH, Jansen R, Bosman RJ et al (2022) Eradication of resistant and susceptible aerobic gram-negative Bacteria from the Digestive Tract in critically ill patients; an Observational Cohort Study. Front Microbiol 12:779805. https://doi.org/10.3389/fmicb.2021.779805
Oostdijk EA, de Smet AM, Blok HE et al (2010) Ecological effects of selective decontamination on resistant gram-negative bacterial colonization. Am J Respir Crit Care Med 181:452–457. https://doi.org/10.1164/rccm.200908-1210OC
Hammond NE, Myburgh J, Seppelt I et al (2022) Association between Selective Decontamination of the Digestive Tract and In-Hospital mortality in Intensive Care Unit patients receiving mechanical ventilation: a systematic review and Meta-analysis. JAMA 328:1922–1934. https://doi.org/10.1001/jama.2022.19709
Lloréns-Villar Y, Tusell F, Canut A et al (2019) Antibiotic susceptibility trend before and after long-term use of selective digestive decontamination: a 16 year ecological study. J Antimicrob Chemother 74:2289–2294. https://doi.org/10.1093/jac/dkz186
Clinical Laboratory Standards Institute. M39-A4 Analysis and Presentation of Cumulative Antimicrobial Susceptibility Test Data. Clinical and Laboratory Standards Institute; Wayne, PA, USA (2022) : 5th Edition
Clinical and Laboratory Standards Institute 2018 Performance Standards for Antimicrobial Susceptibility Testing. 28th ed. CLSI Supplement M100. Wayne, PA, USA
Larrosa MN, Canut-Blasco A, Benito N et al Spanish Antibiogram Committee (COESANT) recommendations for cumulative antibiogram reports 2023. Enferm Infecc Microbiol Clin (Engl Ed) 41:430–435. https://doi.org/10.1016/j.eimce.2022.09.002
WHONET 5.6. Available from http://www.whonet.org/ (Last accessed 20 July 2023)
ENVIN-HELICS. Sociedad Española de Medicina Intensiva, Crítica y Unidades Coronarias (SEMICYUC) Report of 2018. Grupo de Trabajo de Enfermedades Infecciosas. Estudio Nacional de Vigilancia de Infección Nosocomial en UCI.Available from https://hws.vhebron.net/envin-helics/# (Last Accessed 20 July 2023)
WHO Collaborating Centre for Drug Statistics Methodology, https://www.whocc.no/ddd/definition_and_general_considera/ (Last Accessed 20 July 2023)
Bonten MJ (2006) Selective digestive tract decontamination–will it prevent infection with multidrug-resistant gram-negative pathogens but still be applicable in institutions where methicillin-resistant Staphylococcus aureus and Vancomycin-resistant enterococci are endemic? Clin Infect Dis 43(Suppl 2):S70–S74. https://doi.org/10.1086/504482
Bello Gonzalez TDJ, Pham P, Top J et al (2017) Characterization of Enterococcus isolates colonizing the Intestinal Tract of Intensive Care Unit patients receiving selective Digestive Decontamination. Front Microbiol 8:1596. https://doi.org/10.3389/fmicb.2017.01596
van der Bij AK, Frentz D, Bonten MJ, ISIS-AR Study Group (2016) Gram-positive cocci in Dutch ICUs with and without selective decontamination of the oropharyngeal and digestive tract: a retrospective database analysis. J Antimicrob Chemother 71:816–820. https://doi.org/10.1093/jac/dkv396
Del Barrio-Tofiño E, Zamorano L, Cortes-Lara S, GEMARA-SEIMC/REIPI Pseudomonas study Group et al (2019) Spanish nationwide survey on Pseudomonas aeruginosa antimicrobial resistance mechanisms and epidemiology. J Antimicrob Chemother 74:1825–1835. https://doi.org/10.1093/jac/dkz147
Strateva T, Yordanov D (2009) Pseudomonas aeruginosa - a phenomenon of bacterial resistance. J Med Microbiol 58:1133–1148. https://doi.org/10.1099/jmm.0.009142-0
Mesaros N, Nordmann P, Plésiat P et al (2007) Pseudomonas aeruginosa: resistance and therapeutic options at the turn of the new millennium. Clin Microbiol Infect 13:560–578. https://doi.org/10.1111/j.1469-0691.2007.01681.x
Pham TT, Mabrut E, Cochard P et al (2021) Cost of off-label antibiotic therapy for bone and joint infections: a 6-year prospective monocentric observational cohort study in a referral centre for management of complex osteo-articular infections. J Bone Jt Infect 6:337–346. https://doi.org/10.5194/jbji-6-337-2021
Daneman N, Sarwar S, Fowler RA et al (2013) SuDDICU Canadian Study Group. Effect of selective decontamination on antimicrobial resistance in intensive care units: a systematic review and meta-analysis. Lancet Infect Dis 13:328–341. https://doi.org/10.1016/S1473-3099(12)70322-5
Rodríguez-Baño J, Oteo J, Ortega A et al (2013) Epidemiological and clinical complexity of Amoxicillin-clavulanate-resistant Escherichia coli. J Clin Microbiol 51:2414–2417. https://doi.org/10.1128/JCM.00999-13
Marques C, Belas A, Aboim C et al (2019) Evidence of sharing of Klebsiella pneumoniae strains between Healthy Companion Animals and cohabiting humans. J Clin Microbiol 57:e01537–e01518. https://doi.org/10.1128/JCM.01537-18
European Centre for Disease Prevention and Control (2023) Antimicrobial resistance in the EU/EEA (EARS-Net) -Annual Epidemiological Report 2022. ECDC, Stockholm
Carvalho I, Chenouf NS, Carvalho JA et al (2021) Multidrug-resistant Klebsiella pneumoniae harboring extended spectrum β-lactamase encoding genes isolated from human septicemias. PLoS ONE 16:e0250525. https://doi.org/10.1371/journal.pone.0250525
Aires-de-Sousa M, Lopes E, Gonçalves ML et al (2020) Intestinal carriage of extended-spectrum beta-lactamase-producing Enterobacteriaceae at admission in a Portuguese hospital. Eur J Clin Microbiol Infect Dis 39:783–790. https://doi.org/10.1007/s10096-019-03798-3
Zandstra D, Abecasis F, Taylor N et al (2013) For control of colonisation with extended-spectrum β-lactamase-producing bacteria, SDD does work. Intensive Care Med 39:539. https://doi.org/10.1007/s00134-012-2780-0
Ye Y, Tian Y, Kong Y (2022) Trends of Antimicrobial susceptibility in clinically significant coagulase-negative Staphylococci isolated from cerebrospinal fluid cultures in neurosurgical adults: a nine-year analysis. Microbiol Spectr 10:e0146221. https://doi.org/10.1128/spectrum.01462-21
Boak LM, Li J, Spelman D et al (2006) Successful treatment and cerebrospinal fluid penetration of oral linezolid in a patient with coagulase-negative Staphylococcus ventriculitis. Ann Pharmacother 40:1451–1455. https://doi.org/10.1345/aph.1H029
Gostev V, Leyn S, Kruglov A et al (2021) Global expansion of linezolid-resistant coagulase-negative Staphylococci. Front Microbiol 12:661798. https://doi.org/10.3389/fmicb.2021.661798
Wang B, Briegel J, Krueger WA et al (2022) Ecological effects of selective oral decontamination on multidrug-resistance bacteria acquired in the intensive care unit: a case-control study over 5 years. Intensive Care Med 48:1165–1175. https://doi.org/10.1007/s00134-022-06826-7
Shariati A, Dadashi M, Chegini Z et al (2020) The global prevalence of Daptomycin, Tigecycline, Quinupristin/Dalfopristin, and linezolid-resistant Staphylococcus aureus and coagulase-negative staphylococci strains: a systematic review and meta-analysis. Antimicrob Resist Infect Control 9:56. https://doi.org/10.1186/s13756-020-00714-9
Rodríguez-Lucas C, Fernández J, Boga JA et al (2018) Nosocomial ventriculitis caused by a meticillin- and linezolid-resistant clone of Staphylococcus epidermidis in neurosurgical patients. J Hosp Infect 100:406–410. https://doi.org/10.1016/j.jhin.2018.02.011
Rouzé A, Martin-Loeches I, Povoa P et al (2021) Relationship between SARS-CoV-2 infection and the incidence of ventilator-associated lower respiratory tract infections: a European multicenter cohort study. Intensive Care Med 47:188–198. https://doi.org/10.1007/s00134-020-06323-9. Erratum in: Intensive Care Med 2022;48:514–515
Grasselli G, Scaravilli V, Mangioni D et al (2021) Hospital-Acquired infections in critically ill patients with COVID-19. Chest 160:454–465. https://doi.org/10.1016/j.chest.2021.04.002
Biagioni E, Ferrari E, Gatto I et al (2023) Role of selective Digestive Decontamination in the Prevention of Ventilator-Associated Pneumonia in COVID-19 patients: a Pre-post Observational Study. J Clin Med 12:1432. https://doi.org/10.3390/jcm12041432
Acknowledgements
This work was supported by the Department of Education of the Basque Government (IT1587-22).
Funding
This work was supported by the Department of Education of the Basque Government (IT1587-22).
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
Alicia Rodríguez-Gascón and Andrés Canut-Blasco participated in the conception and design of the study. Yanire Lloréns-Villar and Andrés Canut-Blasco participated in the acquisition of data. Yanire Lloréns-Villar, Alicia Rodríguez-Gascón and Andrés Canut-Blasco performed the analysis of the data. All authors participated in the interpretation of the data and in the preparation of the draft version. All authors approved the submitted version.
Corresponding author
Ethics declarations
Ethical approval
This study met the exemption criteria of the ethics committee of clinical research because the data analyzed were collected in the routine practice and did not allow the identification of patients.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rodríguez-Gascón, A., Lloréns-Villar, Y., Solinís, M.Á. et al. Does selective digestive decontamination (SDD) increase antibiotic resistance? Long-term comparison of two intensive care units (with and without SDD) of the same tertiary hospital. Eur J Clin Microbiol Infect Dis 43, 885–893 (2024). https://doi.org/10.1007/s10096-024-04792-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-024-04792-0