Abstract
Antibiotic-resistant Acinetobacter baumannii (A. baumannii) is a common cause of hospital-acquired infections. This study aimed to identify independent factors associated with progression from nosocomial pneumonia to bacteremia in patients infected with carbapenem-resistant A. baumannii (CR-AB). From 2019 to 2021, we conducted a retrospective anaylsis of the medical records of 159 nosocomial CR-AB pneumonia patients in our Intensive Care Unit (ICU). We employed both univariate and multivariable logistic regression models to identify factors associated with the progression of nosocomial CR-AB pneumonia to bacteremia. Among the 159 patients with nosocomial CR-AB pneumonia, 40 experienced progression to bacteremia and 38 died within 28 days following diagnosis. Patients who developed bacteremia had a significantly higher 28-day mortality rate compared to those without bloodstream infection (47.50% vs. 15.97%). Multivariable logistic regression revealed that higher levels of C-Reactive protein (CRP) (OR = 1.01) and the use of continuous veno-venous hemofiltration (CVVH) treatment (OR = 2.93) were independently associated with an elevated risk of developing bacteremia. Among patients who developed bloodstream infection, those who died within 28 days exhibited significantly higher level of interleukin-6 (IL-6), a greater frequency of antifungal drugs usage, and a longer duration of machanical ventilation compared to survivors. Furthermore, the use of antifungal drugs was the only factor that associated with 28-day mortality (OR = 4.70). In ICU patients with central venous catheters who have CR-AB pneumonia and are on mechanical ventilation, higher CRP levels and CVVH treatment are risk factors for developing bacteremia. Among patients with bacteremia, the use of antifungal drugs is associated with 28-day mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acinetobacter baumannii (A. baumannii) is a non-fermentative Gram-negative aerobic coccobacillus and an opportunistic pathogen [1]. The widespread use of antibiotics has led to the emergence of various antibiotic-resistant A. baumannii strains [2], These include carbapenem-resistant A. baumannii (CR-AB) [3], multi-drug resistant A. baumannii (MDR-AB) [4] and pan-resistant A. baumannii (PDR-AB) [5]. The annual global incidence of A. baumannii infections is estimated to be approximately 1,000,000 cases, with 50% exhibiting resistance to multiple antibiotics, including carbapenems [6]. Currently, antibiotic-resistant A.baumannii has become a common cause of hospital-acquired infections, particularly in intensive care units (ICUs) [7, 8]. The antibiotic resistance of A. baumannii significantly limits treatment options [9, 10], leading to prolonged hospital stays and increased mortality among ICU patients [11].
A. baumannii infection primarily presents as pneumonia and bloodstream infections (bacteremia) [12]. Additionally, it can lead to urinary tract infections, post-neurosurgery meningitis, wound infections following trauma or surgery, and osteomyelitis [12]. A meta-analysis, comprising data from 27 worldwide studies, reported an overall mortality rate of 42.6% for A. baumannii causing hospital-acquired and ventilator-associated pneumonia [13]. Among A. baumannii-related infection, bloodstream infections is the most severe clinical type, with attributable mortality as high as 58.24% in cases of CR-AB bacteremia [14]. Several studies have explored risk factors associated with antibiotic- resistant A. baumannii infection, identifying factors such as prolonged hospital stay, current ICU admission, immunosuppression, advanced age, comorbidities, major trauma or burns, prior antibiotic use, invasive procedures, and indwelling catheterization or mechanical ventilation [15,16,17,18,19,20,21,22,23]. In a study by Kim et al. which analyzed 441 colonized patients in the ICU over a 7-year period, endotracheal intubation (odds ratio [OR], 5.88), ventilator support (OR, 3.70), and central venous catheterization (OR, 3.48) were identified as risk factors of bacteremia among patients colonized by MDR-AB [24]. However, little is known about the factors contributing to the progression from focal infection to bacteremia in patients infected with A. baumannii.
A. baumannii is a common pathogen causing nosocomial infections in ICUs in our hospital. However, despite similar clinical procedures being administered to all ICU patients, such as central venous catheterization and mechanical ventilation, we have observed that some patients infected with CR-AB are limited to pulmonary infections, while others are more prone to developing bacteremia. As a result, we hypothesized that, in addition to invasive procedures, there may be other risk factors contributing to bacteremia in CR-AB infected patients. Therefore, this study aimed to investigate the independent factors associated with progression from nosocomial pneumonia to bacteremia, as well as the mortality factors in CR-AB patients.
Material and methods
Study subjects
A retrospective study was conducted in a teaching hospital in southern China, equipped with a 62-bed intensive care unit (ICU). We retrospectively analyzed the medical records of patients diagnosed with nosocomial CR-AB pneumonia from January 2019 to December 2021.
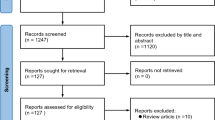
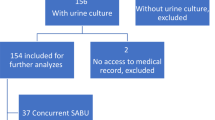
We identified a total of 159 cases, which were divided into two groups for comparison, 40 cases with CR-AB pneumonia associated with bacteremia and 119 cases without bacteremia. The inclusion criteria were: 1) meeting the 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society for nosocomial pneumonia infection and bacteremia [25]; 2) detection of CR-AB through sputum or bronchoalveolar lavage culture conducted more than 48 h after ICU admission; 3) pneumonia-associated bacteremia, defined as the detection of an A. baumannii strain in peripheral blood culture for more than 48 h along with a positive sputum or bronchoalveolar lavage culture, excluding cases with positive transcatheter tip culture only, and no fever or systemic symptoms [26]; 4) aged ≥ 18 years. The exclusion criteria were: incomplete clinical data. This study was approved by the institutional review board of our hospital (Ethical Approval K2021-113). We obtained verbal informed consent from the patients themselves or the their immediate family through a telephone interview.
Data collection
All data were collected from the medical records of 159 nosocomial CR-AB pneumonia patients. The baseline demographic and clinical characteristics included gender, age, APACHE II score at ICU admission, duration of ICU stay, and comorbidities such as hypertension, diabetes, coronary heart disease, chronic heart failure, cerebrovascular disease, chronic kidney disease, chronic liver disease, solid tumors, hematological diseases, and chronic obstructive pulmonary disease.
The treatment-related data were recorded when CR-AB was detected in the sputum or bronchoalveolar lavage samples of the patients. These data included the use of carbapenem, antibiotics, antifungal therapy, acid suppressants, glucocorticoids, continuous veno-venous hemofiltration (CVVH), vasopressors, as well as the durations of mechanical ventilation and central venous catheterization.
The blood biochemical examination data were all recorded within 24 h of detecting CR-AB in the sputum or bronchoalveolar lavage of the patients. These data included white blood cell (WBC) count, neutrophilic granulocyte (NE) count, lymphocyte (L) count, platelet (PLT) count, procalcitonin (PCT) level, interleukin-6 (IL-6) level, C-reactive protein (CRP) level, albumin (ALB) level, prealbumin (PA) level.
Statistical analysis
Continuous data were presented as mean ± standard deviation (SD) while categorical data were reported with number and percentage (%). For comparisons of means between groups, we used either the Student’s independent t-test or Mann-Whitney U test, depending on the normality assumption. Categorical data were assessed using the Chi-square test or Fisher’s exact test (if the expected value was ≤ 5).
We employed univariate and multivariable logistic regression models to investigate the association between independent variables and dichotomous outcomes, which included bloodstream infection and 28-day mortality. Independent variables found to be significant in the univariate analysis were entered into the multivariable model. Additionally, independent variables that remained significant in the multivariable model were identified as associated factors for the dichotomous outcomes. In cases of multicollinearity, we conducted correlation coefficient analyses, including Pearson’s correlation coefficient and point-biserial correlation coefficient, to explore the relationships among independent variables. We considered p ≤ 0.05 as indicating statistical significance for each test, with two-tailed analysis. All the analyses mentioned above were performed using IBM SPSS Version 25 (SPSS Statistics V25, IBM Corporation, Somers, New York).
Results
Patient clinical characteristics
A total of 159 nosocomial CR-AB pneumonia patients were included (mean age = 67.90 ± 14.54 years, 121 males and 38 females). The mean length of stay in the ICU was 25.21 ± 30.46 days, and the Apache II score was 23.62 ± 6.92. Among these patients, 40 developed pneumonia-associated bacteremia, with a mean time from positive sputum or bronchoalveolar lavage samples to bacteremia of 7.46 ± 6.24 days (Fig. 1).
Table 1 compares demographic and clinical characteristics between the two patient groups. The group with pneumonia-related bacteremia exhibited higher rates of CKD, CRP, PCT, IL-6 levels, and infection rates with other bacteria than the pneumonia-only group, while PA levels were lower (all p ≤ 0.05, Table 1). Additionally, patients with pneumonia-associated bacteremia had significantly higher rates of antifungal drug use, CVVH utilization, vasopressor administration, and a greater number of antibiotics, along with prolonged mechanical ventilation times (all p ≤ 0.05, Table 1).
Independent factors associated with bloodstream infection in CR-AB patients
To investigate the factors associated withpneumonia-associated bacteremia, both univariate and multivariable logistic regression models were performed. As shown in Table 2, a higher level of CRP (OR = 1.010, 95% CI = 1.007 to 1.013; p ≤ 0.05) and the utilization of CVVH treatment (OR = 2.93, 95% CI = 1.13 to 7.59; p ≤ 0.05) were found to be associated with an elevated risk of bacteremia.
The highest correlation coefficient among independent variables that were significant in univariate results was 0.60. Thus, no multicollinearity was observed.
28-day mortality
Out of the 159 nosocomial CR-AB pneumonia patients, 38 (23.90%) passed away within 28 days following the definitive diagnosis. The 28-day mortality rate was significantly higher among patients with pneumonia-associated bacteremia compared to those with pneumonia alone (47.50% vs. 15.97%, p ≤ 0.05).
Independent factors associated with mortality in bloodstream-infected CR-AB patients
A subgroup analysis stratified by survival status among those with bacteremia demonstrated that patients who died within 28 days had a significantly higher level of IL-6, a higher rate of use of antifungal drugs, and a longer duration of mechanical ventilation (all p ≤ 0.05, Table 3).
To identify factors associated with 28-day mortality in bacteremia patients, both univariate and multivariable logistic regression models were performed. The results indicated that the use of antifungal drugs was associated with a higher 28-day mortality rate compared to patients without antifungal drug treatment (OR = 4.70, 95% CI = 1.11 to 19.95; p ≤ 0.05, Table 4).
Discussion
We conducted a retrospective study to investigate the independent factors associated with the progression from nosocomial pneumonia to pneumonia-related bacteremia in patients with CR-AB. Our study included a total of 159 ICU patients with CR-AB, of which 40 (25.15%) progressed to pneumonia-related bacteremia. This suggests that the progression from CR-AB pneumonia to pneumonia-related bacteremia is not uncommon. Furthermore, our findings indicate that patients with CR-AB pneumonia-related bacteremia have a significantly higher 28-day mortality rate when compared to the CR-AB pneumonia group, suggesting a worse clinical prognosis.
In previous studies, various risk factors for acquiring antibiotic-resistant A. baumannii bacteremia have been identified, including male gender, prior ICU stay, the use of cefoperazone–sulbactam, carbapenem, penicillins, endotracheal intubation, prior A. baumannii colonization, and cardiovascular failure [27,28,29,30]. In our univariate analysis, we identified risk factors for nosocomial CR-AB pneumonia-associated bacteremia, including the use of antibiotics, extended mechanical ventilation time, and bacteremia with other pathogens. Some of these findings were consistent with previous research [24, 31]. However, in the multivariable analysis, these variables did not reach significance, which may be attributed to the relatively small sample size and inconsistent characteristics of the included population, as tracheal intubation and central venous catheterization were performed in all patients in this study. In addition, discrepancies in patient characteristics and the variables controlled for in the multivariable analysis may also contribute to the differing findings between this study and previous ones.
In this study, CR-AB patients with pneumonia-associated bacteremia had a significantly higher level of CRP, a higher rate of CVVH treatment, and a longer ventilation time compared to those without bacteremia (all p ≤ 0.05). Furthermore, multivariable logistic regression analysis revealed that higher CRP level (OR = 1.01) and CVVH treatment (OR = 2.93) were associated with an increased risk of bacteremia. These variables have not previously been reported as factors for pneumonia-associated bacteremia in CR-AB patients. It's worth noting that CVVH treatment involves the placement of a vascular catheter, which, like other invasive procedures, can serve as a potential source of infection, thereby increasing the risk of bacteremia. CRP, on the other hand, serves as a marker of infections. In a study on bacteremia in community-acquired pneumonia, elevated CRP followed by lower plasma albumin was found to predict a higher risk of community-acquired bacteremia [32]. In addition, Ho et al. conducted a case-control study and found that CRP was highly specific for predicting bacteremia in critically ill patients [33]. In our study, the measurement of CRP and the time to diagnose pneumonia-related bacteremia differed from previous findings. On the one hand, this variation can be attributed to the limitations of retrospective studies, where blood cultures were not systematically collected; on the other hand, the diagnosis of bacteremia through the isolation of microorganisms from blood cultures proved to be a time-consuming process, with a positive reaction time ranging from 24 to 48 h. These two factors could contribute to delays in both CRP measurement and bacteremia diagnosis. CRP reflects the degree of inflammation in the body and can remain elevated for extended periods during ongoing inflammation. Higher CRP values indicated a more severe infection and predict a higher risk of progression from a focal to a systemic infection. This suggests that clinicians should monitor such patients more frequently and intervene early.
Several factors associated with mortality in patients with CR-AB infection have been reported, including old age, ICU stay after bacteremia, readmission within 90 days, tigecycline therapy, septic shock, multiple organ failure, a high Pitt bacteremia score, bacteremia following severe pneumonia, inappropriate empirical antimicrobial treatment, septic shock, chronic liver disease, chronic renal disease, hypertension, neutropenia, immunosuppressant use, and intubation [27, 30, 34]. These findings suggest that severity of baseline condition and inappropriate antibiotic therapy are the primary factors contributing to mortality in patients with CR-AB infections. In this study, among the 159 nosocomial CR-AB pneumonia patients in ICU, 38 (23.90%) patients died within 28 days after the definitive diagnosis. Notably, patients with bacteremia had a significantly higher 28-day mortality rate compared to those without bacteremia (47.50% vs. 15.97%), which is in agreement with the findings of Zhou et al. that bacteremia following severe pneumonia is a risk factor for MDR-AB bacteremia-related mortality [30]. Our results further revealed that among patients with pneumonia-associated bacteremia, those who died within 28 days exhibited significantly higher levels of IL-6, a higher rate of antifungal drug usage, and prolonged mechanical ventilation. The elevated IL-6 levels in bacteremia patients may be attributed, in part, to the higher rate of chronic kidney disease in this group, as it is well-known that CKD patients tend to exhibit increased IL-6 production and decreased clearance [35]. Univariate analysis showed that the use of antifungal drugs and prolonged mechanical ventilation time were associated with 28-day mortality in CR-AB patients. However, multivariable logistic regression demonstrated that the use of antifungal drugs was the only factor that associated with 28-day mortality (OR = 4.70). In our study, most antifungal drugs were used empirically based on the 2016 guideline and assessment scale for ICU patients issued by the Infectious Diseases Society of America [36]. The relatively low rate of fungal infection detection might be attributed to the subtle clinical manifestations of some critically ill patients and -limitation of detection. Nonetheless, the use of antifungal drugs still serves as an indicator of infection severity of infection in patients. This association with mortality should be further validated through future prospective studies. Due to our limited data, we cannot exclude the influence of other factors, such as disease severity, disease progression, and individualized treatment differences. The most significant challenge in treating A. baumannii infection is MDR, which negatively impacts mortality, length of hospital stays, and healthcare costs. A matched-controlled study demostrated that MDR increased mortality from 13 to 34% and extended hospital stay and health care costs in patients with A. baumannii bacteremia [37]. Park et al. identified that underlying malignancy, the need for mechanical ventilation, and CR-AB infection as risk factors associated with higher mortality in patients with A. baumannii bacteremia [38]. However, given that all patients included in this study were infected with CR-AB and required mechanical ventilation, we couldn't assess the individual impact of carbapenem resistance and mechanical ventilation.
We acknowledge several limitations in this study. Firstly, it was single-center study with a relatively small sample size, which may impact the generalizability of the results. Secondly, the lack of systematic blood culture collection for all CR-AB pneumonia patients poses a risk of misclassification between the two groups. Lastly, although our study identify a relationship between CRP and the progression of CR-AB pneumonia to associated bacteremia, we could not analyze the impact of changes in indicators over time or disease evolution, etc. on the final outcome. Future validation through multicenter, large-sample size prospective studies is warranted.
Conclusion
In summary, this study revealed that among ICU ventilated patients with a central venous catheter, who developed CR-AB pneumonia, a higher level of CRP and the use of CVVH treatment were identified as risk factors for nosocomial pneumonia-associated bacteremia. Additionally, among patients with nosocomial pneumonia-associated bacteremia, the use of antifungal drugs emerged as a factor that associated with 28-day mortality. These findings hold the potential to inform recommendations for preventive and therapeutic guidelines for patients with CR-AB infections.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Asif M, Alvi IA, Ur Rehman S (2018) Insight into Acinetobacter baumannii: Pathogenesis, global resistance, mechanisms of resistance, treatment options, and alternative modalities. Infect Drug Resist 11:1249–1260
Vázquez-López R, Solano-Gálvez SG, Vignon-Whaley JJJ, Vaamonde JAA, Alonzo LAP, Reséndiz AR et al (2020) Acinetobacter baumannii resistance: A real challenge for clinicians. Antibiotics 9(4):205
Liu W, Yang Y, Zhang K, Hai Y, Li H, Jiao Y et al (2020) Drug resistance of healthcare-associated pathogenic bacteria and carbapenem-resistant Acinetobacter baumannii homology in the general intensive care unit. Ann Cardiothorac Surg 9(4):1545–1555
Jiang W, Li L, Wen S, Song Y, Yu L, Tan B (2022) Gram-negative multidrug-resistant organisms were dominant in neurorehabilitation ward patients in a general hospital in southwest China. Sci Rep 12(1):11087
Oliva A, Ceccarelli G, De Angelis M, Sacco F, Miele MC, Mastroianni CM et al (2020) Cefiderocol for compassionate use in the treatment of complicated infections caused by extensively and pan-resistant Acinetobacter baumannii. J Glob Antimicrob Resist 23:292–296
Piperaki ET, Tzouvelekis LS, Miriagou V, Daikos GL (2019) Carbapenem-resistant Acinetobacter baumannii: in pursuit of an effective treatment. Clin Microbiol Infect 25(8):951–957
Da Silva KE, Maciel WG, Croda J, Cayô R, Ramos AC, De Sales RO et al (2018) A high mortality rate associated with multidrug-resistant Acinetobacter baumannii ST79 and ST25 carrying OXA-23 in a Brazilian intensive care unit. PLoS ONE 13(12):e0209367
Teerawattanapong N, Panich P, Kulpokin D, Na Ranong S, Kongpakwattana K, Saksinanon A et al (2018) A systematic review of the burden of multidrug-resistant healthcare-associated infections among intensive care unit patients in southeast asia: The rise of multidrug-resistant Acinetobacter baumannii. Infect Control Hosp Epidemiol 39(5):525–533
Gales AC, Castanheira M, Jones RN, Sader HS (2012) Antimicrobial resistance among Gram-negative bacilli isolated from Latin America: Results from SENTRY Antimicrobial Surveillance Program (Latin America, 2008–2010). Diagn Microbiol Infect Dis 73(4):354–360
Kiratisin P, Chongthaleong A, Tan TY, Lagamayo E, Roberts S, Garcia J et al (2012) Comparative in vitro activity of carbapenems against major Gram-negative pathogens: Results of Asia-Pacific surveillance from the COMPACT II study. Int J Antimicrob Agents 39(4):311–316
Maragakis LL, Perl TM (2008) Acinetobacter baumannii: Epidemiology, antimicrobial resistance, and treatment options. Clin Infect Dis 46(8):1254–1263
Moubareck CA, Halat DH (2020) Insights into Acinetobacter baumannii: A review of microbiological, virulence, and resistance traits in a threatening nosocomial pathogen. Antibiotics 9(3):119
Mohd Sazlly Lim S, Zainal Abidin A, Liew SM, Roberts JA, Sime FB (2019) The global prevalence of multidrug-resistance among Acinetobacter baumannii causing hospital-acquired and ventilator-associated pneumonia and its associated mortality: A systematic review and meta-analysis. J Infect 79(6):593–600
Liu CP, Shih SC, Wang NY, Wu AY, Sun FJ, Chow SF et al (2016) Risk factors of mortality in patients with carbapenem-resistant Acinetobacter baumannii bacteremia. J Microbiol Immunol Infect 49(6):934–940
Jang TN, Lee SH, Huang CH, Lee CL, Chen WY (2009) Risk factors and impact of nosocomial Acinetobacter baumannii bloodstream infections in the adult intensive care unit: a case-control study. J Hosp Infect 73(2):143–150
Martín-Aspas A, Guerrero-Sánchez FM, García-Colchero F, Rodríguez-Roca S, Girón-González JA (2018) Differential characteristics of Acinetobacter baumannii colonization and infection: Risk factors, clinical picture, and mortality. Infect Drug Resist 11:861–872
Blanco N, Harris AD, Rock C, Johnson JK, Pineles L, Bonomo RA et al (2017) Risk factors and outcomes associated with multidrug- resistant Acinetobacter baumannii upon intensive care unit admission. Antimicrob Agents Chemother 62(1):e01631-e1717
Sultan AM, Seliem WA (2018) Identifying risk factors for healthcare-associated infections caused by carbapenem-resistant Acinetobacter baumannii in a neonatal intensive care unit. Sultan Qaboos Univ Med J 18(1):e75–e80
Munier AL, Biard L, Legrand M, Rousseau C, Lafaurie M, Donay JL et al (2019) Incidence, risk factors and outcome of multi-drug resistant Acinetobacter baumannii nosocomial infections during an outbreak in a burn unit. Int J Infect Dis 79:179–184
Huang H, Chen B, Liu G, Ran J, Lian X, Huang X et al (2018) A multi-center study on the risk factors of infection caused by multi-drug resistant Acinetobacter baumannii. BMC Infect Dis 18(1):11
Aydemir H, Tüz HI, Piskin N, Celebi G, Kulah C, Kokturk F (2019) Risk factors and clinical responses of pneumonia patients with colistin-resistant Acinetobacter baumannii-calcoaceticus. World J Clin Cases 7(10):1111–1121
Oh DH, Kim YC, Kim EJ, Jung IY, Jeong SJ, Kim SY et al (2019) Multidrug-resistant Acinetobacter baumannii infection in lung transplant recipients: risk factors and prognosis. Infect Dis (Auckl) 51(7):493–501
Al-Gethamy MM, Faidah HS, Adetunji HA, Haseeb A, Ashgar SS, Mohanned TK et al (2017) Risk factors associated with multi-drug-resistant Acinetobacter baumannii nosocomial infections at a tertiary care hospital in Makkah, Saudi Arabia - a matched case–control study. J Int Med Res 45(3):1181–1189
Kim SY, Cho SL, Bang JH (2020) Risk factors associated with bloodstream infection among patients colonized by multidrug-resistant Acinetobacter baumannii: A 7-year observational study in a general hospital. Am J Infect Control 48(5):581–583
Kalil AC, Metersky ML, Klompas M, Muscedere J, Sweeney DA, Palmer LB et al (2016) Management of Adults with Hospital-acquired and Ventilator-associated Pneumonia: 2016 ClinicalPractice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis 63(5):e61–e111
Teng SO, Yen MY, Ou TY, Chen FL, Yu FL, Lee WS (2015) Comparison of pneumonia- and non-pneumonia-related Acinetobacter baumannii bacteremia: Impact on empiric therapy and antibiotic resistance. J MicrobiolImmunol Infect 48(5):525–530
Niu T, Xiao T, Guo L, Yu W, Chen Y, Zheng B et al (2018) Retrospective comparative analysis of risk factors and outcomes in patients with carbapenem-resistant Acinetobacter baumannii bloodstream infections: Cefoperazone–sulbactam associated with resistance and tigecycline increased the mortality. Infect Drug Resist 11:2021–2030
Liu Y, Wang Q, Zhao C, Chen H, Li H, Wang H et al (2020) Prospective multi-center evaluation on risk factors, clinical characteristics and outcomes due to carbapenem resistance in Acinetobacter baumannii complex bacteraemia: Experience from the Chinese Antimicrobial Resistance Surveillance of Nosocomial Infections (CARES) Network. J Med Microbiol 69(7):949–959
Zhou H, Yao Y, Zhu B, Ren D, Yang Q, Fu Y et al (2019) Risk factors for acquisition and mortality of multidrug-resistant Acinetobacter baumannii bacteremia: A retrospective study from a Chinese hospital. Medicine (Baltimore) 98(13):e14937
Baran G, Erbay A, Bodur H, Öngürü P, Akinci E, Balaban N et al (2008) Risk factors for nosocomial imipenem-resistant Acinetobacter baumannii infections. Int J Infect Dis 12(1):16–21
Gu Y, Jiang Y, Zhang W, Yu Y, He X, Tao J et al (2021) Risk factors and outcomes of bloodstream infections caused by Acinetobacter baumannii: a case-control study. Diagn Micr Infect Dis 99(2):115229
Garvik OS, Póvoa P, Magnussen B, Vinholt PJ, Pedersen C, Jensen TG et al (2020) C-reactive protein and albumin kinetics before community-acquired bloodstream infections- A Danish population-based cohort study. Epidemiol Infect 148:e38
Ho KM, Towler SC (2009) A comparison of eosinopenia and C-reactive protein as a marker of bloodstream infections in critically ill patients: A case control study. Anaesth Intensive Care 37(3):450–456
Du X, Xu X, Yao J, Deng K, Chen S, Shen Z et al (2019) Predictors of mortality in patients infected with carbapenem-resistant Acinetobacter baumannii: A systematic review and meta-analysis. Am J Infect Control 47(9):1140–1145
Su H, Lei CT, Zhang C (2017) Interleukin-6 Signaling Pathway and Its Role in Kidney Disease: An Update. Front Immunol 8:405
Pappas PG, Kauffman CA, Clancy CJ, Marr KA, Ostrosky-Zeichner L, Reboli AC et al (2016) Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clin Infect Dis 62(4):e1-50
Park SY, Choo JW, Kwon SH, Yu SN, Lee EJ, Kim TH et al (2013) Risk factors for mortality in patients with Acinetobacter baumannii bacteremia. Infect Chemother 45(3):325–330
Lee NY, Lee HC, Ko NY, Chang CM, Shih HI, Wu CJ et al (2007) Clinical and Economic Impact of Multidrug Resistance in Nosocomial Acinetobacter baumannii Bacteremia. Infect Control Hosp Epidemiol 28(6):713–719
Acknowledgements
We would like to thank the members of our microbiology testing center.
Funding
This study was supported by the Special Fund for the Construction of High-level Hospitals in Guangdong Province (G330102097011).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. HN, XS and JL conceived and designed the study. HN, SC, QS and XS collected the data. MC, HL and GW analyzed the data. HN, XS and MC drafted the manuscript. JL, KZ reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
The studies was approved by the Ethical Committee of Zhongshan People’s Hospital (grant number: K2021-113).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Niu, H., Shen, X., Liang, H. et al. Risk factors for progression to bacteremia among patients with nosocomial carbapenem-resistant Acinetobacter baumannii pneumonia in the Intensive Care Unit. Eur J Clin Microbiol Infect Dis 42, 1337–1346 (2023). https://doi.org/10.1007/s10096-023-04668-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-023-04668-9