Abstract
A checklist is an effective implementation tool, but addressing barriers that might impact on the effectiveness of its use is crucial. In this paper, we explore barriers to the uptake of an antibiotic checklist that aims to improve antibiotic use in daily hospital care. We performed an online questionnaire survey among medical specialists and residents with various professional backgrounds from nine Dutch hospitals. The questionnaire consisted of 23 statements on anticipated barriers hindering the uptake of the checklist. Furthermore, it gave the possibility to add comments. We included 219 completed questionnaires (122 medical specialists and 97 residents) in our descriptive analysis. The top six anticipated barriers included: (1) lack of expectation of improvement of antibiotic use, (2) lack of expected patients’ satisfaction by checklist use, (3) lack of feasibility of the checklist, (4) negative previous experiences with other checklists, (5) the complexity of the antibiotic checklist and (6) lack of nurses’ expectation of checklist use. Remarkably, 553 comments were made, mostly (436) about the content of the checklist. These insights can be used to improve the specific content of the checklist and to develop an implementation strategy that addresses the identified barriers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
A better use of current antibiotic agents is necessary to help control antimicrobial resistance (AMR) [1]. Antibiotic stewardship programs (ASPs) are introduced to coordinate activities to measure and improve appropriate antibiotic use in daily hospital practice [2]. Recently, a set of generic quality indicators (QIs) was developed to measure appropriate antibiotic use in the treatment of bacterial infections in adult patients hospitalised at non-intensive care unit departments [3]. The next step is using these QIs to improve antibiotic use in daily practice. Checklists are effective tools to improve patient care [4–7]. For example, the use of a surgical safety checklist resulted in an absolute risk reduction in perioperative complications of 10.6 % [4], and a large and sustained reduction in rates of catheter-related bloodstream infections was demonstrated after the implementation of an infection control checklist [5]. It can, therefore, be hypothesised that the use of an antibiotic checklist consisting of generic quality indicators (Box 1) improves antibiotic use in the hospital. However, the implementation of a checklist needs to be combined with an understanding of barriers to its uptake, since it has been shown that physicians have resistances and interferences to the use of checklists [8, 9]. Without enough attention for such barriers that hinder implementation, the use of a checklist may fail, even where other studies showed striking improvements using the same checklist [9, 10]. Ideally, potential barriers hindering uptake are analysed before implementation, to influence both the type and content of the implementation activities [11]. In general, implementation can be complicated by barriers concerning the innovation itself, the individual professional, professional interactions, the patient, the organisation, incentives, resources or the socio-political context [12, 13]. Although addressing barriers that influence the effectiveness of an innovation to improve patient care is a crucial step in the knowledge-to-action process [14], very few barrier studies have been done prior to checklist implementation [15].
Box 1: The antibiotic checklist based on generic quality indicators
1 | Take at least two sets of blood cultures before starting systemic antibiotic therapy. |
2 | Take specimens for culture from suspected sites of infection, if possible before starting systemic antibiotic therapy, but at the latest after 24 hours of treatment. |
3 | Prescribe systemic antibiotic treatment according to the local antibiotic guideline. |
4 | a. Determine renal function. |
b. Adapt dose and dosing interval of systemic antibiotics to renal function if necessary. | |
5 | Document the antibiotic treatment in the case notes or electronic medical record (EMR), including: - Indication; - Name; - Dose; - Interval; - Route of administration. |
6 | Determine whether antibiotic therapy can be adapted as soon as culture results become available. |
7 | Switch from intravenous to oral antibiotic therapy after 48–72 hours on the basis of the clinical condition, provided that oral treatment is adequate.a |
a Adequate means:
1: When the antibiotic is available orally;
2: When oral intake and gastrointestinal absorption are adequate;
3: Adequate in terms of diagnosis (exceptions are e.g. endocarditis, meningitis).
Barrier studies performed during or after checklist implementation [16–24] described barriers such as lack of understanding the purpose of the checklist [17], duplication with current work [18, 19] and problems with the method of implementation [24].
The present study aims to identify barriers to the uptake of an antibiotic checklist in Dutch hospitals prior to checklist implementation and to select implementation activities to target the predominant barriers that obstruct checklist uptake.
Methods
We performed an online questionnaire survey among medical specialists and residents to explore anticipated barriers hindering the uptake of an antibiotic checklist.
Development of the antibiotic checklist barrier questionnaire
We based our questionnaire on the Dutch validated measurement instrument for determinants of innovations (MIDI), combined with barriers found in the literature. The MIDI is developed by the Netherlands Organisation for Applied Scientific Research (TNO) and is meant as a tool for researchers to survey determinants that influence the uptake of an innovation [13]. Additionally, we performed a literature search to find publications on barriers to checklist implementation and to appropriate antibiotic use. Box 2 shows the terms we used in our search, which resulted in 168 hits in total. We selected one book [25], three systematic reviews [12, 26, 27], eight relevant barrier studies [15–22, 24] and four studies on barriers to the appropriate use of antibiotics [28–31]. Based on this information, we adapted the MIDI to fit the topic of antibiotic use, so we removed potential barriers that were not relevant and added barriers that were mentioned in the literature.
Box 2: Terms for literature search
Topic | Search terms in title | Hits |
Reviews on barriers | (checklist* OR guideline*) AND (determinant* OR barrier* OR factor*) Filter: systematic reviews | 130 |
Barriers to checklist implementation | (barrier* OR facilitator* OR determinant* OR challenge*) AND checklist* | 13 |
Barriers to appropriate antibiotic use | (antibiotic* OR antimicrobial* OR antibacterial*) AND (barrier* OR behaviour* OR attitude*) AND (appropriate* [Title/Abstract] OR guideline* [Title/Abstract]) | 25 |
The final online questionnaire started with a description of the antibiotic checklist (Box 1) and was followed by 23 statements on anticipated barriers related to the checklist (seven items), the individual professional (six items), professional interactions (seven items), the patient (two items) and to resources (one item) (see Table 1 for the specific statements). To diminish the influence of the physician’s criticisms on the content of the checklist, statements 8 through 23 started with the sentence “Assuming that the checklist is adapted to your comments on its contents”. The level of agreement or disagreement with the statements was measured by a six-point Likert scale (1 = ‘totally agree’ and 6 = ‘totally disagree’). For each statement, it was possible to choose a seventh option: ‘I don’t know’.
Furthermore, physicians could criticise the separate components of the checklist by adding comments, and there was also space for general or organisational comments. The questionnaire was completed anonymously, but we asked for the participant’s function, department and hospital.
Setting and participants
To gain insight into anticipated barriers to the uptake of an antibiotic checklist in hospitals prior to checklist implementation, physicians in nine Dutch hospitals were invited to participate in the survey. These nine hospitals, including two university and seven non-university hospitals, previously agreed to participate in a cluster-randomised trial on the implementation of the antibiotic checklist [32].
We visited the hospitals to inform the local antibiotic stewardship team about the antibiotic checklist and the questionnaire. Following this visit, we emailed the contact physician a link to the questionnaire, and he/she forwarded this email to the target group. The target group consisted of specialists and residents—with all levels of experience and various professional backgrounds—who have direct contact with and prescribe antibiotics to adult patients.
Analysis
We included questionnaires in the analysis if at least half of the statements were appraised. We excluded statements from further analyses if ≥30 % of the participants answered ‘I don’t know’. While taking into account whether the statement was formulated as a barrier hindering uptake (“This checklist is a threat to my professional autonomy”) or as a facilitator helping uptake (“I expect that this checklist will improve the quality of my antibiotic prescriptions”), all answers (1 through 6) were re-coded into dichotomous scores: anticipated barrier ‘yes’ or ‘no’. The answers ‘I don’t know’ were excluded from the analyses. We computed frequencies and percentages and created a top five of the statements that were most often mentioned as barriers.
We categorised the comments on the checklist added by the participants. If comparable comments were mentioned three times or more, the comment was considered to be relevant. We also created a top five of comments.
Results
Participants
The online questionnaire was filled out by participants in eight of the nine hospitals that initially agreed to participate in the cluster-randomised trial on the implementation of the antibiotic checklist. One non-university hospital no longer wanted to participate and was replaced by a similar hospital. In another hospital, the link to the questionnaire was only emailed to physicians of the department of infectious diseases. In total, 250 physicians participated in the survey, of which 219 participants completed 50 % or more of the questionnaire statements. These 219 questionnaires were included in the analyses. The participants’ characteristics are summarised in Table 2. The number of completed questionnaires per hospital ranged from 8 to 90.
Barriers
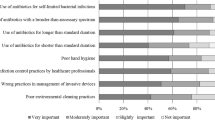
Table 1 shows the survey results. The statement concerning the availability of sufficient financial resources to use the checklist was excluded from further analysis, as more than 30 % of the participants answered ‘I don’t know’. The top five anticipated barriers were: (1) lack of expected quality improvement of the physician’s antibiotic prescribing (26.9 %), (2) lack of expected patients’ satisfaction with checklist use (21.8 %), (3) lack of feasibility of the checklist (20.8 %), (4) negative previous experiences with other checklists (19.9 %) and (5) the complexity of the antibiotic checklist (17.4 %).
To exclude the possibility that the single hospital in which 90 physicians completed the questionnaire influenced the results disproportionally, we compared the appraisals of the 219 participants (nine hospitals) with the appraisals of 129 participants (eight hospitals). The top five anticipated barriers from these eight hospitals differed on one statement: instead of ‘complexity of the checklist’, the statement ‘nurses will expect me to use the checklist’ was in the top five. For this reason, this barrier was added to the list of frequently mentioned barriers (6). This top six contains barriers from four different domains, namely the individual professional (1 and 4), the patient (2), the checklist (3 and 5) and professional interactions (6).
Comments
In total 553, comments were given, of which 436 were comments and suggestions regarding the content of the checklist, 59 were general comments and 58 were organisational comments. These organisational comments described contextual factors that should be taken into account in these specific hospitals, i.e. implementation of a new electronic medical record (EMR) system (14.6 %) and merger of the hospital with another hospital (7.3 %). Comments on the content of the checklist or general comments that were relevant for all hospitals and were mentioned at least three times are presented in Table 3. The five most frequently mentioned comments were: (1c) “the item documentation leads to duplication of work” (11.0 %), (2c) “doubts about the need of blood cultures for several diagnoses” (10.5 %), (3c) “incomplete or too simplistic clarification of ‘adequate in terms of diagnosis’ for the item IV o oral switch” (8.2 %), (4c) “add information about the renal function” (6.8 %) and (5c) “add that one should take different sites for taking two different blood cultures” (6.4 %). Again, we compared the overall top five with the top five after exclusion of the 90 questionnaires of the single (university) hospital, which showed that the top five of the eight hospitals was equal to the overall top five comments.
Addressing identified barriers
We developed an implementation strategy that could be applied in all hospitals to address the top six anticipated barriers.
The barrier (1) “lack of expected quality improvement of the physician’s antibiotic prescribing” can be addressed in the following two ways: first by showing the room for improvement, i.e. giving feedback on their current antibiotic use based on a baseline measurement, and second by providing evidence for a reduction in the length of hospital stay for the patient with adequate antibiotic use [33]. This information can be given during a kick-off lecture at the departments that participate in the cluster-randomised trial on the implementation of the antibiotic checklist [32].
The barrier (2) “lack of expected patients’ satisfaction with checklist use” can be addressed by giving information about the study at the nursery department, i.e. by supplying flyers about antimicrobial resistance and the expected effects of appropriate antibiotic use [33].
Two anticipated barriers, namely (3) “the feasibility of the checklist” and (5) “the complexity of the checklist”, can be addressed by adapting the checklist. Table 4 shows the adapted checklist based on the survey results, which includes tick boxes and pre-printed options.
To address the barrier (4) “negative previous experiences with other checklists”, the differences between these previous checklists (most probably the surgical safety checklist) and this antibiotic checklist should be emphasised, for example in group discussions. Differences are, e.g. that this checklist is short and does not involve other physicians to complete it. In addition, it should be stressed that the antibiotic checklist was adapted based on their comments and the comments of other colleagues participating in the trial.
Finally, to address the barrier (6) “lack of nurses’ expectation of checklist use”, nurses working at participating departments should be informed about the checklist use and its aims, for example through email, e-learnings and/or the department lecture showing the room for improvement and the evidence for reduction in the length of hospital stay for the patient with adequate antibiotic use.
Incorporating comments
We adapted the checklist design based on the top five comments. First (1c), to reduce duplication of documentation, we left out the name, dose, interval and route of administration of the antibiotic, and the item was reduced to ‘documentation of indication’. This is appropriate since all Dutch hospitals are, nowadays, obliged to work with an electronic medical record that already requires this information. Second (2c), since 10.5 % of the participants felt that taking blood cultures was not necessary for all diagnoses, we added the option “In my opinion, not necessary with this diagnosis” in the checklist. Third (3c), we left out the clarification of “adequate in terms of diagnosis” for the item IV to oral switch, and included options in the checklist so that the physicians could tick their reasons for not switching: “Insufficient clinical improvement”; “No oral antibiotic available”; “No adequate oral intake/gastrointestinal absorption” and “No oral therapy possible with this diagnosis”. In addition, we ensured that the information on switch criteria could be easily found: we developed informative posters and laminated pocket versions of the checklist, which included a reference to the website with all necessary information concerning this topic. The information about dosage adaptation when the patient has an impaired renal function (4c) can also be found on this website. Last (5c), we added in the checklist that, for taking two different blood cultures, one should use two different sites.
Discussion
In this study, we identified the barriers that need to be addressed when implementing an antibiotic checklist. We created a top six of anticipated barriers to the uptake of the checklist, and a top five of comments on its components. The top six barriers encompassed four different domains, namely barriers related to the individual professional, to the patient, to professional interactions and to the checklist itself. The top five comments mostly encompassed suggestions to more clearly specify the content of the checklist, i.e. the quality indicators included. We adapted the checklist to the survey results; two barriers and all top five comments could be addressed by adapting the design of the checklist, and by re-phrasing various checklist items (Table 4).
Our most frequently mentioned barrier, the lack of expected improvement of care, has been described by several other barrier studies of checklist implementation [17, 18, 20].
Feasibility is also a known barrier in the implementation of checklists; items in the surgical safety checklist were perceived to be inappropriate for certain surgical procedures [16] or for certain settings [18]. Patient perceptions were only mentioned as a barrier by Russ et al., describing that too many checks can create anxiety and unsafe feelings towards the system [16]. This might be an explanation of our anticipated barrier “lack of patient’s satisfaction”. However, this anticipated barrier can also be explained by the fact that our antibiotic checklist is typically meant as a reminder for physicians; in daily practice, patients probably will not notice the use of the checklist. Next, the literature describes barriers comparable to our sixth anticipated barrier, “lack of nurses’ expectation of checklist use”, since it is associated with both the lack of teamwork [20, 21, 23, 24] and professional hierarchy [17, 18]. Not surprisingly, the barrier “negative previous experiences with other checklists” was not mentioned in previous studies, as those evaluations were performed after the introduction of the checklists, and our inventory was performed before checklist implementation. The complexity of the checklist was once earlier described as a barrier after the implementation of a quality improvement checklist on an inpatient hepatology service [21]. It was, however, not a major barrier, as it was only mentioned by 1 of the 23 participants. The explanation for why this is a top five barrier in our survey and not in previous studies might be that other barrier studies have been done during or after checklist implementation. In that phase, the physicians’ main concerns are the organisational problems they are facing in daily practice and, consequently, these studies mainly describe logistic barriers, such as difficulties in timing (when to fill out the checklist) and lack of time [16–19, 21, 24], and professional barriers such as lack of senior support and professional hierarchy [17, 18, 20, 24]. We identified the barriers before implementation of the checklist, and the physicians, therefore, may have felt that they still had an influence on the contents and design of the checklist. Finally, it appears that the benefit of the use of checklists is not always clear [17–19] and a lack of understanding of the purpose of the checklist might influence its use [17]. This topic was, however, not mentioned as a barrier in our study.
The current study has some strengths. To our knowledge, our study is the first to assess and quantify barriers prior to checklist implementation. We based our questionnaire on a validated instrument to measure determinants of innovations, combined with a literature search. The barriers to the uptake of the antibiotic checklist were reported by a large sample of physicians from nine different hospitals. Furthermore, we included residents and specialists of surgical departments and medical departments, while previous barrier studies only focussed on people in the surgical [16, 18–20, 23, 24] or medical fields [15, 17, 21]. Finally, barrier identification prior to implementation resulted not only in the identification of concerns about the structure of the checklist and the introduction into the clinical workflow, but also concerns about the actual content of the checklist. Although the set of quality indicators has been developed in a RAND-modified Delphi procedure by experts [3], the comment “doubts about the need of blood cultures for several diagnoses” (2c) expresses disagreement among physicians about targeting “taking blood cultures” in the checklist. Disagreement about clinical measures targeted in the checklist can hinder implementation. Therefore, the option “no, in my opinion, not necessary with this diagnosis” was added in the checklist. The cluster-randomised trial [32] will show how often this option in the checklist will be used, and can help to determine whether taking blood cultures is part of agreed-upon standard concerning appropriate antibiotic use among frontline clinicians.
The most important limitation of our study is that the physicians of one university hospital completed 90 of the 219 questionnaires. We compared, however, the appraisals of the 219 participants (nine hospitals) with the appraisals of 129 participants (eight hospitals). This comparison resulted in the addition of a sixth anticipated barrier on nurses’ expectations regarding checklist use. During the implementation process, this barrier should also be addressed. A second limitation has to do with the chosen recruitment method. We emailed the contact physician a link to the questionnaire, and asked him/her to forward this email to the target group. Through this approach, we aimed to invite as many professionals as possible to participate in the questionnaire study. The downside of this approach is that the exact number of physicians that received the link to the questionnaire was unclear and, consequently, that it was impossible to determine the response rate. Based on an analysis in one hospital where we could retrieve information on the number of invitations sent, we estimated the response rate to be about 30 %, which is also found in other studies [34, 35]. Although the response rate is said to be important to determine the non-response bias, Willis et al. showed that increasing the response rate by additional re-contacts had little effect on the key data distribution and, therefore, they suggest in physician surveys to have a larger initial sample (as we did) and to accept a lower final overall response rate [36]. Furthermore, a barrier study prior to implementation also has its limits, as the appraisers lack experience in actually using the checklist. Finally, although the contents of our questionnaire were carefully chosen, some results might seem less informative after all. For the anticipated barrier concerning negative previous experiences, it remains unclear whether the problem with other checklists concerned, for example, the length of the checklist, the lack of time or no perceived benefit. Explicitly discussing this barrier in small groups of physicians—as suggested—might, however, provide insight into the underlying problem.
Performing a barrier analysis prior to checklist implementation creates the opportunity to address barriers in an early stage, and to adapt its design structure and content. This iterative survey resulted in a checklist adapted to the perceptions of frontline clinicians, which should facilitate implementation. The results of our barrier analysis may be specific for our setting and our checklist. Since our developed questionnaire is based, however, on a validated instrument and a literature search, we assume that the checklist questionnaire itself can be used for barrier identification for the implementation of other checklists.
This survey provides insight into the anticipated barriers that have to be addressed when implementing an antibiotic checklist in Dutch hospitals. Taking into account these factors that hinder the uptake of the antibiotic checklist, implementation in daily practice will be challenging. Education, feedback, involvement of the whole healthcare team in the implementation process and adaption of the checklist itself (Table 4) will be necessary to overcome the barriers hindering the uptake of the checklist and to improve the appropriate use of antibiotics. Our assumption that the suggested combination of interventions should facilitate successful checklist implementation will be tested in a cluster-randomised trial [32].
References
World Health Organization (WHO) Antimicrobial resistance. Home page at: http://www.who.int/drugresistance/en. Accessed 2 May 2015
MacDougall C, Polk RE (2005) Antimicrobial stewardship programs in health care systems. Clin Microbiol Rev 18:638–656
van den Bosch CM, Geerlings SE, Natsch S, Prins JM, Hulscher ME (2015) Quality indicators to measure appropriate antibiotic use in hospitalized adults. Clin Infect Dis 60:281–291
de Vries EN, Prins HA, Crolla RM, den Outer AJ, van Andel G, van Helden SH, Schlack WS, van Putten MA, Gouma DJ, Dijkgraaf MG, Smorenburg SM, Boermeester MA; SURPASS Collaborative Group (2010) Effect of a comprehensive surgical safety system on patient outcomes. N Engl J Med 363:1928–1937
Pronovost P, Needham D, Berenholtz S, Sinopoli D, Chu H, Cosgrove S, Sexton B, Hyzy R, Welsh R, Roth G, Bander J, Kepros J, Goeschel C (2006) An intervention to decrease catheter-related bloodstream infections in the ICU. N Engl J Med 355:2725–2732
Diaz-Montes TP, Cobb L, Ibeanu OA, Njoku P, Gerardi MA (2012) Introduction of checklists at daily progress notes improves patient care among the gynecological oncology service. J Patient Saf 8:189–193
Haynes AB, Weiser TG, Berry WR, Lipsitz SR, Breizat AH, Dellinger EP, Herbosa T, Joseph S, Kibatala PL, Lapitan MC, Merry AF, Moorthy K, Reznick RK, Taylor B, Gawande AA; Safe Surgery Saves Lives Study Group (2009) A surgical safety checklist to reduce morbidity and mortality in a global population. N Engl J Med 360:491–499
Bosk CL, Dixon-Woods M, Goeschel CA, Pronovost PJ (2009) Reality check for checklists. Lancet 374:444–445
Leape LL (2014) The checklist conundrum. N Engl J Med 370:1063–1064
Urbach DR, Govindarajan A, Saskin R, Wilton AS, Baxter NN (2014) Introduction of surgical safety checklists in Ontario, Canada. N Engl J Med 370:1029–1038
Bosch M, van der Weijden T, Wensing M, Grol R (2007) Tailoring quality improvement interventions to identified barriers: a multiple case analysis. J Eval Clin Pract 13:161–168
Flottorp SA, Oxman AD, Krause J, Musila NR, Wensing M, Godycki-Cwirko M, Baker R, Eccles MP (2013) A checklist for identifying determinants of practice: a systematic review and synthesis of frameworks and taxonomies of factors that prevent or enable improvements in healthcare professional practice. Implement Sci 8:35
Fleuren MA, Paulussen TG, Van Dommelen P, Van Buuren S (2014) Towards a measurement instrument for determinants of innovations. Int J Qual Health Care 26:501–510
Colquhoun H, Grimshaw J, Wensing M (2013) Mapping KT interventions to barriers and facilitators. In: Straus SE, Tetroe J, Graham ID (eds) Knowledge translation in health care: moving from evidence to practice. Wiley Blackwell, BMJ Books, Chichester, pp 137–149
Blok S, Gouttebarge V, Slebus FG, Sluiter JK, Frings-Dresen MH (2011) Facilitators and barriers in the use of a checklist by insurance physicians during work ability assessments in depressive disorder. Saf Health Work 2:328–335
Russ SJ, Sevdalis N, Moorthy K, Mayer EK, Rout S, Caris J, Mansell J, Davies R, Vincent C, Darzi A (2015) A qualitative evaluation of the barriers and facilitators toward implementation of the WHO surgical safety checklist across hospitals in England: lessons from the “Surgical Checklist Implementation Project”. Ann Surg 261:81–91
Nolan B, Zakirova R, Bridge J, Nathens AB (2014) Barriers to implementing the World Health Organization’s Trauma Care Checklist: a Canadian single-center experience. J Trauma Acute Care Surg 77:679–683
Vats A, Vincent CA, Nagpal K, Davies RW, Darzi A, Moorthy K (2010) Practical challenges of introducing WHO surgical checklist: UK pilot experience. BMJ 340:b5433
Fourcade A, Blache JL, Grenier C, Bourgain JL, Minvielle E (2012) Barriers to staff adoption of a surgical safety checklist. BMJ Qual Saf 21:191–197
Gagliardi AR, Straus SE, Shojania KG, Urbach DR (2014) Multiple interacting factors influence adherence, and outcomes associated with surgical safety checklists: a qualitative study. PLoS One 9:e108585
Tapper EB, Lai M (2014) Factors affecting adherence to a quality improvement checklist on an inpatient hepatology service. Proc (Bayl Univ Med Cent) 27:100–102
Shapiro FE, Fernando RJ, Urman RD (2014) Barriers to the implementation of checklists in the office-based procedural setting. J Healthc Risk Manag 33:35–43
Wæhle HV, Haugen AS, Søfteland E, Hjälmhult E (2012) Adjusting team involvement: a grounded theory study of challenges in utilizing a surgical safety checklist as experienced by nurses in the operating room. BMC Nurs 11:16
O’Connor P, Reddin C, O’Sullivan M, O’Duffy F, Keogh I (2013) Surgical checklists: the human factor. Patient Saf Surg 7:14
Grol R, Wensing M, Eccles M, Davis D (eds) (2013) Improving patient care: the implementation of change in health care. John Wiley & Sons, Chichester
Cabana MD, Rand CS, Powe NR, Wu AW, Wilson MH, Abboud PA, Rubin HR (1999) Why don’t physicians follow clinical practice guidelines? A framework for improvement. JAMA 282:1458–1465
Francke AL, Smit MC, de Veer AJ, Mistiaen P (2008) Factors influencing the implementation of clinical guidelines for health care professionals: a systematic meta-review. BMC Med Inform Decis Mak 8:38
Hulscher ME, Grol RP, van der Meer JW (2010) Antibiotic prescribing in hospitals: a social and behavioural scientific approach. Lancet Infect Dis 10:167–175
Mol PG, Rutten WJ, Gans RO, Degener JE, Haaijer-Ruskamp FM (2004) Adherence barriers to antimicrobial treatment guidelines in teaching hospital, the Netherlands. Emerg Infect Dis 10:522–525
Schouten JA, Hulscher ME, Natsch S, Kullberg BJ, van der Meer JW, Grol RP (2007) Barriers to optimal antibiotic use for community-acquired pneumonia at hospitals: a qualitative study. Qual Saf Health Care 16:143–149
Barlow G, Nathwani D, Myers E, Sullivan F, Stevens N, Duffy R, Davey P (2008) Identifying barriers to the rapid administration of appropriate antibiotics in community-acquired pneumonia. J Antimicrob Chemother 61:442–451
van Daalen FV, Prins JM, Opmeer BC, Boermeester MA, Visser CE, van Hest RM, Hulscher MEJL, Geerlings SE (2015) A cluster randomized trial for the implementation of an antibiotic checklist based on validated quality indicators: the AB-checklist. BMC Infect Dis 15:134
Spoorenberg V, Hulscher ME, Akkermans RP, Prins JM, Geerlings SE (2014) Appropriate antibiotic use for patients with urinary tract infections reduces length of hospital stay. Clin Infect Dis 58:164–169
Cunningham CT, Quan H, Hemmelgarn B, Noseworthy T, Beck CA, Dixon E, Samuel S, Ghali WA, Sykes LL, Jetté N (2015) Exploring physician specialist response rates to web-based surveys. BMC Med Res Methodol 15:32
Kellerman SE, Herold J (2001) Physician response to surveys. A review of the literature. Am J Prev Med 20:61–67
Willis GB, Smith T, Lee HJ (2013) Do additional recontacts to increase response rate improve physician survey data quality? Med Care 51:945–948
Acknowledgments
The authors thank the contact physicians of the nine hospitals. They also thank all physicians who participated in the survey.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the Netherlands Organisation for Health Research and Development (ZonMw) grant 836021001.
Conflict of interest
None.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
van Daalen, F.V., Geerlings, S.E., Prins, J.M. et al. A survey to identify barriers of implementing an antibiotic checklist. Eur J Clin Microbiol Infect Dis 35, 545–553 (2016). https://doi.org/10.1007/s10096-015-2569-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-015-2569-5