Abstract
Introduction
Patients with cluster headache (CH), the most common trigeminal autonomic cephalalgia, often face delayed diagnosis, misdiagnosis and mismanagement.
Objectives
To identify, appraise and synthesise clinical studies on the delays in diagnosis and misdiagnosis of CH in order to determine its causes and help the management of this condition.
Methods
The systematic review was prepared, conducted and reported in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis. It was registered with International Prospective Register of Systematic Reviews. A systematic search of different electronic databases (Medline, EMBASE, PsycINFO, PubMed, CINAHL, BNI, HMIC, AMED, HBE and Cochrane Library) was carried out in May 2017. Reference lists of relevant articles were hand searched.
Results
The search identified 201 unique studies. Fifteen studies met the inclusion criteria of which 13 case series studies and two survey studies. Nine studies assessed the delays in diagnosis and misdiagnosis of CH, five studies the delays in diagnosis and one study the misdiagnosis of CH. The studies included 4661 patients. Delays in diagnosis, misdiagnosis and mismanagement have been reported in many European countries, Japan and in the USA with well-developed health services. The patients with CH often visited many different clinicians, surgeons and dentists and received multiple diagnosis prior to being correctly diagnosed.
Conclusion
This systematic review shows that the delays in the diagnosis of CH are a widespread problem, the time to diagnosis still vary from country to country and both patients and physicians are responsible for the delays in diagnosis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Cluster headache (CH) is the most common of the trigeminal autonomic cephalalgias (TACs) and often described as the most severe pain possible [1]. The prevalence of CH is estimated at 0.5–3/1000, with male preponderance [2]. CH is characterised by attacks of unilateral pain associated with ipsilateral conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, forehead and facial sweating, miosis, ptosis and/or eyelid oedema, and/or with restlessness or agitation [3, 4]. The CH attacks that can last between 15 min and 3 h occur from every other day to eight times a day [3]. Cluster headache is maximal orbitally, supraorbitally, temporally or in any combination of these sites, but may spread to other regions [3]. During the worst attacks, the intensity of pain is excruciating. Patients with CH, unlike those with migraine, are unable to lie down and characteristically pace and rock back and forth. The diagnosis of CH is based entirely on clinical history due to the lack of a diagnostic biomarker. Additionally, CH is uncommon and it is even rarer in the paediatric population, therefore underrecognised [5]. For these reasons, patients often face delays in diagnosis and misdiagnosis which inevitably leads to mismanagement. There have been no rigorous systematic literature reviews on this topic. The aim of this systematic literature review is to identify, appraise and synthesise all relevant clinical studies on the misdiagnosis and delays in the diagnosis of CH.
Methods
The systematic review was prepared in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Protocols (PRISMA-P) 2015 guidelines [6] and was conducted and reported according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [7]. It was registered with International Prospective Register of Systematic Reviews (PROSPERO) on 9/11/2017 (registration number CRD42017081204).
Search strategy
A comprehensive search of different electronic databases was carried out in May 2017 to identify potential studies. The following electronic databases were searched: Medline, EMBASE, PsycINFO, PubMed, CINAHL, BNI, HMIC, AMED, HBE (NICE Healthcare Databases) and Cochrane Library. Pre-specified search criteria were designed with input from a professional librarian search specialist; Medical Subject Heading and free text terms were used to increase the search sensitivity.
To search for misdiagnosis, the search terms were misdiagnosis OR diagnostic error OR hidden diagnosis OR unrecognised diagnosis OR alternate diagnosis OR undiagnosed OR diagnostic mistake OR missed diagnosis. The search terms for delays in diagnosis were delays in diagnosis OR late diagnosis OR delayed diagnosis. These were combined with a search for cluster headache OR cluster-like headache. In addition to the electronic search, we screened the reference lists of the included articles and relevant literature known by the authors. The detailed search criteria are shown in Table 1.
Two authors (AB and JB) independently assessed all titles and abstracts for inclusion. The inclusion/exclusion criteria implemented for all searches are shown in Table 2. Full-text papers were retrieved for those meeting the inclusion criteria and for those articles whose eligibility criteria could not be assessed based only on the title and abstract. Two authors (AB and JB) independently assessed all full-text articles and disagreement was resolved by discussion to reach consensus and if needed with the intervention of a third reviewer (FA). The findings are reported according to PRISMA guidelines [7].
Data extraction, assessment and analysis
The data was independently extracted by two authors (AB and JB). Data extracted included the study design, methods of data acquisition, study population (number of participants, men:women ratio, percentage of patients with episodic cluster headache (ECH) and chronic cluster headache (CCH)), time from disease onset to diagnosis (the patient’s delay: the mean time between the CH attack and first consultation of a clinician, clinician’s delay: the mean time between the first consultation of a clinician and correct diagnosis and the mean total delay: sum of patient’s delay and clinician’s delay), percentage of patients misdiagnosed, diagnosis received prior to CH diagnosis, the type and number of clinicians seen prior to diagnosis, treatment received prior to diagnosis and factors involved in the diagnostic delay. The discrepancies were resolved through discussion with a third reviewer (FA).
Risk of bias in individual studies
The risk of bias in individual studies was conducted in order to assess the quality of the studies included in the SLR. Quality assessment was performed using the Joanna Briggs Institute (JBI) Appraisal Checklist for case series studies [8]. Ten domains of the study design and reporting were assessed, each rated ‘Yes’, ‘No’, ‘Unclear’ or ‘Not applicable’. The Oxford Centre for Evidence-Based Medicine (OCEBM) critical appraisal was used for survey studies [9]. Ten domains of the study design and reporting were assessed, each rated ‘Yes’, ‘No’, ‘Unclear’ or ‘Not applicable’. Studies were not excluded based on their quality appraisal. The studies were independently assessed by two reviewers (AB and JB) and the discrepancies were resolved through discussion with a third author (FA).
Data Availability
All data is fully available without restriction.
Results
Studies included
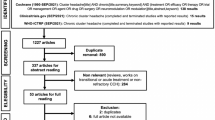
The search carried out in May 2017 on diagnostic delays and misdiagnosis of CH identified 201 unique studies (Fig. 1). The retrieved articles were published between January 1978 and May 2017. All studies were screened by title and abstract and 149 articles were excluded at this stage. Full-text articles were assessed for the remaining 52 studies and 15 studies met our inclusion criteria (Table 2). Thirty-seven articles were excluded after the full-text screening; the reasons for exclusion are shown in the PRISMA flow chart (Fig. 1). The 15 included studies took place in Europe, the USA and Asia. Four studies were from the USA, 3 from Denmark and 1 each from Greece, Serbia, Spain, Norway, Japan, Britain and Flanders. One study was conducted in multiple countries: Italy, Moldova, Ukraine and Bulgaria.
Thirteen case series studies and two survey studies were included. Nine studies assessed the delays in diagnosis and misdiagnosis of CH, five studies the delays in diagnosis and one study the misdiagnosis of CH. The studies included a total of 4661 patients, aged 3–81 years, men and women with ECH and CCH. The percentage of patients with ECH varies from 64 to 100%. The male to female ratio varied from 1.9:1 [10] to 9.6:1 [11]. One included study was in children with CH [12]. The data extracted from case series and survey studies is shown in Table 3 and Table 4. The values in Tables 3 and 4 are extracted from the original (referenced) papers and the percentage values are rounded to the nearest integer. The number of patients with ECH and CCH was converted into percentages where necessary for consistency. The ratio (men:women) was calculated if it was not provided in the cited work.
Non-English articles
Four full-text articles in foreign languages were identified and translated [13,14,15,16]. The articles were excluded as they did not meet the inclusion criteria (the studies were not on delays in diagnosis or misdiagnosis of CH).
Risk of bias in individual studies
The 13 case series assessed using JBI Appraisal Checklist (Table 5) were consecutive case series [11, 12, 17,18,19,20] and non-consecutive case series [21,22,23] which scored ‘YES’ to all JBI domains as well as retrospective case series [10, 24] and one study with unclear inclusion of participants [25]. The two survey studies were assessed using OCEBM critical appraisal of a survey (Table 6). Using this tool, we identified studies that did not assess the statistical significance [26, 27] and did not give the confidence intervals for the main results [27]. We did not exclude studies based on their quality appraisal.
Diagnostic delays
Fourteen of the 15 studies investigated the total delay in diagnosis (i.e. the time from disease onset to correct diagnosis). The studies reported different statistics for time to correct diagnosis (mean, median or percentage). Ten studies assessed the mean time to correct diagnosis [10,11,12, 18,19,20,21, 23, 26, 28], three studies the median time [17, 22, 24] and one study the percentage of patients that experienced delays in diagnosis [29]. The mean time to correct diagnosis recorded in the UK was 2.6 years (between 1990 and 1999) [21], in Flanders 3.6 years [11], in Spain 4.9 years [18], in Italy and East European countries 5.3 ± 6.4 years [28], in Denmark between 6.2 years [23] and 9 years [20], in the USA between 6.6 [26] and 8.5 years [12], in Japan 7.3 ± 6.9 years [19] and in Serbia 7.8 ± 8 years (quoted verbatim form the original paper) [10]. The median time to correct diagnosis was 1 year (range 0–7) in Greece [17], 3 years (range 1–48) in Denmark [22] and 4 years (range 0–30) in Norway [24]. In one study performed in the USA, 42% of patients waited more than 5 years to receive a correct diagnosis of cluster headache [29].
Two studies showed a reduction in delay in the diagnosis of CH over time, from 22.3 years (before 1959) to 2.6 years (between 1990 and 1999) in the UK [21] and from 20 years (prior to 1989) to 1 year (between 2010 and 2015) in Greece [17]. Two studies looked at patient’s and clinician’s delays in the diagnosis of CH [11, 28]. Van Alboom et al. showed that the mean time between the first cluster headache attack and the first consultation was 11 months [11] and Voiticovski-Iosob et al. found patient’s delay in almost one quarter of cases [28].
While Bahra and Goadsby found no significant difference in time to diagnosis between men and women [21], Lund et al. showed that men waited a mean time of 6.56 years and women waited 5.5 years [23]. Gender difference was also recorded by Vikelis and Rapoport where a median of 0 years (range 0–6) was found for men and 3 years (range 0–7) for women [17]. One study assessed the influence of age of onset on the diagnostic delay [10]. Zidverc-Trajkovic et al. showed that the condition is less recognised in patients with early onset of CH (less than 20 years of age) [10]. People with late onset of CH (> 40 years of age) were more rapidly diagnosed than subjects with typical age of onset of CH (20–40 years of age) [10]. In the study conducted by Van Vliet et al., the patients with ECH had longer delays in diagnosis compared to CCH patients [22], probably due to longer remission periods.
Misdiagnoses prior to correct CH diagnosis
Migraine, trigeminal neuralgia, sinusitis and dental/jaw disease are the most common misdiagnoses. Other diagnoses received by the CH patients were tension-type headache; ophthalmic disease; ear, nose and throat (ENT) disease; cervical spine disease; idiopathic intracranial hypertension; allergies; short lasting neuralgiform headache with conjunctival injection and tearing (SUNCT) and psychiatric disorders. Migraine was the most received misdiagnosis [11, 17, 18] followed by trigeminal neuralgia, [17, 18, 28]. Sinusitis was often diagnosed in patients with CH, most likely due to presence of rhinorrhoea, nasal congestion and seasonal variation, although there was no significant statistical correlation between these features and the diagnosis of CH [11]. The mean number of diagnosis received per patient was 2.2 in Italy and Eastern Europe [28] and 3.9 in the USA [26]. In Flanders, 65% of the patients studied were misdiagnosed [11] and in Italy and East Europe 77% were misdiagnosed [28]. In Denmark, more women (61%) were misdiagnosed as migraine compared to men (45.5%) [23].
Clinicians seen prior to correct CH diagnosis
Patients with CH were often seen by different clinicians before the correct diagnosis was made. Vikelis and Rapoport showed that nearly two thirds of their Greek patients (63.5%) consulted a general practitioner or internist, around one third an ENT specialist, ophthalmologist or dentist, and a small proportion (8.5%) a neurosurgeon [17]. In the same study, 40% of the patients were seen by neurologists who missed the diagnosis [17]. In Flanders, neurologists correctly diagnosed 80% of cases [11]. Patients often sought help from alternative medicine specialists (acupuncturists and chiropractors) [11, 24, 25, 28]. Even children consulted many different specialists prior to diagnosis (internists, general practitioners, otolaryngologists, opthalmologists, psychiatrists, chiropractors, orthopaedic surgeons and allergists) [12]. Self-diagnosis using different sources of information (internet, reading about CH and discussion with other people suffering with CH) with subsequent medical confirmation was the second most common way of diagnosis after clinician’s diagnosis [17] and it was reported in 4%, 13% and 15% of patients in Flanders [11], the UK [21] and Italy and East European countries respectively [28]. Patients consulted between 2 and 5 clinicians before the correct diagnosis was made [11, 17, 18, 28] frequently including a dentist, ENT specialists or ophthalmologist who exceptionally made the diagnosis [11]. Vikelis and Rapoport found that patients with CCH consulted more clinicians than patients with ECH (median 4 vs 2) [17] and no differences in the number of clinicians consulted by men and women were found [17]. Most patients with CH have never been seen by specialists in emergency medicine [29]. The most obvious explanation would be the short duration of the attacks.
Mismanagement prior to correct CH diagnosis
General neurologists frequently offered non-evidence-based CH treatments [12, 17, 28]. Dentists and ENT specialists performed tooth extractions, fillings, sinus washout and surgery for nasal septum deviation without any success. Dentists, ENT specialists or other clinicians that did not recognise the disorder often recommend unnecessary investigations (MRI head, CT head, EEC, cervical spine X-ray, skull X-ray) to diagnose a secondary headache [28]. Patients underwent alternative medicine treatments such as acupuncture [11, 24, 25, 27], homoeotherapy [28], chirotherapy [24, 25, 28], relaxation techniques [28], cold therapy [28], reflexology [11], hypnosis [11], osteopathy [11], spiritual healing [11] and illicit drug use [24, 28]. Even after correct diagnosis of CH, the patients complained of lack of information regarding the cause of the disorder and available treatments [18]. Some patients received incorrect information as to the cause of CH (psychiatric, vascular disorder, genetic/familial, brain injury, alcohol, tobacco) and others no information [18].
Factors involved in the diagnostic delay and misdiagnosis
Three studies assessed the factors involved in the diagnostic delay [11, 17, 22]. Van Vliet et al. showed that the presence of ECH, nausea, vomiting during attacks, photophobia or phonophobia, nocturnal onset of attacks, restlessness, pain radiating to the jaw, alternating attack side and circadian rhythm delayed the diagnosis of CH [22]. The male gender and interictal headache did not influence the correct diagnosis of CH [22]. However, Vikelis and Rapoport showed that the side shift between bouts, jaw location of pain, the cheek location of pain, lower teeth location of pain, ear location of pain, aggravation by physical activity, the presence of forehead and facial sweating, the presence of photophobia and the absence of cranial autonomic features delayed the correct diagnosis of CH [17]. The authors have also shown that the decade of onset of CH influenced the correct diagnosis [17]. Patients with onset before the year 2000 waited a median of 13 years (range 0–45) to be diagnosed compared to patients with onset after the year 2010 who waited a median of 1 year (range 1–7) [17]. A lower age of onset and pain that does not reach the maximum intensity within the first 5 min were also features that contributed to diagnostic delay [11].
Discussion
It is evident from the studies that diagnostic delay in CH is not confined to a geographical area. Although some countries had less delay than others, delays in diagnosis were recorded in multiple countries in Europe, the USA and Japan. One possible reason could be limited knowledge about the characteristics of CH across countries. However, these results should be interpreted with caution as each study does not reflect the whole CH population of a country. Only one nationwide survey study performed in the USA that included a sample of 1134 patients was retrieved by our searches and could be considered representative for a large cohort of patients with CH [29]. The studies were performed over a period of 25 years and are not directly comparable as the International Classification of Headache Disorders has suffered amendments over the years.
The studies included in this review showed that patient’s delay in diagnosis is as important as clinician’s delay [11, 28]. The reason why patients with CH do not seek timely medical advice is not well understood. The short duration of the attacks could be an explanation although there are currently no studies that assessed this.
It has been shown that the episodic pattern of attacks, a specific feature of CH, does not seem to contribute to an earlier diagnosis [22]. Moreover, extended periods of remissions only prolong the diagnostic delay. Improved awareness of the condition is the most probable reason for the reduction of time to correct diagnosis in the UK, Greece and Denmark [17, 20, 21, 23]. It is unclear why patients with late onset CH were more rapidly diagnosed than those with early onset [10]. It is possible that clinicians erroneously view CH as a disorder with onset predominantly in late adulthood. Another explanation might be that clinicians are more suspicious of a sinister cause for the symptoms if the patient is older, and therefore have a lower threshold to refer to a neurologist although there are no studies that have assessed this.
A lack of knowledge of the characteristics of CH is likely to influence the clinician to seek an alternative diagnosis. Some CH characteristics could lead the clinician astray. For example, migraine features (e.g. aura, photophobia, phonophobia, nausea, vomiting) and a family history of migraine are often encountered in patients with CH [22]. The features of the pain in CH may also mislead the clinician in making the wrong diagnosis. Although CH affects the first division of the trigeminal nerve while trigeminal neuralgia the second or third and exceptionally the first division, trigeminal neuralgia was the second most received misdiagnosis in two studies [17, 18]. The presence of stereotyped attacks associated with cranial autonomic symptoms, the absence of triggers and the totally different duration and pain quality still qualify trigeminal neuralgia as one of the most received misdiagnosis [17, 18, 28]. It is possible that clinicians are more aware of trigeminal neuralgia, even though CH is more common (incidence 53/100.000 [30] vs 4.5/100.00 [31]) but there are no studies that validated this. The presence of side shift between attacks was also correlated with diagnostic delay possibly because CH is defined as ‘unilateral pain’ as per ICHD-3 criteria [3].
Misdiagnosis invariably leads to mismanagement. In CH, due to the severity of the symptoms, patients desperately seek the opinion of several specialists until the symptoms are alleviated. It is possible that some specialists feel the need to offer invasive procedures in an attempt to provide some form of relief, even if the chance of success is small. A high proportion of patients with CH undergo invasive procedures from dental surgeons and ENT specialists when a clear indication for such interventions was lacking. These results suggest that further awareness is required, particularly in the dental and ENT professions regarding the pain and cranial autonomic symptoms of CH mimicking dental and sinus pathologies, to avoid unnecessary and potentially harmful procedures.
In an attempt to treat their symptoms, patients with CH are more likely to employ extreme measures. The use of illicit drugs among CH sufferers is common [24, 28]. They are also more inclined to have recourse to non-evidence-based and non-pharmocological treatments [11, 24]. This further supports the need for timely diagnosis and initiation of evidence-based treatments, and patient education. The evidence suggests that even after the correct diagnosis is reached, some patients received poor or incorrect information about the nature of their disability [18]. Suboptimal management is not limited to the cluster headache sufferers since most headache patients are undertreated, hence the importance of headache centres and promoting education of GPs [32].
Strengths
This is the first rigorously conducted systematic review on delays in diagnosis and misdiagnosis of cluster headache. A detailed search strategy of 10 electronic databases was used with no date or language restrictions. We included larger studies that could demonstrate rigorous analysis and we have excluded studies with less than 10 patients and case reports.
Limitations
It is possible that relevant studies were missed despite a comprehensive search strategy across multiple databases with no date or language restrictions. Due to the paucity of studies in this area, we did not exclude studies on the basis of quality appraisal.
Future work
As CH is a life-long severe and debilitating condition that requires prompt diagnosis and management, it is essential to establish what factors are involved in the diagnostic delay and misdiagnosis. Educational activities for general practitioners, ENT specialists, ophthalmologists and other medical specialities and even for neurologists are important to raise awareness of CH, its diagnosis and management. Getting medical and emotional support are important priorities for CH sufferers. Clinicians of all specialities should be aware of the existence of CH and long-term support should be in place so that patients with CH can live a normal life. Future work regarding biomarkers could help in the misdiagnosis and delays in the diagnosis of CH.
Conclusions
Delays in diagnosis, misdiagnosis and mismanagement of CH are a widespread problem and have been reported in many countries with well-developed health services, including several European countries, Japan and in the USA. Both patient and clinician factors account for the delays in diagnosis. Patients with CH often waited before seeking medical advice and when they did, they visited many clinicians and received multiple misdiagnosis prior to being correctly diagnosed. The failure to diagnose patients with CH leads to poor management, disability and misuse of healthcare resources. If a clinician has a suspicion of CH, this should trigger referral to specialised headaches centres for a correct diagnosis and initiation of appropriate treatment and to minimise the wastage of healthcare resources and unnecessary procedures.
Abbreviations
- CH:
-
cluster headache
- PRISMA:
-
Preferred Reporting Items for Systematic Review and Meta-Analysis
- PROSPERO:
-
International Prospective Register of Systematic Reviews
- TACs:
-
trigeminal autonomic cephalalgias
- ECH:
-
episodic cluster headache
- CCH:
-
chronic cluster headache
- OCEBM:
-
Oxford Centre for Evidence-Based Medicine
- JBI:
-
Joanna Briggs Institute
- ENT:
-
ear, nose and throat
- SUNCT:
-
short lasting neuralgiform headache with conjunctival injection and tearing
References
Bahra A, May A, Goadsby PJ (2002) Cluster headache: a prospective clinical study with diagnostic implications. Neurology 58(3):354–356. https://doi.org/10.1212/WLN.58.3.354
D’Arrigo G et al (2018) High dosage of methylprednisolone in cluster headache. Neurol Sci 39(1):157–158. https://doi.org/10.1007/s10072-018-3383-5
ICHD-3 (2018) Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia 38(1):1–211. https://doi.org/10.1177/0333102417738202
Bussone G (2014) Strictly unilateral headaches: considerations of a clinician. Neurol Sci 35(1):71–75. https://doi.org/10.1186/1750-1172-3-20
Lambru G, Byrne S (2018) Trigeminal autonomic cephalalgias in children and adolescents. Neurol Sci 239(1):105–106. https://doi.org/10.1007/s10072-018-3359-5
Shamseer L, Moher D, Clarke M, Ghersi D, Liberati A, Petticrew M, Shekelle P, Stewart LA, the PRISMA-P Group (2015) Preferred Reporting Items for Systematic Review and Metanalysis Protocols (PRISMA-P): elaboration and explanation. BMJ 350:g7647. https://doi.org/10.1136/bmj.g7647
Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009) Preferred Reporting Items for Systematic Reviews and Meta-Analyses: the PRISMA Statement. PLoS Med 6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097
Moola S, Munn Z, Sears K, Sfetcu R, Currie M, Lisy K, Tufanaru C, Qureshi R, Mattis P, Mu P (2015) Conducting systematic reviews of association (etiology): the Joanna Briggs Institute’s approach. Int J Evid Based Healthc 13(3):163–169. https://doi.org/10.1097/XEB.0000000000000064
CEBM - Centre for Evidence Based Medicine (2014) Critical Appraisal Tools. https://www.cebm.net/2014/06/critical-appraisal. Accessed 5 May 2017
Zidverc-Trajkovic J, Markovic K, Radojicic A, Podgorac A, Sternic N (2014) Cluster headache: is age of onset important for clinical presentation? Cephalalgia 34(9):664–670. https://doi.org/10.1177/0333102413520085
Van Alboom E et al (2009) Diagnostic and therapeutic trajectory of cluster headache patients in Flanders. Acta Neurol Belg 109(1):10–17
Maytal J, Lipton RB, Solomon S, Shinnar S (1992) Childhood onset cluster headaches. Headache 32(6):275–279
Holle D, Nagel S, Obermann M (2012) Therapie trigemino-autonomer kopfschmerzen treatment of trigeminal autonomic cephalgias. Arzneimitteltherapie 30(7):221–226
Lanteri-Minet M (2015) Epidemiology, clinical presentation, diagnosis, natural history and screening of cluster headache. Presse Med 44(11):1176–1179. https://doi.org/10.1177/0333102417738202
Kim H (2005) The characteristics of sinus headache resembling the primary headaches. Japanese Journal of Clinical Medicine 63(10):1771–1776
Yang Y, Huang W (2011) Clinical manifestations of cluster headache accompanied by chronic nasosinusitis. Nihon Rinsho 25(21):981–983
Vikelis M, Rapoport AM (2016) Cluster headache in Greece: an observational clinical and demographic study of 302 patients. J Headache Pain 17(1):88. https://doi.org/10.1186/s10194-016-0683-0
Sanchez Del Rio M et al (2014) Errors in recognition and management are still frequent in patients with cluster headache. Eur Neurol 72(3–4):209–212. https://doi.org/10.1159/000362517
Imai N, Yagi N, Kuroda R, Konishi T, Serizawa M, Kobari M (2011) Clinical profile of cluster headaches in Japan: low prevalence of chronic cluster headache, and uncoupling of sense and behaviour of restlessness. Cephalalgia 31(5):628–633. https://doi.org/10.1177/0333102410391486
Jensen RM, Lyngberg A, Jensen RH (2007) Burden of cluster headache. Cephalalgia 27(6):535–541. https://doi.org/10.1111/j.1468-2982.2007.01330.x
Bahra A, Goadsby PJ (2004) Diagnostic delays and mis-management in cluster headache. Acta Neurol Scand 109(3):175–179. https://doi.org/10.1046/j.1600-0404.2003.00237.x
Van Vliet JA et al (2003) Features involved in the diagnostic delay of cluster headache. J Neurol Neurosurg Psychiatry 74(8):1123–1125
Lund N, Barloese M, Petersen A, Haddock B, Jensen R (2017) Chronobiology differs between men and women with cluster headache, clinical phenotype does not. Neurology 88(11):1069–1076
Bekkelund SI, Ofte HK, Alstadhaug KB (2014) Patient satisfaction with conventional, complementary, and alternative treatment for cluster headache in a Norwegian cohort. Scand J Prim Health Care 32(3):111–116
Bittar G, Graff-Radford SB (1992) A retrospective study of patients with cluster headaches. Oral Surg Oral Med Oral Pathol 73(5):519–525
Klapper JA, Klapper A, Voss T (2000) The misdiagnosis of cluster headache: nonclinic, population-based, Internet survey. Headache 40(9):730–735
Rozen TD (2010) Cluster headache as the result of secondhand cigarette smoke exposure during childhood. Headache 50(1):130–132. https://doi.org/10.1111/j.1526-4610.2009.01542.x
Voiticovschi-Iosob C, Allena M, de Cillis I, Nappi G, Sjaastad O, Antonaci F (2014) Diagnostic and therapeutic errors in cluster headache: a hospital-based study. J Headache Pain 15:56. https://doi.org/10.1186/1129-2377-15-56
Rozen TD, Fishman RS (2012) Cluster headache in the United States of America: demographics, clinical characteristics, triggers, suicidality, and personal burden. Headache 52(1):99–113. https://doi.org/10.1111/j.1526-4610.2011.02028.x
Fischera M, Marziniak M, Gralow I, Evers S (2008) The incidence and prevalence of cluster headache: a meta-analysis of population-based studies. Cephalalgia 28(6):614–618. https://doi.org/10.1111/j.1468-2982.2008.01592.x
Bangash TH (2011) Trigeminal neuralgia: frequency of occurrence in different nerve branches. Anesth Pain Med 1(2):70–72. https://doi.org/10.5812/kowsar.22287523.2164
D’Amico D et al (2011) Undertreatment in patients with primary headaches attending headache centres. Neurol Sci 32(1):181–183. https://doi.org/10.1007/s10072-011-0528-1
Acknowledgements
The authors would like to acknowledge the library personnel from Hull York Medical School for their help in setting up the search criteria for the systematic literature review.
Author information
Authors and Affiliations
Contributions
All authors designed the review. AB and JB identified and screened the articles and summarised and analysed the data. The discrepancies were resolved through discussion with FA. AB wrote the initial version of manuscript. All authors read, edited and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
All authors have read and approved the manuscript for publication.
Competing interests
The authors declare that they have no competing interests.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Buture, A., Ahmed, F., Dikomitis, L. et al. Systematic literature review on the delays in the diagnosis and misdiagnosis of cluster headache. Neurol Sci 40, 25–39 (2019). https://doi.org/10.1007/s10072-018-3598-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-018-3598-5