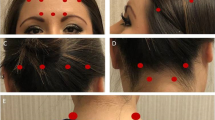
Abstract
The use of Onabotulinumtoxin A as treatment for different neurological conditions is more common in the last decades; its application has been consolidated on the basis of significant clinical results. The clinical experiences with Onabotulinumtoxin A for chronic migraine are on the increase in Italy: at the moment, clinical results are encouraging and enforce the application of the toxin for chronic migraine, according to the results of the PREEMPT studies. The possibility for the patients to be treated with a second cycle of therapy after the first year of treatment is under discussion, in particular for patients who obtained significant clinical benefit from the first period of treatment. In this report, a group of patients treated with Onabotulinumtoxin A for 1 year, according to the PREEMPT protocol, has been retreated for one more year in order to confirm the clinical benefit obtained after the first year of treatment.
Similar content being viewed by others
References
Bigal ME, Serrano D, Reed M, Lipton RB (2008) Chronic migraine in the population: burden, diagnosis, and satisfaction with treatment. Neurology 71:559–566
Manack AN, Buse DC, Lipton RB (2011) Chronic migraine: epidemiology and disease burden. Curr Pain Headache Rep 15:70–78
International Headache Society (2013) International classification of headache disorders, 3rd edition (beta version). Cephalalgia 33:628–808
Grazzi L, Andrasik F, Usai S, Bussone G (2009) Treatment of chronic migraine with medication overuse: is drug withdrawal crucial? Neurol Sci 30(Suppl 1):85–88
Ranoux D, Attal N, Morain F, Bouhasira D (2008) Botulinum toxin type A induces direct analgesic effects in chronic neuropathic pain. Ann Neurol 64:274–283
Simpson DM, Hallett M, Ashman EJ, Comella CI, Green MW, Gronseth GS, Armostrong MJ, Gloss D, Potrebic S, Jankovic J, Karp BR, Naumann M, So YT, Yablon SA (2016) Practice guideline update summary: botulinum neurotoxin for the treatment of blepharospasm, cervical dystonia, adult spasticity, and headache. Neurology 86:1818–1826
Colosimo C, Tiple D, Berardelli A (2012) Efficacy and safety of long-term botulinum toxin treatment in craniocervical dystonia: a systematic review. Neurotox Res 22:265–273
Aurora SK, Dodick DW, Turkel CC, DeGryse RE, Silberstein SD, Lipton RB, Diener HC, Brin MF, PREEMPT 1 Chronic Migraine Study Group (2010) Onabotulinumtoxin A for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 1 trial. Cephalalgia 30:793–803
Diener HC, Dodick DW, Aurora SK, Turkel CC, DeGryse RE, Lipton RB, Silberstein SD, Brin MF, PREEMPT 2 Chronic Migraine Study Group (2010) Onabotulinumtoxin A for treatment of chronic migraine: results from the double-blind, randomized, placebo-controlled phase of the PREEMPT 2 trial. Cephalalgia 30:804–814
Aurora SK, Winner P, Freeman MC, Spierings EL, Heiring JO, DeGryse RE, VanDenburgh AM, Nolan ME, Turkel CC (2011) Onabotulinumtoxin A for treatment of chronic migraine: pooled analyses of the 56-week PRE-EMPT clinical program. Headache 51:1358–1373
Aoki KR (2003) Evidence for antinociceptive activity of botulinum toxin type A in pain management. Headache 43(Suppl):S9–S15
Aoki KR (2005) Review of a proposed mechanisms for the antinociceptive action of botulinum toxin type A. Neurotoxicology 26:785–793
Gazerani P, Pedersen NS, Staahl C, Drewes AM, Arendt-Nielsen L (2009) Subcutaneous botulinum toxin type A reduces capsaicin-induced trigeminal pain and vasomotor reactions in human skin. Pain 141:60–69
Sandrini G, Perrotta A, Tassorelli C, Torelli P, Brighina F, Sances G, Nappi G (2011) Botulinum toxin type-A in the prophylaxis treatment of medication-overuse headache: a multi center, double blind, randomized, placebo-controlled, parallel group study. J Headache Pain 12:427–433
Grazzi L, Usai S (2015) Onabotulinumtoxin A (Botox) for chronic migraine treatment: an Italian experience. Neurol Sci 36(Suppl1):S33–S35
Farinelli I, Coloprisco G, De Filippis S, Martelletti P (2006) Long-term benefits of Botulinum Toxin A (BOTOX) in chronic daily headache: a five-year long experience. J Headache Pain 7:407–412
Cernuda-Morollon E, Ramon C, Larrosa D, Alvarez R, Riesco N, Pascual J (2015) Long-term experience with Onabotulinumtoxin A in the treatment of chronic migraine: what happens after one year? Cephalalgia 35:864–868
Kollewe K, Escher CM, Wulff DU, Fathi D, Paracka L, Mohammadi B, Karst M, Dressler D (2016) Long-term treatment of chronic migraine with Onabotulinumtoxin A: efficacy, quality of life and tolerability in a real-life setting. J Neural Transm 123:533–540
Guerzoni S, Pellesi L, Beraldi C, Pini LA (2016) Increased efficacy of regularly repeated cycles with Onabotulinumtoxin A in MOH patients beyond the first year of treatment. J Headache Pain 17:48
Negro A, Curto M, Lionetto L, Martelletti P (2016) A two years open-label prospective study of Onabotulinumtoxin A 195U in medication overuse headache: a real-world experience. J Headache Pain 17:1–9
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We certify that there is no actual or potential conflict of interest in relation to this article.
Rights and permissions
About this article
Cite this article
Grazzi, L. Onabotulinumtoxin A for chronic migraine with medication overuse: clinical results of a long-term treatment. Neurol Sci 38 (Suppl 1), 141–143 (2017). https://doi.org/10.1007/s10072-017-2864-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-017-2864-2