Abstract
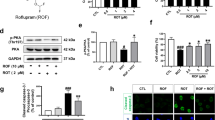
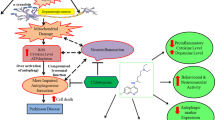
Rifampicin is a macrocyclic antibiotic used extensively for the treatment of Mycobacterium tuberculosis and other mycobacterial infections. Recently, it was discovered that rifampicin exhibits neuroprotective effects. It has been shown to protect PC12 cells against MPP+-induced apoptosis and inhibit the expression of α-synuclein multimers. In in vitro studies, rifampicin pretreatment protects PC12 cells against rotenone-induced cell death. Qualitative and quantitative analyses uncover that rifampicin significantly suppresses rotenone-induced apoptosis by ameliorating mitochondrial oxidative stress. It reduces microglial inflammation and improves neuron survival. Our results indicate that rifampicin is cytoprotective under a variety of experimental conditions, and suggest that it may be useful in PD therapeutics. It is the aim of this paper to review the experimental neuroprotection data reported using rifampicin with a focus on the molecular and cellular mechanisms of cytoprotective effect in in vitro models of PD.


Similar content being viewed by others
References
Langston JW (2002) Parkinson’s disease: current and future challenges. Neurotoxicology 23(4–5):443–450
Heisters D (2011) Parkinson’s: symptoms, treatments and research. Br J Nurs 20(9):548–554
Eriksen JL, Wszolek Z, Petrucelli L (2005) Molecular pathogenesis of Parkinson disease. Arch Neurol 62(3):353–357
Babi T, Mahovi D (2008) Parkinson’s disease-challenges in new drug development. Coll Antropol 32(4):1275–1281
Wirdefeldt K, Adami HO, Cole P, Trichopoulos D, Mandel J (2011) Epidemiology and etiology of Parkinson’s disease: a review of the evidence. Eur J Epidemiol 26(Suppl 1):S1–S58
Olanow CW (2007) The pathogenesis of cell death in Parkinson’s disease-2007. Mov Disord 22(Suppl 17):S335–S342
Meissner WG, Frasier M, Gasser T, Goetz CG, Lozano A, Piccini P, Obeso JA, Rascol O, Schapira A, Voon V, Weiner DM, Tison F, Bezard E (2011) Priorities in Parkinson’s disease research. Nat Rev Drug Discov 10(5):377–393
Yulug B, Kilic U, Kilic E, Bähr M (2004) Rifampicin attenuates brain damage in focal ischemia. Brain Res 996(1):76–80
Namba Y, Kawatsu K, Izumi S, Ueki A, Ikeda K (1992) Neurofibrillary tangles and senile plaques in brain of elderly leprosy patients. Lancet 340(8825):978
Chui DH, Tabira T, Izumi S, Koya G, Ogata J (1994) Decreased beta-amyloid and increased abnormal tau deposition in the brain of aged patients with leprosy. Am J Pathol 145(4):771–775
Tomiyama T, Kaneko H, Kataoka K, Asano S, Endo N (1997) Rifampicin inhibits the toxicity of pre-aggregated amyloid peptides by binding to peptide fibrils and preventing amyloid-cell interaction. Biochem J 322(Pt3):859–865
Tomiyama T, Shoji A, Kataoka K, Suwa Y, Asano S, Kaneko H, Endo N (1996) Inhibition of amyloid beta protein aggregation and neurotoxicity by rifampicin. Its possible function as a hydroxyl radical scavenger. J Biol Chem 271(12):6839–6844
Tomiyama T, Asano S, Suwa Y, Morita T, Kataoka K, Mori H, Endo N (1994) Rifampicin prevents the aggregation and neurotoxicity of amyloid beta protein in vitro. Biochem Biophys Res Commun 204(1):76–83
Kapurniotu A (2004) Targeting alpha-synuclein in Parkinson’s disease. Chem Biol 11(11):1476–1478
Bradbury J (2005) New hope for mechanism-based treatment of Parkinson’s disease. Drug Discov Today 10(2):80–81
Kilic U, Kilic E, Lingor P, Yulug B, Bähr M (2004) Rifampicin inhibits neurodegeneration in the optic nerve transection model in vivo and after 1-methyl-4-phenylpyridinium intoxication in vitro. Acta Neuropathol 108(1):65–68
Oida Y, Kitaichi K, Nakayama H, Ito Y, Fujimoto Y, Shimazawa M, Nagai H, Hara H (2006) Rifampicin attenuates the MPTP-induced neurotoxicity in mouse brain. Brain Res. 1082(1):196–204
Forno LS (1996) Neuropathology of Parkinson’s disease. J Neuropathol Exp Neurol 55:259–272
Martin FL, Williamson SJ, Paleologou KE, Allsop D, El-Agnaf OM (2004) Alpha-synuclein and the pathogenesis of Parkinson’s disease. Protein Pept Lett 11:229–237
Bennett MC (2005) The role of alpha-synuclein in neurodegenerative diseases. Pharmacol Ther 105:311–331
Uversky VN, Eliezer D (2009) Biophysics of Parkinson’s disease: structure and aggregation of alpha-synuclein. Curr Protein Pept Sci 10(5):483–499
Xu J, Wei C, Xu C, Bennett MC, Zhang G, Li F, Tao E (2007) Rifampicin protects PC12 cells against MPP+-induced apoptosis and inhibits the expression of an α-synuclein multimer. Brain Res 1139:220–225
Lee SJ (2003) alpha-Synuclein aggregation: a link between mitochondrial defects and Parkinson’s disease? Antioxid Redox Signal 3:337–348
Ono K, Hirohata M, Yamada M (2008) Alpha-synuclein assembly as a therapeutic target of Parkinson’s disease and related disorders. Curr Pharm Des 14(30):3247–3266
Li J, Zhu M, Rajamani S, Uversky VN, Fink AL (2004) Rifampicin inhibits alpha-synuclein fibrillation and disaggregates fibrils. Chem Biol 11:1513–1521
Greenamyre JT, Betarbet R, Sherer TB (2003) The rotenone model of Parkinson’s disease: genes, environment and mitochondria. Parkinsonism Relat Disord 9(Suppl 2):S59–S64
Testa CM, Sherer TB, Greenamyre JT (2005) Rotenone induces oxidative stress and dopaminergic neuron damage in organotypic substantia nigra cultures. Brain Res Mol Brain Res 134(1):220–225
Watanabe Y, Himeda T, Araki T (2005) Mechanisms of MPTP toxicity and their implications for therapy of Parkinson’s disease. Med Sci Monit 11:RA17–RA23
Sherer TB, Betarbet R, Stout AK, Lund S, Baptista M, Panov AV, Cookson MR, Greenamyre JT (2002) An in vitro model of Parkinson’s disease: linking mitochondrial impairment to altered alpha-synuclein metabolism and oxidative damage. J Neurosci 22:7006–7015
Starkov AA (2008) The role of mitochondria in reactive oxygen species metabolism and signaling. Ann N Y Acad Sci 1147:37–52
Tretter L, Sipos I, Adam-Vizi V (2004) Initiation of neuronal damage by complex I deficiency and oxidative stress in Parkinson’s disease. Neurochem Res 29(3):569–577
Zhu J, Chu CT (2010) Mitochondrial dysfunction in Parkinson’s disease. J Alzheimers Dis 20(Suppl 2):S325–S334
Tobón-Velasco JC, Carmona-Aparicio L, Ali SF, Santamaría A (2010) Biomarkers of cell damage induced by oxidative stress in Parkinson’s disease and related models. Cent Nerv Syst Agents Med Chem 10(4):278–286
Fariss MW, Chan CB, Patel M, Van Houten B, Orrenius S (2005) Role of mitochondria in toxic oxidative stress. Mol Interv 5(2):94–111
Beal MF (1992) Does impairment of energy metabolism result in excitotoxic neuronal death in neurodegenerative illnesses? Ann Neurol 31(2):119–130
Adams JD Jr, Chang ML, Klaidman L (2001) Parkinson’s disease—redox mechanisms. Curr Med Chem 8(7):809–814
Böttcher T, Gerber J, Wellmer A, Smirnov AV, Fakhrjanali F, Mix E, Pilz J, Zettl UK, Nau R (2000) Rifampin reduces production of reactive oxygen species of cerebrospinal fluid phagocytes and hippocampal neuronal apoptosis in experimental Streptococcus pneumoniae meningitis. J Infect Dis 181(6):2095–2098
Chen S, Sun Y, Zeng Z, Tao E (2010) Rifampicin inhibits apoptosis in rotenone-induced differentiated PC12 cells by ameliorating mitochondrial oxidative stress. Neural Regen Res 5(4):251–256
Qian L, Flood PM, Hong JS (2010) Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J Neural Transm 117(8):971–979
Lu L, Li F, Wang X (2010) Novel anti-inflammatory and neuroprotective agents for Parkinson’s disease. CNS Neurol Disord Drug Targets 9(2):232–240
Meda L, Cassatella MA, Szendrei GI, Otvos L Jr, Baron P, Villalba M (1995) Activation of microglial cells by β-amyloid protein and interferon-γ. Nature 373:647–650
Zielasek J, Hartung HP (1996) Molecular mechanisms of microglial activation. Adv Neuroimmunol 6:191–202
Bi W, Zhu L, Wang C, Liang Y, Liu J, Shi Q, Tao E (2011) Rifampicin inhibits microglial inflammation and improves neuron survival against inflammation. Brain Res 1395:12–20
Bellahsène A, Forsgren A (1980) Effect of rifampin on the immune response in mice. Infect Immun 27:15–20
Tsiskarishvili NV, Tsiskarishvili NI (2009) The anti-tubercular drugs in the treatment of psoriasis. Georgian Med News 174:25–28
Hamid Namazi MD (2008) Practice pearl: a novel use of rifampicin for treatment of carpal tunnel syndrome. J Pain 9:380–381
Calleja C, Pascussi JM, Mani JC, Maurel P, Vilarem MJ (1998) The antibiotic rifampicin is a nonsteroidal ligand and activator of the human glucocorticoid receptor. Nat Med 4:92–96
Kim SK, Kim YM, Yeum CE, Jin SH, Chae GT, Lee SB (2009) Rifampicin Inhibits the LPS-induced Expression of Toll-like Receptor 2 via the Suppression of NF-kappaB DNA-binding Activity in RAW 264.7 Cells. Korean J Physiol Pharmacol 13:475–482
Dutta G, Zhang P, Liu B (2008) The lipopolysaccharide Parkinson’s disease animal model: mechanistic studies and drug discovery. Fundam Clin Pharmacol 22(5):453–464
Liu B (2006) Modulation of microglial pro-inflammatory and neurotoxic activity for the treatment of Parkinson’s disease. AAPS J 8(3):E606–E621
Qian L, Flood PM, Hong JS (2010) Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J Neural Transm 117:971–979
Acknowledgments
This work was funded by the Guangdong Provincial Department of Science and Technology (0400935505B33801003) and the Ph.D. Programs Foundation of Ministry of Education of China (No. 20070558257).
Conflict of interest
This paper is free of conflicts of interest. We declare that we have no financial or personal relationships with other people or organizations that could inappropriately bias our work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bi, W., Zhu, L., Jing, X. et al. Rifampicin and Parkinson’s disease. Neurol Sci 34, 137–141 (2013). https://doi.org/10.1007/s10072-012-1156-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-012-1156-0