Abstract
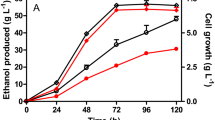
The acetic acid bacterium Acetobacter pasteurianus is used for vinegar fermentation with ethanol as a substrate. However, growth of A. pasteurianus is inhibited by high ethanol concentrations. The ethanol resistance of A. pasteurianus CGMCC 3089 was improved using a continuous ethanol stress adaptation culture. Characterization studies of strains during evolutionary processes were performed for improved ethanol resistance by comparison of cell growth and alcohol dehydrogenase (ADH) and aldehyde dehydrogenases (ALDH) activities. Improved resistance against ethanol was an inheritable phenotype instead of a transient physiologic adaptation. The evolutionary response of ADH and ALDH to high concentrations of ethanol was responsible for the ethanol resistance of A. pasteurianus, instead of mutations in the open reading frames of ADH and ALDH, or long nucleotide sequence insertion or deletion in the genome.
Similar content being viewed by others
References
Matsutani M, Nishikura M, Saichana N, Hatano T, Masud-Tippayasak U, Theergool G, Yakushia T, Matsushita K. Adaptive mutation of Acetobacter pasteurianus SKU1108 enhances acetic acid fermentation ability at high temperature. J. Biotechnol. 165: 109–119 (2013)
Nakano S, Fukaya M. Analysis of proteins responsive to acetic acid in Acetobacter: Molecular mechanisms conferring acetic acid resistance in acetic acid bacteria. Int. J. Food Microbiol. 125: 54–59 (2008)
Hanada T, Kashima Y, Kosugi A, Koizumi Y, Yanagida F, Udaka S. A gene encoding phosphatidylethanolamine N-methyltransferase from Acetobacter aceti and some properties of its disruptant. Biosci. Biotech. Bioch. 65: 2741–2748 (2001)
Zheng Y, Zhang KP, Wang CX, Liu HX, Luo JM, Wang M. Improving acetic acid production of Acetobacter pasteurianus AC2005 in hawthorn vinegar fermentation by using beer for seed culture. Int. J. Food Sci. Tech. 45: 2394–2399 (2010)
Chinnawirotpisan P, Theeragool G, Limtong S, Toyama H, Adachi OO, Matsushita K. Quinoprotein alcohol dehydrogenase is involved in catabolic acetate production, while NAD-dependent alcohol dehydrogenase in ethanol assimilation in Acetobacter pasteurianus SKU1108. J. Biosci. Bioeng. 96: 564–571 (2003)
Tayama K, Fukaya M, Okumura H, Kawamura Y, Beppu T. Purification and characterization of membrane-bound alcohol dehydrogenase from Acetobacter polyoxogenes sp. nov. Appl. Microbiol. Biot. 32: 181–185 (1989)
Berraud C. Production of highly concentrated vinegar in fed-batch culture. Biotechnol. Lett. 22: 451–454 (2000)
Trcek J, Raspor P, Teuber M. Molecular identification of Acetobacter isolates from submerged vinegar production, sequence analysis of plasmid pJK2-1 and application in the development of a cloning vector. Appl. Microbiol. Biot. 53: 289–295 (2000)
Krisch J, Szajani B. Ethanol and acetic acid tolerance in free and immobilized cells of Saccharomyces cerevisiae and Acetobacter aceti. Biotechnol. Lett. 19: 525–528 (1997)
Azuma Y, Hosoyama A, Matsutani M, Furuya N, Horikawa H, Harada T, Hirakawa H, Kuhara S, Fujita N, Shirai M. Whole-genome analyses reveal genetic instability of Acetobacter pasteurianus. Nucleic Acids Res. 37: 5768–5783 (2009)
Shafiei R, Delvigne F, Babanezhad M, Thonart P. Evaluation of viability and growth of Acetobacter senegalensis under different stress conditions. Int. J. Food Microbiol. 163: 204–213 (2013)
Steiner P, Sauer U. Proteins induced during adaptation of Acetobacter aceti to high acetate concentrations. Appl. Environ. Microb. 67: 5474–5481 (2001)
Takemura H, Horinouchi S, Beppu T. Novel insertion sequence IS1380 from Acetobacter pasteurianus is involved in loss of ethanol-oxidizing ability. J. Bacteriol. 173: 7070–7076 (1991)
Steiner P, Sauer U. Long-term continuous evolution of acetate resistant Acetobacter aceti. Biotechnol. Bioeng. 84: 40–44 (2003)
González Á, Hierro N, Poblet M, Mas A, Guillamón JM. Application of molecular methods to demonstrate species and strain evolution of acetic acid bacteria population during wine production. Int. J. Food Microbiol. 102: 295–304 (2005)
Williams JG, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 18: 6531–6535 (1990)
Ameyama M, Adachi O. Alcohol dehydrogenase from acetic acid bacteria, membrane-bound. Methods Enzymol. 89: 450–457 (1982)
Ameyama M, Adachi O. Aldehyde dehydrogenase from acetic acid bacteria, membrane-bound. Methods Enzymol. 89: 491–497 (1982)
Trcek J, Toyama H, Czuba J, Misiewicz A, Matsushita K. Correlation between acetic acid resistance and characteristics of PQQ-dependent ADH in acetic acid bacteria. Appl. Microbiol. Biot. 70: 366–373 (2006)
Takemura H, Kondo K, Horinouchi S, Beppu T. Induction by ethanol of alcohol dehydrogenase activity in Acetobacter pasteurianus. J. Bacteriol. 175: 6857–6866 (1993)
Nanda K, Taniguchi M, Ujike S, Ishihara N, Mori H, Ono H, Murooka Y. Characterization of acetic acid bacteria in traditional acetic acid fermentation of rice vinegar (komesu) and unpolished rice vinegar (kurosu) produced in Japan. Appl. Environ. Microb. 67: 986–990 (2001)
Raja N, Goodson M, Smith DG, Rowbury RJ. Decreased DNA damage by acid and increased repair of acid-damaged DNA in acid-habituated Escherichia coli. J. Appl. Bacteriol. 70: 507–511 (1991)
Hanna MN, Ferguson RJ, Li YH, Cvitkovitch DG. uvrA is an acid-inducible gene involved in the adaptive response to low pH in Streptococcus mutans. J. Bacteriol. 183: 5964–5973 (2001)
Steiner P, Sauer U. Overexpression of the ATP-dependent helicase RecG improves resistance to weak organic acids in Escherichia coli. Appl. Microbiol. Biot. 63: 293–299 (2003)
Zheng Y, Han Q, Jiang CY, Nie ZQ, Wang M. Deletion of gene recG and its susceptibility to acetic acid in Escherichia coli. Lect. Notes Electr. Eng. 249: 351–358 (2014)
Andrés-Barrao C, Saad MM, Chappuis ML, Boffa M, Perret X, Ortega Pérez R, Barja F. Proteome analysis of Acetobacter pasteurianus during acetic acid fermentation. J. Proteomics 75: 1701–1717 (2012)
Ishikawa M, Okamoto-Kainuma A, Jochi T, Suzuki I, Matsui K, Kaga T, Koizumi Y. Cloning and characterization of grpE in Acetobacter pasteurianus NBRC 3283. J. Biosci. Bioeng. 109: 25–31 (2010)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, Y., Zhang, K., Su, G. et al. The evolutionary response of alcohol dehydrogenase and aldehyde dehydrogenases of Acetobacter pasteurianus CGMCC 3089 to ethanol adaptation. Food Sci Biotechnol 24, 133–140 (2015). https://doi.org/10.1007/s10068-015-0019-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-015-0019-x