Abstract
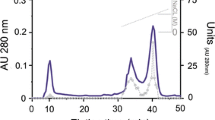
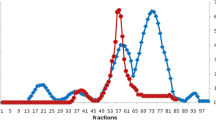
Cathepsin B from the gut of sea cucumber (Stichopus japonicas) was purified 81-fold with a 3% recovery by ammonium sulfate fractionation and a series chromatography on DEAE Sepharose CL-6B, Sephadex G-75, and TSK-Gel 3000 SWxl. The purified protein appeared as a single band on Native-PAGE but showed 2 bands of 23 and 26 kDa on SDS-PAGE. The optimum activity was found at pH 5.5 and 45°C. The enzyme was stable at pH 4.5–6.0 and the thermal stability was up to 50oC. The enzyme was strongly inhibited by E-64, iodoacetic acid, and antipain, demonstrating it is a cysteine protease containing sulfhydryl groups. Cu2+, Ni2+, and Zn2+ could strongly inhibit the enzyme activity. The amino acid sequences of the purified enzyme were acquired by mass spectrometer, which did not show any homology with previously described cathepsins, suggesting it may be a novel member.
Similar content being viewed by others
References
Saito M, Kunisaki N, Urano N, Kimura S. Collagen as the major edible component of sea cucumber (Stichopus japonicus). J. Food Sci. 67: 1319–1322 (2002)
Ho ML, Chen GH, Jiang ST. Effect of mackerel cathepsins L and Llike, and calpain on the degradation of mackerel surimi. Fisheries Sci. 66: 558–568 (2000)
Cao MJ, Hara K, Osatomi K, Tachibana K, Izumi T, Ishihara T. Myofibril-bound serine proteinase (MBP) and its degradation of myofibrillar proteins. J. Food Sci. 64: 644–647 (1999)
Zhu BW, Zheng J, Zhang ZS, Dong XP, Zhao LL, Mikiro T. Autophagy plays a potential role in the process of sea cucumber body wall “melting” induced by UV irradiation. Wuhan Univ. J. Nat. Sci. 13: 232–238 (2008)
Zhu BW, Zhao LL, Sun LM, Li DM, Murata Y, Yu L, Zhang L. Purification and characterization of a cathepsin L-like enzyme from the body wall of the sea cucumber Stichopus japonicus. Biosci. Biotech. Bioch. 72: 1430–1437 (2008)
Qi H, Dong XP, Cong LN, Gao Y, Liu L, Mikiro T, Zhu BW. Purification and characterization of a cysteine-like protease from the body wall of the sea cucumber Stichopus japoricus. Fish Physiol. Biochem. 33: 181–188 (2007)
Kirschke H, Barrett AJ, Rawlings ND. Lysosomal cysteine proteinases. Vol. 2, pp. 1587–1643. In: Protein Profile. Sheterline P (ed). Oxford University Press, Oxford, UK (1998)
Ahmad S, Agarwal SK, Khan MY. Purification and some properties of buffalo spleen cathepsin B. J. Bioscience 14: 261–268 (1989)
Bradley JD, Whitaker JN. Isolation and characterization of cathepsin B from bovine brain. Neurochem. Res. 11: 851–867 (1986)
Sherekar SV, Gor MS, Ninjoor V. Purification and characterization of cathepsin B from the skeletal muscle of fresh water fish, Tilapia mossambica. J. Food Sci. 53: 1018–1023 (1988)
Jiang ST, Lee JJ, Chen HC. Purification and characterization of a novel cysteine proteinase from mackerel (Scomber australasicus). J. Agr. Food Chem. 42: 1639–1646 (1994)
Corticchiato O, Cajot JF, Abrahamson M, Chan SJ, Keppler D, Sordat B. Cystatin C and cathepsin B in human colon carcinoma: Expression in cell lines and matrix degradation. Int. J. Cancer 52: 645–652 (1992)
Coulibaly S, Schwihla H, Abrahamson M, Albini A, Cerni C, Clark JL, Ng KM, Katunuma N, Schlappack O, Glossl J, Mach L. Modulation of the invasive properties of murine squamous carcinoma cells by heterologous expression of cathepsin B and cystatin C. Int. J. Cancer 83: 526–531 (1999)
De Nanteuil G, Portevin B, Benoist A. Disease-modifying antiosteoarthritic drugs: Current herapies and new prospects around protease inhibition. Farmaco 56: 107–112 (2001)
Lowry OH, Rosebrough NJ, Farr, AL, Randall RJ. Protein measurement with the Folin phehol reagent. J. Biol. Chem. 193: 265–275 (1951)
Barrett AJ, Kirschke H. Cathepsin B, cathepsin H, and cathepsin L. Method. Enzymol. 80: 535–561 (1981)
Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685 (1970)
Aranishi F, Hara K, Osatomi K, Ishihara T. Purification and characterization of cathepsin B from hepatopancreas of carp Cyprinus carpio. Comp. Biochem. Phys. B 118: 531–537 (1997)
Towatari T, Kawabata Y, Katunuma N. Crystallization and properties of cathepsin B from rat liver. Eur. J. Biochem. 102: 279–289 (1979)
Takio K, Towatari T, Katunuma N, Teller DC, Titani K. Homology of amino acid sequences of rat liver cathepsins B and H with that of papain. P. Natl. Acad. Sci. USA 80: 3666–3670 (1983)
Kominami E, Tsukahara T, Hara K, Katunuma N. Biosyntheses and processing of lysosomal cysteine proteinases in rat macrophages. FEBS Lett. 231: 225–228 (1988)
Lee JJ, Chen HC, Jiang ST. Comparison of the kinetics of cathepsins B, L, L-like, and X from the dorsal muscle of mackerel on the hydrolysis of methylcoumarylaminde substrates. J. Agr. Food Chem. 44: 774–778 (1996)
Hara K, Suzumatsu A, Ishihara T. Purification and characterization of cathepsin B from carp ordinary muscle. Nippon Suisan Gakk. 54: 1243–1252 (1988)
Swanson AA, Martin BJ, Spicer SS. Human placental cathepsin B1. Isolation and some physical properties. Biochem. J. 137: 223–228 (1974)
An H, Peters MY, Seymour TA, Morrissey MT. Isolation and activation of cathepsin L-inhibitor complex from Pacific whiting (Merluccius products). J. Agr. Food. Chem. 43: 327–330 (1995)
Mark N, Berg J. Rat brain cathepsin L: Characterization and differentiation from cathepsin B utilizing opioid peptides. Arch. Biochem. Biophys. 259: 131–143 (1987)
Yamashita M, Konagaya S. Purification and characterization of cathepsin L from the white muscle of chum salmon, Oncorhynchus keta. Comp. Biochem. Phys. B 96: 247–252 (1990)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, LM., Zhu, BW., Wu, Ht. et al. Purification and characterization of cathepsin B from the gut of the sea cucumber (Stichopus japonicas). Food Sci Biotechnol 20, 919–925 (2011). https://doi.org/10.1007/s10068-011-0127-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-011-0127-1