Abstract
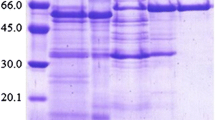
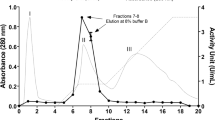
In order to investigate the optimal conditions for raffinose synthesis, α-galactosidase was purified from Absidia corymbifera IFO8084 with a recovery yield of approximately 8.1% (8.36 mg). The molecular weight of the wild-type α-galactosidase was about 83 kDa by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The native molecular mass of the enzyme was approximately 330 kDa by gel filtration chromatography, indicating that α-galactosidase from A. corymbifera IFO8084 is a homotetrameric enzyme. The purified enzyme displayed optimal enzyme activity at pH 4.5 and 60°C. When the purified α-galactosidase was incubated in a substrate solution of sucrose and D-galactose for 48 hr at 37°C, raffinose was synthesized and was confirmed by thin layer chromatography (TLC), high performance liquid chromatography (HPLC), and 13C-nuclear magnetic resonance (13C-NMR) spectrometer analysis. Maximum rates of conversion were observed with 1.67 M galactose, 2.04 M sucrose, and 100 U α-galactosidase at pH 6.0 and 70°C. Under the optimized conditions, the overall conversion ratio was 10%(w/v), representing 2.5 times the synthesis yield that would be possible without the optimized conditions.
Similar content being viewed by others
References
Ballongue J, Bifidobacteria and probiotic action. pp. 357–442. In: Lactic Acid Bacteria, Microbiology, and Functional Aspects. Salminen S, von Wright A (eds). Marcel Dekker, Inc, New York, NY, USA (1998)
Mituoka T. Bifidobacteria and their role in human health. J. Ind. Microbiol. 6: 263–268 (1990)
Gibson GR, Roberfroid MB. Dietary modulation of human colonic microbiota: Introducing the concept of prebiotic action. J. Nutr. 125: 1401–1412 (1995)
Mitsuoka T, Hidaka H, Eida T. Effect of fructooligosaccharides on intestinal microflora. Nahrung 31: 5–6 (1987)
Lehle L, Tanner W. The function of myo-inositol in the biosynthesis of raffinose. Eur. J. Biochem. 38: 103–110 (1973)
Schmidt RR. New methods for the synthesis of glycosides and oligosaccharides. Are there alternatives to the Koenigs-Knorr methods? Angew. Chem. Int. Edit. 25: 212–235 (1986)
Wong CH, Haynie SL, Whitesides GM. Enzyme-catalyzed synthesis of N-acetu;actpsa, ome wotj in situ regeneration of uridine 5′-diphosphate glucose and uridine 5′-diphosphate galactose. J. Org. Chem. 47: 5416–5418 (1982)
Lowry OH, Farr NJ, Randall RL. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275 (1951)
Laemmli UK. Cleavage of structural protein during the assembly of the head of bacteriophage T4. Nature 227: 680–685 (1970)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baik, SH. Synthesis of raffinose by fungal α-galacotosidase from Absidia corymbifera . Food Sci Biotechnol 19, 83–87 (2010). https://doi.org/10.1007/s10068-010-0012-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-010-0012-3