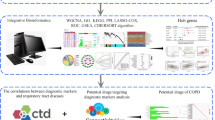
Abstract
Background
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease, whose development is associated with immune cells and persistent inflammation. Exploring the biomarkers of RA holds immense significance in terms of the prevention, diagnosis, and treatment of RA.
Material and methods
The differentially expressed genes (DEGs) in RA patients and the control group were screened by limma package. Through DEGs intersection overlapping 200 inflammatory response-related genes and 2498 immune-related genes, differentially expressed immune and inflammation-related genes (DE-IIRGs) were identified. Lasso regression analysis screened RA diagnostic biomarkers and constructed PPI networks. Finally, immune infiltration analysis and drug prediction were performed.
Results
A total of 20 DE-IIRGs were identified by overlapping DEGs with 2498 immune-related genes and 200 inflammatory response-related genes. These DE-IIRGs were primarily enriched in the cytokine-cytokine receptor interaction and other biological processes, and then five biomarker genes (TNFSF10, IL1R1, CXCL9, ACVR1B, and IL15) were identified. It was found that the expression levels of CXCL9, IL15, and TNFSF10 in the disease samples were significantly higher than those in the control group. These biomarker genes have more effective diagnostic potential. The RA samples exhibited significantly higher levels of cell infiltration compared to the control samples. hsa-miR-199a-5p’s connections to the ACVR1B and CCR7 genes were identified by creating ceRNA networks from 20 screened DE-IIRGs. There was a connection between CCL5 and AEMA4D and hsa-miR-214-3p.
Conclusion
We identified immune- and inflammation-related biomarkers in RA based on bioinformatics analysis and screened TNFSF10, IL1R1, CXCL9, ACVR1B, and IL15 as diagnostic markers for RA.
Key Points • TNFSF10, IL1R1, CXCL9, ACVR1B, and IL15 may be new diagnostic biomarkers for RA. • These findings may provide a theoretical basis for early RA diagnosis. |







Similar content being viewed by others
Data availability
All relevant data are within the manuscript and its Additional files.
References
Radu AF, Bungau SG (2021) Management of rheumatoid arthritis: an overview. Cells 10:11. https://doi.org/10.3390/cells10112857
Smolen JS, Aletaha D, Mcinnes IB (2016) Rheumatoid arthritis. Lancet (London, England) 388(10055):2023–2038. https://doi.org/10.1016/S0140-6736(16)30173-8
Scherer HU, Häupl T, Burmester GR (2020) The etiology of rheumatoid arthritis. J Autoimmun 110:102400. https://doi.org/10.1016/j.jaut.2019.102400
Littlejohn EA, Monrad SU (2018) Early diagnosis and treatment of rheumatoid arthritis. Prim Care 45(2):237–255. https://doi.org/10.1016/j.pop.2018.02.010
Wang C, Lutes LK, Barnoud C, Scheiermann C (2022) The circadian immune system. Sci Immunol 7(72):2465. https://doi.org/10.1126/sciimmunol.abm2465
Jang S, Kwon EJ, Lee JJ (2022) Rheumatoid arthritis: pathogenic roles of diverse immune cells. Int J Mol Sci 23:2. https://doi.org/10.3390/ijms23020905
Ikeda H, Togashi Y (2022) Aging, cancer, and antitumor immunity. Int J Clin Oncol 27(2):316–322. https://doi.org/10.1007/s10147-021-01913-z
Weyand CM, Goronzy JJ (2021) The immunology of rheumatoid arthritis. Nat Immunol 22(1):10–18
Gravallese EM, Firestein GS (2023) Rheumatoid arthritis - common origins, divergent mechanisms. New Engl J Med 388(6):529–542. https://doi.org/10.1056/NEJMra2103726
Khandia R, Munjal A (2016) Interplay between inflammation and cancer. Adv Protein Chem Struct Biol 119:199–245. https://doi.org/10.1016/bs.apcsb.2019.09.004
Suzuki K (2019) Chronic inflammation as an immunological abnormality and effectiveness of exercise. Biomolecules 9:6. https://doi.org/10.3390/biom9060223
Liberzon A, Birger C, Thorvaldsdóttir H et al (2015) The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst 1(6):417–425. https://doi.org/10.1016/j.cels.2015.12.004
Liu S, Wang Z, Zhu R, et al (2021) Three differential expression analysis methods for RNA sequencing: limma, EdgeR, DESeq2. J Vis Exp: JoVE (175). https://doi.org/10.3791/62528
Cotto KC, Wagner AH, Feng YY et al (2018) DGIdb 3.0: a redesign and expansion of the drug-gene interaction database. Nucleic Acids Res 46(D1):D1068–D1073. https://doi.org/10.1093/nar/gkx1143
Smith MH, Berman JR (2022) What is rheumatoid arthritis? JAMA 327(12):1194. https://doi.org/10.1001/jama.2022.0786
Daëron M (2022) The immune system as a system of relations. Front Immunol 13:984678. https://doi.org/10.3389/fimmu.2022.984678
Negrini S, Emmi G, Greco M et al (2022) Sjögren’s syndrome: a systemic autoimmune disease. Clin Exp Med 22(1):9–25. https://doi.org/10.1007/s10238-021-00728-6
Jiang Q, Yang G, Liu Q et al (2021) Function and role of regulatory T cells in rheumatoid arthritis. Front Immunol 12:626193. https://doi.org/10.3389/fimmu.2021.626193
Maeda K, Yoshida K, Nishizawa T et al (2022) Inflammation and bone metabolism in rheumatoid arthritis: molecular mechanisms of joint destruction and pharmacological treatments. Int J Mol Sci 23:5. https://doi.org/10.3390/ijms23052871
Li Z, Chen Y, Zulipikaer M et al (2022) Identification of PSMB9 and CXCL13 as immune-related diagnostic markers for rheumatoid arthritis by machine learning. Curr Pharm Des 28(34):2842–2854. https://doi.org/10.2174/1381612828666220831085608
Li A, Zhang Z, Ru X et al (2022) Identification of SLAMF1 as an immune-related key gene associated with rheumatoid arthritis and verified in mice collagen-induced arthritis model. Front Immunol 13:961129. https://doi.org/10.3389/fimmu.2022.961129
Balasundaram A, Udhaya Kumar S, George Priya DC (2022) A computational model revealing the immune-related hub genes and key pathways involved in rheumatoid arthritis (RA). Adv Protein Chem Struct Biol 129:247–273. https://doi.org/10.1016/bs.apcsb.2021.11.006
Fang S, Xu X, Zhong L et al (2021) Bioinformatics-based study to identify immune infiltration and inflammatory-related hub genes as biomarkers for the treatment of rheumatoid arthritis. Immunogenetics 73(6):435–448. https://doi.org/10.1007/s00251-021-01224-7
Tsubaki T, Takegawa S, Hanamoto H et al (2005) Accumulation of plasma cells expressing CXCR3 in the synovial sublining regions of early rheumatoid arthritis in association with production of Mig/CXCL9 by synovial fibroblasts. Clin Exp Immunol 141(2):363–371. https://doi.org/10.1111/j.1365-2249.2005.02850.x
Shi Y, Xie F, Rao P et al (2020) TRAIL-expressing cell membrane nanovesicles as an anti-inflammatory platform for rheumatoid arthritis therapy. J Control Release 320:304–313. https://doi.org/10.1016/j.jconrel.2020.01.054
Wu YL, Ke J, Zhang BY et al (2022) Hepatitis virus reactivation in rheumatoid arthritis. World J Clin Cases 10(1):12–22. https://doi.org/10.12998/wjcc.v10.i1.12
Lobbes H, Dervout C, Toussirot E et al (2020) Rituximab for rheumatoid arthritis-associated large granular lymphocytic leukemia, a retrospective case series. Semin Arthritis Rheum 50(5):1109–1113. https://doi.org/10.1016/j.semarthrit.2020.05.020
Anang DC, Ramwadhdoebe TH, Hähnlein JS et al (2022) Increased frequency of CD4(+) follicular helper T and CD8(+) follicular T cells in human lymph node biopsies during the earliest stages of rheumatoid arthritis. Cells 11:7. https://doi.org/10.3390/cells11071104
Cao G, Mao Z, Niu T et al (2023) Oxymatrine ameliorates rheumatoid arthritis by regulation of Tfr/Tfh cell balance via the TLR9-MyD88-STAT3 signaling pathway. J Sci Food Agric 103(12):6017–6024. https://doi.org/10.1002/jsfa.12674
Funding
The Anhui Provincial Natural Science Foundation,2308085MH252
Author information
Authors and Affiliations
Contributions
Xijie Bao independently designed the research, completed data download and analysis work, drafted all the figures of the manuscript, and composed the full text of the article. The author has read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
All figures submitted have been created by the authors, who confirm that the images are original with no duplication and have not been previously published in whole or in part.
Disclosures
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bao, X. Validation of new immune and inflammation-related diagnostic biomarkers for RA. Clin Rheumatol 43, 949–958 (2024). https://doi.org/10.1007/s10067-024-06882-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-024-06882-y