Abstract
Introduction/objectives
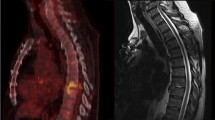
Cervical spine involvement is one of the most serious complications in rheumatoid arthritis (RA). The study aim was to assess the clinical significance of atlantoaxial (AA) joint involvement detected by 18F-fluorodeoxyglucose (FDG)-positron emission tomography (PET) with computed tomography (CT) in patients with RA.
Method
A prospective cross-sectional study was conducted to evaluate AA joint involvement detected by FDG-PET/CT in consecutive RA patients from December 2017 to February 2022. We investigated the relationship between AA joint involvement and clinical data, including disease activity and patients’ cervical symptoms.
Results
Among 48 patients enrolled, abnormal FDG uptake at AA joint was detected in 13 (27%). Rheumatoid factor titre, initial disease activity score 28-erythrocyte sedimentation rate and total standardized uptake value were significantly higher in the 13 patients than in the others (P = 0.004, P < 0.001 and P = 0.001, respectively). All patients with abnormal FDG uptake at AA joint had some cervical symptoms regardless of cervical spine X-ray abnormalities. Neck pain on movement and at rest were more frequent in the 13 patients than in the others (P = 0.001 and P = 0.004, respectively). The most sensitive symptom associated with AA joint involvement was neck pain on movement (sensitivity, 69%), and the most specific symptom was neck pain at rest (specificity, 100%).
Conclusions
AA joint involvement was commonly observed by FDG-PET/CT in patients with active RA, independent of radiographic findings. Specific cervical symptoms can be important surrogate markers for detection of potential AA synovitis associated with active RA.
Key Points • AA joint involvement was frequently seen in RA with high disease activity independent of radiographic findings. • Neck pain was a hallmark of AA joint involvement reflecting disease activity, and resting pain was highly specific. |

Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
McInnes IB, Schett G (2017) Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet 389:2328–2337. https://doi.org/10.1016/S0140-6736(17)31472-1
Gillick JL, Wainwright J, Das K (2015) Rheumatoid arthritis and the cervical spine: a review on the role of surgery. Int J Rheumatol 2015:252456. https://doi.org/10.1155/2015/252456
Kotecki M, Sotniczuk M, Gietka P, Gasik R, Sudol-Szopinska I (2021) Imaging of cervical spine involvement in inflammatory arthropathies: a review. Pol J Radiol 86:e620–e629. https://doi.org/10.5114/pjr.2021.111363
Wasserman BR, Moskovich R, Razi AE (2011) Rheumatoid arthritis of the cervical spine--clinical considerations. Bull NYU Hosp Jt Dis 69:136–148
Neva MH, Hakkinen A, Makinen H, Hannonen P, Kauppi M et al (2006) High prevalence of asymptomatic cervical spine subluxation in patients with rheumatoid arthritis waiting for orthopaedic surgery. Ann Rheum Dis 65:884–888. https://doi.org/10.1136/ard.2005.042135
Shlobin NA, Dahdaleh NS (2021) Cervical spine manifestations of rheumatoid arthritis: a review. Neurosurg Rev 44:1957–1965. https://doi.org/10.1007/s10143-020-01412-1
Zhang T, Pope J (2015) Cervical spine involvement in rheumatoid arthritis over time: results from a meta-analysis. Arthritis Res Ther 17:148. https://doi.org/10.1186/s13075-015-0643-0
Manczak M, Gasik R (2017) Cervical spine instability in the course of rheumatoid arthritis - imaging methods. Reumatologia 55:201–207. https://doi.org/10.5114/reum.2017.69782
Baker JF, Conaghan PG, Gandjbakhch F (2018) Update on magnetic resonance imaging and ultrasound in rheumatoid arthritis. Clin Exp Rheumatol 36(Suppl 114):16–23
Kono M, Kamishima T, Yasuda S, Sakamoto K, Abe S et al (2017) Effectiveness of whole-body magnetic resonance imaging for the efficacy of biologic anti-rheumatic drugs in patients with rheumatoid arthritis: a retrospective pilot study. Mod Rheumatol 27:953–960. https://doi.org/10.1080/14397595.2016.1276425
Magarelli N, Simone F, Amelia R, Leone A, Bosello S et al (2010) MR imaging of atlantoaxial joint in early rheumatoid arthritis. Radiol Med 115:1111–1120. https://doi.org/10.1007/s11547-010-0574-4
Beckers C, Ribbens C, Andre B, Marcelis S, Kaye O et al (2004) Assessment of disease activity in rheumatoid arthritis with (18)F-FDG PET. J Nucl Med 45:956–964
Fosse P, Kaiser MJ, Namur G, de Seny D, Malaise MG et al (2018) (18)F- FDG PET/CT joint assessment of early therapeutic response in rheumatoid arthritis patients treated with rituximab. Eur J Hybrid Imaging 2:6. https://doi.org/10.1186/s41824-017-0022-y
Elzinga EH, van der Laken CJ, Comans EF, Boellaard R, Hoekstra OS et al (2011) 18F-FDG PET as a tool to predict the clinical outcome of infliximab treatment of rheumatoid arthritis: an explorative study. J Nucl Med 52:77–80. https://doi.org/10.2967/jnumed.110.076711
Kubota K, Ito K, Morooka M, Mitsumoto T, Kurihara K et al (2009) Whole-body FDG-PET/CT on rheumatoid arthritis of large joints. Ann Nucl Med 23:783–791. https://doi.org/10.1007/s12149-009-0305-x
Okamura K, Yonemoto Y, Okura C, Higuchi T, Tsushima Y et al (2014) Evaluation of tocilizumab therapy in patients with rheumatoid arthritis based on FDG-PET/CT. BMC Musculoskelet Disord 15:393. https://doi.org/10.1186/1471-2474-15-393
Suto T, Yonemoto Y, Okamura K, Okura C, Kaneko T et al (2017) Predictive factors associated with the progression of large-joint destruction in patients with rheumatoid arthritis after biologic therapy: a post-hoc analysis using FDG-PET/CT and the ARASHI (assessment of rheumatoid arthritis by scoring of large-joint destruction and healing in radiographic imaging) scoring method. Mod Rheumatol 27:820–827. https://doi.org/10.1080/14397595.2016.1266132
Yonemoto Y, Okamura K, Takeuchi K, Kaneko T, Kobayashi T et al (2016) [18F] fluorodeoxyglucose uptake as a predictor of large joint destruction in patients with rheumatoid arthritis. Rheumatol Int 36:109–115. https://doi.org/10.1007/s00296-015-3331-0
Suto T, Okamura K, Yonemoto Y, Okura C, Tsushima Y et al (2016) Prediction of large joint destruction in patients with rheumatoid arthritis using 18F-FDG PET/CT and disease activity score. Medicine (Baltimore) 95:e2841. https://doi.org/10.1097/MD.0000000000002841
Yonemoto Y, Okamura K, Kobayashi T, Kaneko T, Okura C et al (2017) Predictive factors related to shoulder joint destruction in rheumatoid arthritis patients treated with biologics: a prospective study. Mod Rheumatol 27:587–592. https://doi.org/10.1080/14397595.2016.1245125
Kaneta T, Hakamatsuka T, Yamada T, Takase K, Sato A et al (2006) Atlantoaxial osteoarthritis in rheumatoid arthritis: FDG PET/CT findings. Clin Nucl Med 31:209. https://doi.org/10.1097/01.rlu.0000204124.79497.bb
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581. https://doi.org/10.1002/art.27584
Goerres GW, Forster A, Uebelhart D, Seifert B, Treyer V et al (2006) F-18 FDG whole-body PET for the assessment of disease activity in patients with rheumatoid arthritis. Clin Nucl Med 31:386–390. https://doi.org/10.1097/01.rlu.0000222678.95218.42
Matsui T, Nakata N, Nagai S, Nakatani A, Takahashi M et al (2009) Inflammatory cytokines and hypoxia contribute to 18F-FDG uptake by cells involved in pannus formation in rheumatoid arthritis. J Nucl Med 50:920–926. https://doi.org/10.2967/jnumed.108.060103
Chaudhari AJ, Ferrero A, Godinez F, Yang K, Shelton DK et al (2016) High-resolution (18)F-FDG PET/CT for assessing disease activity in rheumatoid and psoriatic arthritis: findings of a prospective pilot study. Br J Radiol 89:20160138. https://doi.org/10.1259/bjr.20160138
Lee SJ, Jeong JH, Lee CH, Ahn BC, Eun JS et al (2019) Development and validation of an (18) F-fluorodeoxyglucose-positron emission tomography with computed tomography-based tool for the evaluation of joint counts and disease activity in patients with rheumatoid arthritis. Arthritis Rheumatol 71:1232–1240. https://doi.org/10.1002/art.40860
Zhu S, Xu W, Luo Y, Zhao Y, Liu Y (2017) Cervical spine involvement risk factors in rheumatoid arthritis: a meta-analysis. Int J Rheum Dis 20:541–549. https://doi.org/10.1111/1756-185X.13096
American College of Rheumatology Ad Hoc Committee on Clinical Guidelines (1996) Guidelines for the initial evaluation of the adult patient with acute musculoskeletal symptoms. Arthritis Rheum 39:1–8. https://doi.org/10.1002/art.1780390102
Elzinga EH, van der Laken CJ, Comans EF, Lammertsma AA, Dijkmans BA et al (2007) 2-Deoxy-2-[F-18]fluoro-D-glucose joint uptake on positron emission tomography images: rheumatoid arthritis versus osteoarthritis. Mol Imaging Biol 9:357–360. https://doi.org/10.1007/s11307-007-0113-4
Chaudhari AJ, Bowen SL, Burkett GW, Packard NJ, Godinez F et al (2010) High-resolution (18)F-FDG PET with MRI for monitoring response to treatment in rheumatoid arthritis. Eur J Nucl Med Mol Imaging 37:1047. https://doi.org/10.1007/s00259-009-1364-x
Monet A, Massonnat R, Merino B, Riviere A, Richez C (2014) Crowned dens syndrome diagnosed on (1)(8)F-FDG PET/CT. Clin Nucl Med 39:1041–1042. https://doi.org/10.1007/s40336-021-00445-w
Casali M, Lauri C, Altini C, Bertagna F, Cassarino G et al (2021) State of the art of (18)F-FDG PET/CT application in inflammation and infection: a guide for image acquisition and interpretation. Clin Transl Imaging 9:299–339. https://doi.org/10.1097/RLU.0000000000000544
Acknowledgements
We thank all of the medical staff in the Department of Internal Medicine, Radiology Section, and in the Nuclear Medicine Centre in our hospital for their clinical contributions. The authors would like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Contributions
TK contributed to the study conception and design. AN collected patients’ clinical data and performed the statistical analysis. KM interpreted FDG-PET/CT images including SUV scoring. The first draft of the manuscript was written by AN and TK. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical standards
Our study complies with the principles of the Declaration of Helsinki, the locally appointed ethics committee approved the research protocol and written informed consent was obtained from the subjects.
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Noguchi, A., Kurita, T. & Matsuzawa, K. Clinical features of atlantoaxial involvement in patients with rheumatoid arthritis using 18F-fluorodeoxyglucose-positron emission tomography with computed tomography. Clin Rheumatol 42, 1875–1884 (2023). https://doi.org/10.1007/s10067-023-06586-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-023-06586-9