Abstract
Introduction/objectives
Systemic sclerosis (SSc) is characterized by generalized vasculopathy affecting mainly small vessels while macrovascular involvement is less investigated. The aim of this study was to examine associations between asymmetric dimethylarginine (ADMA) — a biomarker of atherosclerosis — and assessments of macrovascular endothelial function in patients with SSc.
Methods
This was a cross-sectional study including consecutive SSc patients attending the Scleroderma Outpatient Clinic. ADMA measurement in serum samples was based on an enzyme immunoassay technique. Participants underwent blood pressure measurement according to 2018 ESC/ESH Guidelines, applanation tonometry for the evaluation of arterial stiffness, and carotid ultrasound for the measurement of the intima-media thickness (cIMT).
Results
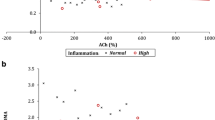
Eighty-one Caucasians (82.3% female) SSc individuals with mean age 55.44 ± 13.4 years were included in this analysis. The correlation analysis of ADMA levels (unadjusted and adjusted values) with functional and morphological parameters of atherosclerosis revealed no statistically significant associations. Subgroup analysis based on disease duration (≤ 4 years), immunologic profile (SCL-70 and ACA antibodies), disease type (limited, diffuse), and inflammatory status (erythrocyte sedimentation rate [ESR] > 25 mm/h and C-reactive protein [CRP] > 5 mg/L) showed no associations, except from a significant positive correlation between ADMA levels and cΙΜΤmean (r = 0.370, p = 0.044) in individuals with early SSc.
Conclusions
The results of the study suggest that ADMA may be related with accelerated atherosclerosis in early stages of the disease. However, the lack of association between other morphological and functional parameters of endothelial dysfunction may suggest that other regulators of nitric oxide metabolism may contribute to macrovascular injury in SSc in various phases of the disease.
Key Points • ADMA is a biomarker of atherosclerosis and has been linked with microvascular complications of SSc. •ADMA was not correlated with morphological and functional parameters of atherosclerosis in the population of the study. •The demonstrated association between ADMA and cIMT in patients with early SSc may suggest a role of NO/ADMA pathway in the initiation of macrovascular injury in SSc. |


Similar content being viewed by others
Abbreviations
- ADMA:
-
Asymmetrical dimethylarginine
- SSc:
-
Systemic sclerosis
- cIMT:
-
Carotid intima-media thickness
- ACA:
-
Anti-centromeric antibodies
- SCL-70:
-
Anti-topoisomerase IIa
References
Elhai M, Meune C, Avouac J, Kahan A, Allanore Y (2012) Trends in mortality in patients with systemic sclerosis over 40 years: a systematic review and meta-analysis of cohort studies. Rheumatology (Oxford) 51:1017–1026. https://doi.org/10.1093/rheumatology/ker269
Poudel DR, Jayakumar D, Danve A, Sehra ST, Derk CT (2018) Determinants of mortality in systemic sclerosis: a focused review. Rheumatol Int 38:1847–1858. https://doi.org/10.1007/s00296-017-3826-y
Ngian GS, Sahhar J, Proudman SM, Stevens W, Wicks IP, Van Doornum S (2012) Prevalence of coronary heart disease and cardiovascular risk factors in a national cross-sectional cohort study of systemic sclerosis. Ann Rheum Dis 71:1980–1983. https://doi.org/10.1136/annrheumdis-2011-201176
Tyndall AJ, Bannert B, Vonk M et al (2010) Causes and risk factors for death in systemic sclerosis: a study from the EULAR Scleroderma Trials and Research (EUSTAR) database. Ann Rheum Dis 69:1809–1815. https://doi.org/10.1136/ard.2009.114264
Aviña-Zubieta JA, Man A, Yurkovich M, Huang K, Sayre EC, Choi HK (2016) Early cardiovascular disease after the diagnosis of systemic sclerosis. Am J Med 129:324–331. https://doi.org/10.1016/j.amjmed.2015.10.037
Au K, Singh MK, Bodukam V et al (2011) Atherosclerosis in systemic sclerosis: a systematic review and meta-analysis. Arthritis Rheum 63:2078–2090. https://doi.org/10.1002/art.30380
Sandoo A, Veldhuijzen van Zanten JJ, Metsios GS, Carroll D, Kitas GD (2011) Vascular function and morphology in rheumatoid arthritis: a systematic review. Rheumatology (Oxford) 50:2125–2139. https://doi.org/10.1093/rheumatology/ker275
Kravvariti E, Konstantonis G, Sfikakis PP, Tektonidou MG (2018) Progression of subclinical atherosclerosis in systemic lupus erythematosus versus rheumatoid arthritis: the impact of low disease activity. Rheumatology (Oxford) 57:2158–2166. https://doi.org/10.1093/rheumatology/key233
Cypiene A, Laucevicius A, Venalis A, Dadoniene J, Ryliskyte L, Petrulioniene Z, Kovaite M, Gintautas J (2008) The impact of systemic sclerosis on arterial wall stiffness parameters and endothelial function. Clin Rheumatol 27:1517–1522. https://doi.org/10.1007/s10067-008-0958-1
Meiszterics Z, Tímár O, Gaszner B, Faludi R, Kehl D, Czirják L, Szűcs G, Komócsi A (2016) Early morphologic and functional changes of atherosclerosis in systemic sclerosis—a systematic review and meta-analysis. Rheumatology (Oxford) 55:2119–2130. https://doi.org/10.1093/rheumatology/kew236
Lieb W, Benndorf RA, Benjamin EJ et al (2009) Plasma asymmetric dimethylarginine, L-arginine and left ventricular structure and function in a community-based sample. Atherosclerosis 204:282–287. https://doi.org/10.1016/j.atherosclerosis.2008.08.018
Zhang H, Xiang S, Dai Z, Fan Y (2021) Asymmetric dimethylarginine level as biomarkers of cardiovascular or all-cause mortality in patients with chronic kidney disease: a meta-analysis. Biomarkers 26:579–585. https://doi.org/10.1080/1354750x.2021.1954694
Sheikh Rezaei S, Weisshaar S, Litschauer B, Gouya G, Ohrenberger G, Wolzt M (2019) ADMA and NT pro-BNP are associated with overall mortality in elderly. Eur J Clin Invest 49:e13041. https://doi.org/10.1111/eci.13041
Cziráki A, Lenkey Z, Sulyok E, Szokodi I, Koller A (2020) L-arginine-nitric oxide-asymmetric dimethylarginine pathway and the coronary circulation: translation of basic science results to clinical practice. Front Pharmacol 11:569914. https://doi.org/10.3389/fphar.2020.569914
Dimitroulas T, Sandoo A, Kitas GD (2012) Asymmetric dimethylarginine as a surrogate marker of endothelial dysfunction and cardiovascular risk in patients with systemic rheumatic diseases. Int J Mol Sci 13:12315–12335. https://doi.org/10.3390/ijms131012315
Surdacki A, Martens-Lobenhoffer J, Wloch A et al (2007) Elevated plasma asymmetric dimethyl-L-arginine levels are linked to endothelial progenitor cell depletion and carotid atherosclerosis in rheumatoid arthritis. Arthritis Rheum 56:809–819. https://doi.org/10.1002/art.22424
Sandoo A, Dimitroulas T, Veldhuijzen van Zanten JJ, Smith JP, Metsios GS, Nightingale P, Stavropoulos-Kalinoglou A, Kitas GD (2012) Lack of association between asymmetric dimethylarginine and in vivo microvascular and macrovascular endothelial function in patients with rheumatoid arthritis. Clin Exp Rheumatol 30:388–396
Dimitroulas T, Hodson J, Sandoo A, Smith J, Kitas GD (2017) Endothelial injury in rheumatoid arthritis: a crosstalk between dimethylarginines and systemic inflammation. Arthritis Res Ther 19:32. https://doi.org/10.1186/s13075-017-1232-1
Dimitroulas T, Giannakoulas G, Sfetsios T, Karvounis H, Dimitroula H, Koliakos G, Settas L (2008) Asymmetrical dimethylarginine in systemic sclerosis-related pulmonary arterial hypertension. Rheumatology (Oxford) 47:1682–1685. https://doi.org/10.1093/rheumatology/ken346
Silva I, Teixeira A, Oliveira J, Almeida I, Almeida R, Águas A, Vasconcelos C (2015) Endothelial dysfunction and nailfold videocapillaroscopy pattern as predictors of digital ulcers in systemic sclerosis: a cohort study and review of the literature. Clin Rev Allergy Immunol 49:240–252. https://doi.org/10.1007/s12016-015-8500-0
Dimitroulas T, Giannakoulas G, Papadopoulou K et al (2010) Early detection of cardiac involvement in systemic sclerosis assessed by tissue-Doppler echocardiography: relationship with neurohormonal activation and endothelial dysfunction. J Rheumatol 37:993–999. https://doi.org/10.3899/jrheum.090931
Rajagopalan S, Pfenninger D, Kehrer C et al (2003) Increased asymmetric dimethylarginine and endothelin 1 levels in secondary Raynaud’s phenomenon: implications for vascular dysfunction and progression of disease. Arthritis Rheum 48:1992–2000. https://doi.org/10.1002/art.11060
van den Hoogen F, Khanna D, Fransen J et al (2013) 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League Against Rheumatism Collaborative Initiative. Ann Rheum Dis 72:1747–1755. https://doi.org/10.1136/annrheumdis-2013-204424
Williams B, Mancia G, Spiering W et al (2018) 2018 ESC/ESH Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J Hypertens 36:1953–2041. https://doi.org/10.1097/HJH.0000000000001940
Saito K, Hishiki Y, Takahashi H (2020) Validation of the Omron HBP-1320 for professional use according to the ANSI/AAMI/ISO 81060–2: 2013 protocol and the 2010 revision of the European Society of Hypertension International Protocol. Blood Press Monit 25:162–166. https://doi.org/10.1097/mbp.0000000000000437
Frech TM, Penrod J, Battistone MJ, Sawitzke AD, Stults BM (2012) The prevalence and clinical correlates of an auscultatory gap in systemic sclerosis patients. Int J Rheumatol 2012:590845. https://doi.org/10.1155/2012/590845
Roman MJ, Naqvi TZ, Gardin JM, Gerhard-Herman M, Jaff M, Mohler E (2006) Clinical application of noninvasive vascular ultrasound in cardiovascular risk stratification: a report from the American Society of Echocardiography and the Society of Vascular Medicine and Biology. J Am Soc Echocardiogr 19:943–954. https://doi.org/10.1016/j.echo.2006.04.020
DeLoach SS, Townsend RR (2008) Vascular stiffness: its measurement and significance for epidemiologic and outcome studies. Clin J Am Soc Nephrol 3:184–192. https://doi.org/10.2215/CJN.03340807
Townsend RR, Wilkinson IB, Schiffrin EL et al (2015) Recommendations for improving and standardizing vascular research on arterial stiffness: a scientific statement from the American Heart Association. Hypertension 66:698–722. https://doi.org/10.1161/HYP.0000000000000033
Schulze F, Wesemann R, Schwedhelm E, Sydow K, Albsmeier J, Cooke JP, Boger RH (2004) Determination of asymmetric dimethylarginine (ADMA) using a novel ELISA assay. Clin Chem Lab Med 42:1377–1383. https://doi.org/10.1515/CCLM.2004.257
Dooley A, Gao B, Bradley N, Abraham DJ, Black CM, Jacobs M, Bruckdorfer KR (2006) Abnormal nitric oxide metabolism in systemic sclerosis: increased levels of nitrated proteins and asymmetric dimethylarginine. Rheumatology (Oxford) 45:676–684. https://doi.org/10.1093/rheumatology/kei276
Romero LI, Zhang DN, Cooke JP, Ho HK, Avalos E, Herrera R, Herron GS (2000) Differential expression of nitric oxide by dermal microvascular endothelial cells from patients with scleroderma. Vasc Med 5:147–158. https://doi.org/10.1177/1358836x0000500304
Matucci Cerinic M, Kahaleh MB (2002) Beauty and the beast. The nitric oxide paradox in systemic sclerosis. Rheumatology (Oxford) 41:843–847. https://doi.org/10.1093/rheumatology/41.8.843
Zhang L, Wan YN, Zhao JH, Wang YJ, Wang YX, Yan JW, Huang XL, Wang J (2015) The association between systemic sclerosis, arginine and asymmetric dimethylarginine. Inflammation 38:218–223. https://doi.org/10.1007/s10753-014-0025-9
Jud P, Meinitzer A, Strohmaier H et al (2021) Evaluation of endothelial dysfunction and clinical events in patients with early-stage vasculopathy in limited systemic sclerosis. Clin Exp Rheumatol 39(131):57–65. https://doi.org/10.55563/clinexprheumatol/243mpp
Moschetti L, Piantoni S, Vizzardi E, Sciatti E, Riccardi M, Franceschini F, Cavazzana I (2022) Endothelial dysfunction in systemic lupus erythematosus and systemic sclerosis: a common trigger for different microvascular diseases. Front Med (Lausanne) 9:849086. https://doi.org/10.3389/fmed.2022.849086
Maas R (2005) Pharmacotherapies and their influence on asymmetric dimethylargine (ADMA). Vasc Med 10(Suppl 1):S49-57. https://doi.org/10.1191/1358863x05vm605oa
Zinellu A, Mangoni AA (2022) An updated systematic review and meta-analysis of the effect of statins on asymmetric dimethylarginine. Nitric Oxide 120:26–37. https://doi.org/10.1016/j.niox.2022.01.001
Allanore Y, Distler O, Matucci-Cerinic M, Denton CP (2018) Review: defining a unified vascular phenotype in systemic sclerosis. Arthritis Rheumatol 70:162–170. https://doi.org/10.1002/art.40377
Psarras A, Soulaidopoulos S, Garyfallos A, Kitas G, Dimitroulas T (2017) A critical view on cardiovascular risk in systemic sclerosis. Rheumatol Int 37:85–95. https://doi.org/10.1007/s00296-016-3530-3
Butt SA, Jeppesen JL, Torp-Pedersen C, Sam F, Gislason GH, Jacobsen S, Andersson C (2019) Cardiovascular manifestations of systemic sclerosis: a Danish nationwide cohort study. J Am Heart Assoc 8:e013405. https://doi.org/10.1161/jaha.119.013405
Ferraz-Amaro I, Delgado-Frías E, Hernández-Hernández V, Sánchez-Pérez H, de Armas-Rillo L, Armas-González E, Machado JD, Diaz-González F (2021) HDL cholesterol efflux capacity and lipid profile in patients with systemic sclerosis. Arthritis Res Ther 23:62. https://doi.org/10.1186/s13075-021-02443-9
Man A, Zhu Y, Zhang Y, Dubreuil M, Rho YH, Peloquin C, Simms RW, Choi HK (2013) The risk of cardiovascular disease in systemic sclerosis: a population-based cohort study. Ann Rheum Dis 72:1188–1193. https://doi.org/10.1136/annrheumdis-2012-202007
Nordin A, Jensen-Urstad K, Björnådal L, Pettersson S, Larsson A, Svenungsson E (2013) Ischemic arterial events and atherosclerosis in patients with systemic sclerosis: a population-based case-control study. Arthritis Res Ther 15:R87. https://doi.org/10.1186/ar4267
el Tarek G, Yasser AE, Gheita T (2006) Coronary angiographic findings in asymptomatic systemic sclerosis. Clin Rheumatol 25:487–490. https://doi.org/10.1007/s10067-005-0073-5
Wang Z, Tang WH, Cho L, Brennan DM, Hazen SL (2009) Targeted metabolomic evaluation of arginine methylation and cardiovascular risks: potential mechanisms beyond nitric oxide synthase inhibition. Arterioscler Thromb Vasc Biol 29:1383–1391. https://doi.org/10.1161/atvbaha.109.185645
Dimitroulas T, Kitas GD (2019) Genetic regulation of dimethylarginines and endothelial dysfunction in rheumatoid arthritis. Amino Acids 51:983–990. https://doi.org/10.1007/s00726-019-02740-3
Łuczak A, Madej M, Kasprzyk A, Doroszko A (2020) Role of the eNOS uncoupling and the nitric oxide metabolic pathway in the pathogenesis of autoimmune rheumatic diseases. Oxid Med Cell Longev 2020:1417981. https://doi.org/10.1155/2020/1417981
Sandoo A, Dimitroulas T, Hodson J, Smith JP, Douglas KM, Kitas GD (2015) Cumulative inflammation associates with asymmetric dimethylarginine in rheumatoid arthritis: a 6 year follow-up study. Rheumatology (Oxford) 54:1145–1152. https://doi.org/10.1093/rheumatology/keu349
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
None
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pagkopoulou, E., Soulaidopoulos, S., Katsiki, N. et al. The role of asymmetric dimethylarginine in endothelial dysfunction and abnormal nitric oxide metabolism in systemic sclerosis: results from a pilot study. Clin Rheumatol 42, 1077–1085 (2023). https://doi.org/10.1007/s10067-022-06472-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06472-w