Abstract
Objective
To investigate the frequency of glutathione S-transferase (GST), catalase, and SOD2 genetic polymorphisms and their correlation with SLE.
Methods
A total of 290 females (patients = 151; controls= 139) were recruited. Multiplex PCR was performed for genotyping GSTM1 and GSTT1 genes, whereas real-time qPCR was used for determination of SNPs: CAT C262T, SOD2 C47T, GSTP1 A313G and GSTP1 IVS6 -C16T.
Results
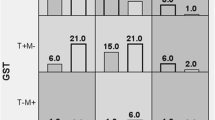
Thiol levels are decreased in SLE patients (p<0.001), while MDA levels were significantly higher (p<0.001) and those carrying the polymorphisms had higher rates of oxidative stress. Patients with double null deletion GSTT1null/GSTM1null had a frequency almost five times higher than the controls (p<0.001, OR 4.81, CI 1.98–12.11). SLE patients had a lower wild-type frequency of SOD2CC allele compared to controls (12.4% vs 27.3%). Statistical significances were observed on the association between the GSTT1null and GSTM1null with SOD2mut (p<0.001, OR 0.15, CI 0.05–0.47), with GSTP1 A303G (p=0.012, OR 0.19, CI 0.05–0.69), and with GSTP1 IVS6 (p=0.008, OR 0.14, CI 0.03–0.63). The same was observed between SOD2 C47T with GSTP1 A303G (p=0.09, OR 0.27, CI 0.09–0.74) and GSTP1 IVS6 (p=0.036, OR 0.41, CI 0.18–0.92).
Conclusions
The deletion GSTT1null/GSTM1null may contribute to the increased of the oxidative stress in SLE patients. Isolated GSTP1 and CAT polymorphisms do not seem to influence the increased oxidative stress, neither SLE clinical manifestations. SOD2 47CT/TT allele may have greater oxidative stress due to structural change in the protein and decreased H2O2 production. The combination of polymorphic genes may be involved in the pathogenesis of the disease.
Key points • Major question of our paper: Many studies have shown that the antioxidant status levels are decreased in patients with SLE, especially in severe stages of disease. We believe that this paper will be of interest to the readership of your journal had the involvement of polymorphisms and mutations in several genes that contribute to the genetic etiology of SLE, suggesting that these may influence the mechanisms of disease. • Our results. Thiol level was significantly (p<.001) lower and MDA level significantly increased (p<.001) among SLE patients. Those carrying the polymorphisms had higher rates of oxidative stress. SLE Patients had a frequency almost five times higher of double null deletion GSTT1null/GSTM1null than the controls. SLE Patients had a lower wild type frequency of SOD2CC allele compared to controls (12.4% vs 27.3%). We believed the deletion GSTT1null/GSTM1null may contribute to the increased of the oxidative stress in SLE patients while carriers of the mutant SOD2 47CT/TT allele may have greater oxidative stress due to structural change in the protein and decreased H2O2 production. The combination of polymorphic genes may be involved in the pathogenesis of the disease. • Implications of our results: Evidence for the involvement of genetic factors in severe clinical to lupus is compelling. This manuscript shows genetic insights in pathogenic pathways that may lead to severe clinical implications to LES. Therefore, it is necessary to understand their impact on overall disease pathogenesis and prognosis in these patients. We understand from general consensus about environmental factors can modify disease, however, maybe just in individuals who have a permissive genetic background. Even that no single gene predisposes some individuals to LES, we believe the genetic factors described in this manuscript are important elements in susceptibility to severe clinical to LES |



Similar content being viewed by others
References
Zhang Q, Ye DQ, Chen GP, Zheng Y (2010 Apr) Oxidative protein damage and antioxidant status in systemic lupus erythematosus. Clin Exp Dermatol 35(3):287–294. https://doi.org/10.1111/j.1365-2230.2009.03437.x
Perl A (2013 Nov) Oxidative stress in the pathology and treatment of systemic lupus erythematosus. Nat Rev Rheumatol 9(11):674–686. https://doi.org/10.1038/nrrheum.2013.147
Wang G, Pierangeli SS, Papalardo E, Ansari GA, Khan MF (2010 Jul) Markers of oxidative and nitrosative stress in systemic lupus erythematosus: correlation with disease activity. Arthritis Rheum 62(7):2064–2072. https://doi.org/10.1002/art.27442
Pérez YG, Pérez LCG, de Netto R CM, de Lima DSN, Lima ES (2012 Aug) Malondialdehyde and sulfhydryl groups as biomarkers of oxidative stress in patients with systemic lupus erythematosus. Rev Bras Reumatol 52(4):658–660
Sirivarasai J, Wananukul W, Kaojarern S, Chanprasertyothin S, Thongmung N, Ratanachaiwong W, Sura T, Sritara P (2013) Association between inflammatory marker, environmental lead exposure, and glutathione S-transferase gene. Biomed Res Int 2013:474963. https://doi.org/10.1155/2013/474963
Khodayari S, Salehi Z, Fakhrieh Asl S, et al (2013 May) Catalase gene C-262T polymorphism: importance in ulcerative colitis. J Gastroenterol Hepatol 28(5):819-822. https://doi.org/10.1111/jgh.12141
Bona N, Pezzarini E, Balbi B, Daniele SM, Rossi MF, Monje AL, Basiglio CL, Pelusa HF, Arriaga SMM (2020 Mar) Oxidative stress, inflammation and disease activity biomarkers in lupus nephropathy. Lupus 29(3):311–323. https://doi.org/10.1177/0961203320904784
Ye Z, Song H (2005 May) Glutathione s-transferase polymorphisms (GSTM1, GSTP1 and GSTT1) and the risk of acute leukaemia: a systematic review and metaanalysis. Eur J Cancer 41(7):980–989. https://doi.org/10.1016/j.ejca.2005.01.014
Thorn CF, Ji Y, Weinshilboum RM, Altman RB, Klein TE (2012 Aug) PharmGKB summary: very important pharmacogene information for GSTT1. Pharmacogenet Genomics 22(8):646–651. https://doi.org/10.1097/FPC.0b013e3283527c02
Mo Z, Gao Y, Cao Y, Gao F, Jian L (2009 May 1) An updating meta-analysis of the GSTM1, GSTT1, and GSTP1 polymorphisms and prostate cancer: a HuGE review. Prostate 69(6):662–688. https://doi.org/10.1002/pros.20907
Jia CY, Liu YJ, Cong XL, Ma YS, Sun R, Fu D, Lv ZW (2014 Apr) Association of glutathione S-transferase M1, T1, and P1 polymorphisms with renal cell carcinoma: evidence from 11 studies. Tumour Biol 35(4):3867–3873. https://doi.org/10.1007/s13277-013-1513-5
Pandey SN, Jain M, Nigam P, Choudhuri G, Mittal B (2006 May-Jun) Genetic polymorphisms in GSTM1, GSTT1, GSTP1, GSTM3 and the susceptibility to gallbladder cancer in North India. Biomarkers 11(3):250–261. https://doi.org/10.1080/13547500600648697
Cowell IG, Dixon KH, Pemble SE, Ketterer B, Taylor JB (1988) The structure of the human gltathione S-transferase pi gene. Biochem J 255(1):79–83. https://doi.org/10.1042/bj2550079
Mannervik B, Danielson UH (1988) Glutathione transferases--structure and catalytic activity. CRC Crit Rev Biochem 23(3):283–337. https://doi.org/10.3109/10409238809088226
Spurdle AB, Webb PM, Purdie DM, Chen X, Green A, Chenevix-Trench G (2001 Jan) Polymorphisms at the glutathione S-transferase GSTM1, GSTT1 and GSTP1 loci: risk of ovarian cancer by histological subtype. Carcinogenesis 22(1):67–72. https://doi.org/10.1093/carcin/22.1.67
Gulubova M, Vlaykova T (2010 Dec) Expression of the xenobiotic- and reactive oxygen species-detoxifying enzymes, GST-pi, Cu/Zn-SOD, and Mn-SOD in the endocrine cells of colorectal cancer. Int J Colorectal Dis 25(12):1397–1405. https://doi.org/10.1007/s00384-010-1041-3
Sánchez-Gómez FJ, Díez-Dacal B, García-Martín E, Agúndez JA, Pajares MA, Pérez-Sala D (2016) Detoxifying enzymes at the cross-roads of inflammation, oxidative stress, and drug hypersensitivity: role of glutathione transferase P1-1 and aldose reductase. Front Pharmacol 7:237. Published 2016 Aug 4 https://doi.org/10.3389/fphar.2016.00237
Moyer AM, Salavaggione OE, Wu TY, Moon I, Eckloff BW, Hildebrandt MA, Schaid DJ, Wieben ED, Weinshilboum RM (2008 Jun 15) Glutathione s-transferase p1: gene sequence variation and functional genomic studies. Cancer Res 68(12):4791–4801. https://doi.org/10.1158/0008-5472.CAN-07-6724
Crawford A, Fassett RG, Coombes JS, Kunde DA, Ahuja KD, Robertson IK, Ball MJ, Geraghty DP (2011 Sep) Glutathione peroxidase, superoxide dismutase and catalase genotypes and activities and the progression of chronic kidney disease. Nephrol Dial Transplant 26(9):2806–2813. https://doi.org/10.1093/ndt/gfq828
Xu Z, Zhu H, Luk JM, Wu D, Gu D, Gong W, Tan Y, Zhou J, Tang J, Zhang Z, Wang M, Chen J (2012 Nov 15) Clinical significance of SOD2 and GSTP1 gene polymorphisms in Chinese patients with gastric cancer. Cancer 118(22):5489–5496. https://doi.org/10.1002/cncr.27599
Crawford A, Fassett RG, Geraghty DP, Kunde DA, Ball MJ, Robertson IK, Coombes JS (2012 Jun 15) Relationships between single nucleotide polymorphisms of antioxidant enzymes and disease. Gene 501(2):89–103. https://doi.org/10.1016/j.gene.2012.04.011
dos Santos KG, Canani LH, Gross JL, Tschiedel B, Souto KE, Roisenberg I (2006) The catalase -262C/T promoter polymorphism and diabetic complications in Caucasians with type 2 diabetes. Dis Markers 22(5–6):355–359. https://doi.org/10.1155/2006/983408
Crispín JC, Hedrich CM, Tsokos GC (2013 Aug) Gene-function studies in systemic lupus erythematosus. Nat Rev Rheumatol 9(8):476–484. https://doi.org/10.1038/nrrheum.2013.78
Rhodes B, Vyse TJ (2008 Nov) The genetics of SLE: an update in the light of genome-wide association studies. Rheumatology (Oxford) 47(11):1603–1611. https://doi.org/10.1093/rheumatology/ken247
Lalwani P, de Souza GK, de Lima DS, Passos LF, Boechat AL, Lima ES. Serum thiols as a biomarker of disease activity in lupus nephritis. PLoS One. 2015 Mar 23;10(3):e0119947. https://doi.org/10.1371/journal.pone.0119947
Abd El Azeem RA, Zedan MM, Saad EA, Mutawi TM, Attia ZR (2021 Feb) Single-nucleotide polymorphisms (SNPs) of antioxidant enzymes SOD2 and GSTP1 genes and SLE risk and severity in an Egyptian pediatric population. Clin Biochem 88:37–42. https://doi.org/10.1016/j.clinbiochem.2020.11.010
Uribe AG, Vilá LM, McGwin G Jr, Sanchez ML, Reveille JD, Alarcón GS (2004 Oct) The systemic lupus activity measure-revised, the Mexican Systemic Lupus Erythematosus Disease Activity Index (SLEDAI), and a modified SLEDAI-2K are adequate instruments to measure disease activity in systemic lupus erythematosus. J Rheumatol 31(10):1934–1940
Weening JJ, D'Agati VD, Schwartz MM, Seshan SV, Alpers CE, Appel GB, Balow JE, Bruijn JA, Cook T, Ferrario F, Fogo AB, Ginzler EM, Hebert L, Hill G, Hill P, Jennette JC, Kong NC, Lesavre P, Lockshin M, Looi LM, Makino H, Moura LA, Nagata M, International Society of Nephrology Working Group on the Classification of Lupus Nephritis, Renal Pathology Society Working Group on the Classification of Lupus Nephritis (2004 Feb) The classification of glomerulonephritis in systemic lupus erythematosus revisited. Kidney Int 65(2):521–530. https://doi.org/10.1111/j.1523-1755.2004.00443.x Erratum in: Kidney Int. 2004 Mar;65(3):1132
Saloum de Neves Manta F, Pereira R, Vianna R, Rodolfo Beuttenmüller de Araújo A, Leite Góes Gitaí D, Aparecida da Silva D, de Vargas Wolfgramm E, da Mota Pontes I, Ivan Aguiar J, Ozório Moraes M, Fagundes de Carvalho E, Gusmão L (2013 Sep 20) Revisiting the genetic ancestry of Brazilians using autosomal AIM-Indels. PLoS One 8(9):e75145. https://doi.org/10.1371/journal.pone.0075145
Aycicek A, Erel O, Kocyigit A (2005 Dec) Decreased total antioxidant capacity and increased oxidative stress in passive smoker infants and their mothers. Pediatr Int 47(6):635–639. https://doi.org/10.1111/j.1442-200x.2005.02137.x
Harma M, Harma M, Erel O (2005 Feb) Oxidative stress in women with preeclampsia. Am J Obstet Gynecol 192(2):656–657. https://doi.org/10.1016/j.ajog.2004.07.094
Faul F, Erdfelder E, Buchner A, Lang AG (2009 Nov) Statistical power analyses using G*Power 3.1: tests for correlation and regression analyses. Behav Res Methods 41(4):1149–1160. https://doi.org/10.3758/BRM.41.4.1149
Goncalves M, Neto J, Souza C, Melo P, Reis M (2009) Evaluating glutathione S-Transferase (GST) null genotypes (GSTT1 and GSTM1) as a potential biomarker of predisposition for developing leukopenia. Int J Lab Hematol 32:e49–e56. https://doi.org/10.1111/j.1751-553X.2009.01169.x
Buttgereit F, da Silva JA, Boers M, Burmester GR, Cutolo M, Jacobs J et al (2002) Standardised nomenclature for glucocorticoid dosages and glucocorticoid treatment regimens: current questions and tentative answers in rheumatology. Ann Rheum Dis 61(8):718–722
dos Santos KSC, de Almeida MF, Maciel TKP, de Lima DSN (jan./dez. - 2010) Perfil Clínico e Laboratorial de Pacientes com Lúpus Eritematoso Sistêmico (LES). Revista HUGV - Revista do Hospital Universitário Getúlio Vargas 9:1–2
Systemic Lupus Erythematosus (SLE) (2005) Clinical and laboratory profile of patients followed at the Onofre Lopes University Hospital (UFRN - Natal/Brazil) and early organ damage in patients with recently diagnosed disease. Rev Bras Reumatol [online] 45(6):339–342. ISSN 1809-4570. https://doi.org/10.1590/S0482-50042005000600002
Liu K, Kurien BT, Zimmerman SL et al (2016) X chromosome dose and sex bias in autoimmune diseases: increased prevalence of 47, XXX in systemic lupus erythematosus and sjögren's syndrome. Arthritis Rheumatol 68(5):1290–1300. https://doi.org/10.1002/art.39560
Altindag O, Karakoc M, Kocyigit A, Celik H, Soran N (2007 Feb) Increased DNA damage and oxidative stress in patients with rheumatoid arthritis. Clin Biochem 40(3-4):167–171. https://doi.org/10.1016/j.clinbiochem.2006.10.006
Zhong S, Huang M, Yang X, Liang L, Wang Y, Romkes M, Duan W, Chan E, Zhou SF (2006 Oct) Relationship of glutathione S-transferase genotypes with side effects of pulsed cyclophosphamide therapy in patients with systemic lupus erythematosus. Br J Clin Pharmacol 62(4):457–472. https://doi.org/10.1111/j.1365-2125.2006.02690.x
Karaca S, Karaca M, Cesuroglu T, Erge S, Polimanti R (2015 May-Jun) GSTM1, GSTP1, and GSTT1 genetic variability in Turkish and worldwide populations. Am J Hum Biol 27(3):310–316. https://doi.org/10.1002/ajhb.22671
Gattás GJF, Kato M, Soares-Vieira JA, Siraque MS, Kohler P, Gomes L et al (2004) Ethnicity and glutathione S-transferase (GSTM1/GSTT1) polymorphisms in a Brazilian population. Braz J Med Biol Res [online] 37(4):451–458. ISSN 1414-431X. https://doi.org/10.1590/S0100-879X2004000400002
Hatagima A, Klautau-Guimarães MN, Da Silva FP, Cabello PH (Dec. 2000) Glutathione S-transferase M1 (GSTM1) polymorphism in two Brazilian populations. Genet. Mol. Biol (São Paulo) 23(4). https://doi.org/10.1590/S1415-47572000000400003
De Oliveira C, De Oliveira SF, Hatagima A, Ferreira LB, Grisolia CK, Klautau- MDN (2007) Glutathione S-Transferase M1 and T1 polymorphisms in Brazilian African descendants. Hum Biol 79(1):131–140. Project MUSE, https://doi.org/10.1353/hub.2007.0025
Klautau-guimarães MDN, Hiragi CDO, Ascenção RFD, Oliveira SF, Grisolia CK, Hatagima A, et al (Jan./Mar. 2005) Distribution of glutathione S-transferase GSTM1 and GSTT1 null phenotypes in Brazilian Amerindians. Genet Mol Biol (São Paulo) 28(1). https://doi.org/10.1590/S1415-47572005000100005
de Hiragi C O, Miranda-Vilela AL, Rocha DMS, de Oliveira SF, Hatagima A, de Klautau-Guimarães MN (2011) Superoxide dismutase, catalase, glutathione peroxidase and gluthatione s-transferases M1 and T1 gene polymorphisms in three brazilian population groups. Genet Mol Biol [online]. 34(1):11-18. ISSN 1415-4757. https://doi.org/10.1590/S1415-47572010005000102
Taufer M, Peres A, de Andrade VM, de Oliveira G, Sá G, do Canto ME, dos Santos AR, Bauer ME, da Cruz IB (2005 Apr) Is the Val16Ala manganese superoxide dismutase polymorphism associated with the aging process? J Gerontol A Biol Sci Med Sci 60(4):432–438. https://doi.org/10.1093/gerona/60.4.432.
Freedman JE (2008) Oxidative stress and platelets. Arterioscler Thromb Vasc Biol 28:s11–s16. https://doi.org/10.1161/ATVBAHA.107.159178
Yang M et al (2015) Hematocrit level could reflect inflammatory response and disease activity in patients with systemic lupus erythematosus. Clin Lab 61(7):801–807
Morgan PE, Sturgess AD, Davies MJ (2005 Jul) Increased levels of serum protein oxidation and correlation with disease activity in systemic lupus erythematosus. Arthritis Rheum 52(7):2069–2079. https://doi.org/10.1002/art.21130
Anderer G, Schrappe M, Brechlin AM, Lauten M, Muti P, Welte K, Stanulla M (2000 Nov) Polymorphisms within glutathione S-transferase genes and initial response to glucocorticoids in childhood acute lymphoblastic leukaemia. Pharmacogenetics 10(8):715–726. https://doi.org/10.1097/00008571-200011000-00006
Salimi S, Nakhaee A, Jafari M, Jahantigh D, Sandooghi M, Zakeri Z, Shahrakipour M, Naghavi A, Farajian-Mashhadi F (2015 Jun) Combination effect of GSTM1, GSTT1 and GSTP1 polymorphisms and risk of systemic lupus erythematosus. Iran J Public Health 44(6):814–821
Economopoulos KP, Sergentanis TN (2010 Jun) GSTM1, GSTT1, GSTP1, GSTA1 and colorectal cancer risk: a comprehensive meta-analysis. Eur J Cancer 46(9):1617–1631. https://doi.org/10.1016/j.ejca.2010.02.009
Tew MB, Reveille JD, Arnett FC, Friedman AW, McNearney T, Fischbach M, Ahn C, Tan FK (2001 Jun) Glutathione S-transferase genotypes in systemic sclerosis and their association with clinical manifestations in early disease. Genes Immun 2(4):236–238. https://doi.org/10.1038/sj.gene.6363756
Rupasree Y, Naushad SM, Rajasekhar L, Kutala VK (2013) Association of genetic variants of xenobiotic metabolic pathway with systemic lupus erythematosus. Indian J Biochem Biophys 50:447–452
Jufeng Z, Jingui D, David Z, Yanxin L, Li L, Qi W, Yong S, Jie Z, Hong Y, Bo Y, Jun W (2010) Association of GSTT1, GSTM1 and CYP1A1 polymorphisms with susceptibility to systemic lupus erythematosus in the Chinese population. Clin Chim Acta; Int J Clin Chem 411:878–881. https://doi.org/10.1016/j.cca.2010.03.007
Kang TY, El-Sohemy A, Comelis MC, Eny KM, Bae SC (2005) Glutathione S-transferase genotype and risk of systemic lupus erythematosus in Koreans. Lupus 14(5):381–384. https://doi.org/10.1191/0961203305lu2100oa
Fraser PA, Ding WZ, Mohseni M, Treadwell EL, Dooley MA, St Clair EW, Gilkeson GS, Cooper GS (2003) Glutathione S-transferase M null homozygosity and risk of systemic lupus erythematosus associated with sun exposure: a possible gene-environment interaction for autoimmunity. J Rheumatol
Sobkowiak A, Lianeri M, Wudarski M, Lacki JK, Jagodziński PP (2008 Jul) Manganese superoxide dismutase Ala-9Val mitochondrial targeting sequence polymorphism in systemic lupus erythematosus in Poland. Clin Rheumatol 27(7):827–831. https://doi.org/10.1007/s10067-007-0796-6
Glesse N, Rohr P, Monticielo OA, Rech TF, Brenol JC, Xavier RM, Kvitko K, Chies JA (2014 Sep) Genetic polymorphisms of glutathione S-transferases and cytochrome P450 enzymes as susceptibility factors to systemic lupus erythematosus in southern Brazilian patients. Mol Biol Rep 41(9):6167–6179. https://doi.org/10.1007/s11033-014-3496-8
Harris SE, Fox H, Wright AF, Hayward C, Starr JM, Whalley LJ, Deary IJ (2007 Jul 2) A genetic association analysis of cognitive ability and cognitive ageing using 325 markers for 109 genes associated with oxidative stress or cognition. BMC Genet 8:43. https://doi.org/10.1186/1471-2156-8-43
Melén E, Nyberg F, Lindgren CM, Berglind N, Zucchelli M, Nordling E, Hallberg J, Svartengren M, Morgenstern R, Kere J, Bellander T, Wickman M, Pershagen G (2008 Aug) Interactions between glutathione S-transferase P1, tumor necrosis factor, GSTP1and traffic-related air pollution for development of childhood allergic disease. Environ Health Perspect 116(8):1077–1084. https://doi.org/10.1289/ehp.11117
Joubert BR, Reif DM, Edwards SW, Leiner KA, Hudgens EE, Egeghy P, Gallagher JE, Hubal EC (2011 Feb 14) Evaluation of genetic susceptibility to childhood allergy and asthma in an African American urban population. BMC Med Genet 12:25. https://doi.org/10.1186/1471-2350-12-25
Levinsson A, Olin AC, Modig L, Dahgam S, Björck L, Rosengren A, Nyberg F (2014 Jun 10) Interaction effects of long-term air pollution exposure and variants in the GSTP1, GSTT1 and GSTCD genes on risk of acute myocardial infarction and hypertension: a case-control study. PLoS One 9(6):e99043. https://doi.org/10.1371/journal.pone.0099043
Ghaly MS, Ghattas MH, Labib SM (2012 Oct) Association of catalase gene polymorphisms with catalase activity and susceptibility to systemic lupus erythematosus in the Suez Canal area. Egypt Lupus 21(11):1244–1249. https://doi.org/10.1177/0961203312451505
Eny KM, El-Sohemy A, Cornelis MC, Sung YK, Bae SC (2005) Catalase and PPARgamma2 genotype and risk of systemic lupus erythematosus in Koreans. Lupus 14(5):351–355. https://doi.org/10.1191/0961203305lu2091oa
D'souza A, Kurien BT, Rodgers R, Shenoi J, Kurono S, Matsumoto H, Hensley K, Nath SK, Scofield RH (2008 Jul 7) Detection of catalase as a major protein target of the lipid peroxidation product 4-HNE and the lack of its genetic association as a risk factor in SLE. BMC Med Genet 9:62. https://doi.org/10.1186/1471-2350-9-62
Acknowledgments
The authors wish to thank all blood donors attending the FHEMOAM for contributing and participating in this study. Also, the authors thank the staff of the Molecular Biology Laboratory of the Federal University of Amazonas (UFAM) and Gonçalo Moniz Institute (IGM) (FIOCRUZ/BAHIA) for the technical support. Finally, the authors thank the Foundation for Research Support of the State of Amazonas (FAPEAM) for financial support. The sponsors of this study are public or nonprofit organizations that support science in general. They had no role in gathering, analyzing, or interpreting the data.
Funding
Financial support was provided by grants from the Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM)–Processo: 1094/2013-FAPEAM and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors alone are responsible for the content and writing of this article and report no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
de Oliveira, M.A.A., Mallmann, N.H., de Souza, G.K.B.B. et al. Glutathione S-transferase, catalase, and mitochondrial superoxide dismutase gene polymorphisms modulate redox potential in systemic lupus erythematosus patients from Manaus, Amazonas, Brazil. Clin Rheumatol 40, 3639–3649 (2021). https://doi.org/10.1007/s10067-021-05680-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-021-05680-0