Abstract
Objective
To identify the effects of Sirtuin 1 (Sirt1) on gouty arthritis and investigate the underlying mechanisms.
Methods
A gouty arthritis model was established by intra-articular injection of monosodium urate (MSU, 1 mg) crystal solution into the left foot pad of C57BL/6 mice. After pretreating the gouty arthritis mice with intra-articular injection of Sirt1 agonist (Resveratrol, RSV, 20 mg/kg) or peroxisome proliferator-activated receptor γ (PPARγ) inhibitor (T0070907, 1 mg/kg), the degree of joint inflammation of the gouty arthritis mice was evaluated by clinical integration of joint inflammation and hematoxylin and eosin (H&E) staining. The mRNA expression of Sirt1 and PPARγ were determined by real-time polymerase chain reaction (PCR). The expression profiling of inflammatory cytokines and chemokines in mouse joint tissues were determined by multi-factor assay kits. Peritoneal macrophages were isolated from mice and tested the effects of RSV and/or PPARγ on pro-inflammatory cytokines secretion by PCR.
Results
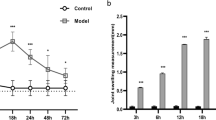
Sirt1 agonist significantly suppressed the onset of gouty arthritis induced by MSU and reduced the infiltration of inflammatory cells in the joints. Sirt1 agonist significantly promoted the expression of PPARγ, while decreased the expression of interleukin (IL)-1β, IL-1α, IL-6, interferon-γ (IFN-γ), monocyte chemotactic protein 1(MCP-1), tumor necrosis factor a (TNF-α), and chemokines (CXCL-1, CXCL-5, CCL-22) induced by MSU in joint tissues. After blocking PPARγ with T0070907 or by siRNA, the anti-inflammatory effect of Sirt1 agonist on gouty arthritis disappeared and the expression of pro-inflammatory molecules were not significantly reduced.
Conclusions
Sirt1 may control the acute onset of gouty arthritis in mice by inhibiting the infiltration of inflammatory cells and the secretion of pro-inflammatory molecules through PPARγ.
Key Points • Sirt1 and its activator, RSV, attenuate the severity of gouty arthritis in mice. • Sirt1 inhibits the infiltration of inflammatory cells and the secretion of pro-inflammatory molecules in MSU-induced arthritis. • Sirt1 inhibits inflammation partially dependent on PPARγ. |




Similar content being viewed by others
References
Schumacher HR (1996) Crystal-induced arthritis: an overview. Am J Med 100(2A):46S–52S
Di Giovine FS, Malawista SE, Nuki G, Duff GW (1987) Interleukin 1 (IL 1) as a mediator of crystal arthritis. Stimulation of T cell and synovial fibroblast mitogenesis by urate crystal-induced IL 1. J Immunol 138(10):3213–3218
Guerne PA, Terkeltaub R, Zuraw B, Lotz M (1989) Inflammatory microcrystals stimulate interleukin-6 production and secretion by human monocytes and synoviocytes. Arthritis Rheum 32(11):1443–1452
Matsukawa A, Yoshimura T, Maeda T, Takahashi T, Ohkawara S, Yoshinaga M (1998) Analysis of the cytokine network among tumor necrosis factor alpha, interleukin-1beta, interleukin-8, and interleukin-1 receptor antagonist in monosodium urate crystal-induced rabbit arthritis. Lab Investig 78(5):559–569
di Giovine FS, Malawista SE, Thornton E, Duff GW (1991) Urate crystals stimulate production of tumor necrosis factor alpha from human blood monocytes and synovial cells. Cytokine mRNA and protein kinetics, and cellular distribution. J Clin Invest 87(4):1375–1381. https://doi.org/10.1172/JCI115142
Wang J, Zhu XX, Liu L, Xue Y, Yang X, Zou HJ (2016) SIRT1 prevents hyperuricemia via the PGC-1alpha/PPARgamma-ABCG2 pathway. Endocrine 53(2):443–452. https://doi.org/10.1007/s12020-016-0896-7
Chen H, Zheng S, Wang Y, Zhu H, Liu Q, Xue Y, Qiu J, Zou H, Zhu X (2014) The effect of resveratrol on the recurrent attacks of gouty arthritis. Clin Rheumatol. https://doi.org/10.1007/s10067-014-2826-5
Borra MT, Smith BC, Denu JM (2005) Mechanism of human SIRT1 activation by resveratrol. J Biol Chem 280(17):17187–17195. https://doi.org/10.1074/jbc.M501250200
Knight JR, Milner J (2012) SIRT1, metabolism and cancer. Curr Opin Oncol 24(1):68–75. https://doi.org/10.1097/CCO.0b013e32834d813b
Pham J, Arul Nambi Rajan K, Li P, Parast MM (2018) The role of Sirtuin1-PPARgamma axis in placental development and function. J Mol Endocrinol 60(4):R201–R212. https://doi.org/10.1530/JME-17-0315
Engler A, Tange C, Frank-Bertoncelj M, Gay RE, Gay S, Ospelt C (2016) Regulation and function of SIRT1 in rheumatoid arthritis synovial fibroblasts. J Mol Med (Berl) 94(2):173–182. https://doi.org/10.1007/s00109-015-1332-9
Siebert A, Goren I, Pfeilschifter J, Frank S (2016) Anti-inflammatory effects of rosiglitazone in obesity-impaired wound healing depend on adipocyte differentiation. PLoS One 11(12):e0168562. https://doi.org/10.1371/journal.pone.0168562
Denko CW, Whitehouse MW (1976) Experimental inflammation induced by naturally occurring microcrystalline calcium salts. J Rheumatol 3(1):54–62
Martin WJ, Walton M, Harper J (2009) Resident macrophages initiating and driving inflammation in a monosodium urate monohydrate crystal-induced murine peritoneal model of acute gout. Arthritis Rheum 60(1):281–289. https://doi.org/10.1002/art.24185
Orlowsky EW, Stabler TV, Montell E, Verges J, Kraus VB (2014) Monosodium urate crystal induced macrophage inflammation is attenuated by chondroitin sulphate: pre-clinical model for gout prophylaxis? BMC Musculoskelet Disord 15:318. https://doi.org/10.1186/1471-2474-15-318
Yeung F, Hoberg JE, Ramsey CS, Keller MD, Jones DR, Frye RA, Mayo MW (2004) Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J 23(12):2369–2380. https://doi.org/10.1038/sj.emboj.7600244
Kauppinen A, Suuronen T, Ojala J, Kaarniranta K, Salminen A (2013) Antagonistic crosstalk between NF-kappaB and SIRT1 in the regulation of inflammation and metabolic disorders. Cell Signal 25(10):1939–1948. https://doi.org/10.1016/j.cellsig.2013.06.007
Akahoshi T, Namai R, Murakami Y, Watanabe M, Matsui T, Nishimura A, Kitasato H, Kameya T, Kondo H (2003) Rapid induction of peroxisome proliferator-activated receptor gamma expression in human monocytes by monosodium urate monohydrate crystals. Arthritis Rheum 48(1):231–239. https://doi.org/10.1002/art.10709
Calleri E, Pochetti G, Dossou KSS, Laghezza A, Montanari R, Capelli D, Prada E, Loiodice F, Massolini G, Bernier M, Moaddel R (2014) Resveratrol and its metabolites bind to PPARs. Chembiochem 15(8):1154–1160. https://doi.org/10.1002/cbic.201300754
Picard F, Kurtev M, Chung N, Topark-Ngarm A, Senawong T, Machado De Oliveira R, Leid M, McBurney MW, Guarente L (2004) Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-gamma. Nature 429(6993):771–776. https://doi.org/10.1038/nature02583
Qiang L, Wang L, Kon N, Zhao W, Lee S, Zhang Y, Rosenbaum M, Zhao Y, Gu W, Farmer SR, Accili D (2012) Brown remodeling of white adipose tissue by SirT1-dependent deacetylation of Ppargamma. Cell 150(3):620–632. https://doi.org/10.1016/j.cell.2012.06.027
Han L, Zhou R, Niu J, McNutt MA, Wang P, Tong T (2010) SIRT1 is regulated by a PPAR{gamma}-SIRT1 negative feedback loop associated with senescence. Nucleic Acids Res 38(21):7458–7471. https://doi.org/10.1093/nar/gkq609
Lee MF, Liou TH, Wang W, Pan WH, Lee WJ, Hsu CT, Wu SF, Chen HH (2013) Gender, body mass index, and PPARgamma polymorphism are good indicators in hyperuricemia prediction for Han Chinese. Genet Test Mol Biomarkers 17(1):40–46. https://doi.org/10.1089/gtmb.2012.0231
Ruiz-Miyazawa KW, Staurengo-Ferrari L, Pinho-Ribeiro FA, Fattori V, Zaninelli TH, Badaro-Garcia S, Borghi SM, Andrade KC, Clemente-Napimoga JT, Alves-Filho JC, Cunha TM, Fraceto LF, Cunha FQ, Napimoga MH, Casagrande R, Verri WA Jr (2018) 15d-PGJ2-loaded nanocapsules ameliorate experimental gout arthritis by reducing pain and inflammation in a PPAR-gamma-sensitive manner in mice. Sci Rep 8(1):13979. https://doi.org/10.1038/s41598-018-32334-0
Rossi A, Kapahi P, Natoli G, Takahashi T, Chen Y, Karin M, Santoro MG (2000) Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IkappaB kinase. Nature 403(6765):103–108. https://doi.org/10.1038/47520
Straus DS, Pascual G, Li M, Welch JS, Ricote M, Hsiang CH, Sengchanthalangsy LL, Ghosh G, Glass CK (2000) 15-deoxy-delta 12,14-prostaglandin J2 inhibits multiple steps in the NF-kappa B signaling pathway. Proc Natl Acad Sci U S A 97(9):4844–4849
Tani Y, Isobe Y, Imoto Y, Segi-Nishida E, Sugimoto Y, Arai H, Arita M (2014) Eosinophils control the resolution of inflammation and draining lymph node hypertrophy through the proresolving mediators and CXCL13 pathway in mice. FASEB J 28(9):4036–4043. https://doi.org/10.1096/fj.14-251132
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
All the animal procedures were approved by the ethics committee of the Animal Experiments Committee of Shanghai Jiao Tong University.
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Juan Wang and Guangliang Chen are co-first authors.
Rights and permissions
About this article
Cite this article
Wang, J., Chen, G., Lu, L. et al. Sirt1 inhibits gouty arthritis via activating PPARγ. Clin Rheumatol 38, 3235–3242 (2019). https://doi.org/10.1007/s10067-019-04697-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-019-04697-w