Abstract
Psoriatic arthritis (PsA) is a chronic condition characterized by a diverse set of symptoms, from swollen joints to nail disease to skin disease. A variety of treatment options are available, including tumor necrosis factor inhibitors (TNFis). Little is known about treatment persistence in patients with PsA who initiate TNFi therapy, with and without prior biologic use. This study assessed persistence in these subgroups of patients with PsA and identified factors associated with persistence. This retrospective study utilized data from the Corrona registry of patients with PsA—with or without prior biologic experience—who initiated TNFi therapy between October 1, 2002, and March 21, 2013. Kaplan-Meier curves estimated median time to nonpersistence (discontinuation or switch to another biologic). Cox proportional hazards models identified factors associated with TNFi nonpersistence. A total of 1241 TNFi initiations were identified: 549 by biologic-naïve and 692 by biologic-experienced patients. Through 4 years of follow-up, more biologic-naïve than biologic-experienced patients remained persistent. Biologic-naïve patients had a greater mean time to nonpersistence compared with biologic-experienced patients: 32 vs 23 months (p = 0.0002). Moderate and high disease activities based on clinical disease activity index and disease duration were associated with persistence in both biologic-naïve and biologic-experienced patients. Additionally, in the biologic-experienced patients, the number of prior medications and skin disease were associated with persistence. The majority of patients with PsA in this study were persistent with their TNFi therapy; biologic-naïve patients had greater persistence compared with biologic-experienced patients. Predictors of persistence differed slightly between biologic-naïve and biologic-experienced patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Psoriatic arthritis (PsA) is an increasingly common chronic systemic inflammatory disease that affects both the skin and the musculoskeletal system [1–3]. PsA can be characterized by pain and swelling of the joints, dactylitis, enthesitis, nail disease, and skin disease and increases the risk of cardiovascular disease and metabolic syndrome [4, 5]. The use of effective treatment improves patients’ functionality and quality of life by reducing skin and joint involvement and preventing permanent joint damage [6]. Treatment options for PsA include nonsteroidal antiinflammatory drugs (NSAIDs), nonbiologic disease-modifying antirheumatic drugs (nbDMARDs), biologic agents (including tumor necrosis factor inhibitors [TNFis]), and small molecules (including phosphodiesterase and Janus kinase inhibitors) [7]. From the rheumatoid arthritis (RA) literature, it has been demonstrated that use of these therapies is associated with a high rate of nonpersistence, attributed to dosing, route of administration, efficacy, and side effects.
Treatment persistence and prior biologic use are well documented in patients with RA and psoriasis; however, less is known about persistence in patients with PsA [8–14]. PsA is manifested in a diverse range of symptoms, and symptom severity varies over time, which complicates treatment and may influence treatment persistence [15, 16]. Factors associated with persistence differ based on prior biologic experience. The current study was undertaken to address this knowledge gap by describing persistence with TNFi in patients with PsA and to identify predictors of persistence, including the role of prior biologic use. This study provides a better understanding of the factors that influence discontinuation and switching of medications among patients with PsA with different biologic experience.
Materials and methods
Study design
The present study was a retrospective analysis of observational data collected on patients with PsA who participated in the Corrona registry and who initiated TNFi therapy.
Data source
Corrona is a large, US-based, independent, prospective, observational, disease-based registry with information on rheumatology patients, including those with PsA, since 2001. Patients are enrolled in the registry by participating rheumatologists, and questionnaires are completed by both patients and physicians as part of routine clinical care during clinical encounters. Data collection is not mandated at regular intervals; however, rheumatologists are encouraged to complete the questionnaires at least every 6 months. This registry encompasses over 160 sites across 40 states, including over 600 rheumatologists and 6000 patients with PsA (about 13% of the registry). This study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Study population
Patients from the Corrona registry were included in this study if they had a physician-recorded diagnosis of PsA; initiated therapy (i.e., the index date) with a TNFi (i.e., adalimumab, etanercept, golimumab, or infliximab) between October 1, 2002, and March 21, 2013; and were at least 18 years of age at the index date. Included patients also had ≥1 follow-up visit captured in the registry after initiation of TNFi therapy. Patients were excluded from this study if they had a diagnosis other than PsA for which any of the qualifying TNFis might be used (e.g., ankylosing spondylitis, Crohn’s disease, plaque psoriasis, RA, and ulcerative colitis).
Patients in this study were dichotomized as being persistent or nonpersistent. Patients were observed from the index date until the earliest instance of one of the following: last follow-up visit or nonpersistence (defined as discontinuation of TNFi therapy, switch to another biologic or small-molecule therapy, or a ≥1-year gap between follow-up visits).
Patients were further stratified by biologic experience. Biologic-naïve patients had no record of prior biologic use at TNFi initiation. Biologic-experienced patients had any record of prior biologic use at TNFi initiation.
Statistical analyses
Demographic and clinical characteristics were described at the index date. Kaplan-Meier curves were used to estimate median time to nonpersistence (i.e., discontinuation or switch to another biologic). Follow-up extended through the last visit for patients who were still on therapy at their last follow-up visit. Follow-up time was censored at the start of a gap in data if a patient had a gap ≥1 year between two visits before experiencing a discontinuation/switch event or last follow-up visit. A log-rank test was used to assess differences in persistence between patients who were biologic-naïve and biologic-experienced at the index date. Multivariate models were created to identify factors associated with TNFi nonpersistence with separate models for biologic-naïve and biologic-experienced patients. Univariate Cox proportional hazards models evaluated potential predictors: history of biologic use, patient demographics, history of comorbid conditions, and treatment history as of the index date; with the exception of gender, race, education, and treatment history as of the index date, all factors could vary over time. All factors that demonstrated a significant association with nonpersistence in the univariate models (p < 0.20) were then evaluated in multivariate Cox proportional hazards models, with the hazard ratios (HRs) from these two final models quantifying the effect of each factor as a predictor of nonpersistence for the biologic-naïve and biologic-experienced subpopulations.
Results
After applying the patient eligibility criteria, a total of 1241 TNFi initiations were identified for this study. These patients had a mean (standard deviation [SD]) age of 51 (12) years, 55% were female, 93% were white, and the study population had a mean (SD) PsA duration of 8 (9) years (Table 1). At the index date, mean (SD) clinical disease activity index (CDAI) was 14.6 (12), and the largest proportion of patients (44%) had CDAI ≤10 (i.e., low disease activity [LDA]; Table 2). The majority of patients had used a biologic (56%) or nonbiologic disease-modifying antirheumatic drug (nbDMARD) (86%) prior to initiating the index TNFi; 543 (44%) received monotherapy and 698 (56%) received combination therapy (Table 3). Tables 1, 2, and 3 also provide patient characteristics for biologic-naïve and biologic-experienced patients.
Time to TNFi nonpersistence
Overall, 46% of patients discontinued TNFi therapy during this study, with the number of persistent patients dropping from 83.2% at 6 months to 36.1% at 48 months. The follow-up time observed in this study was 1810.67 person-years. The median (95% confidence interval [CI]) time to discontinuation among all patients was 27 (25, 31) months. Among the 1241 TNFi initiations, there were 572 instances of nonpersistence, of which 309 instances were of switching.
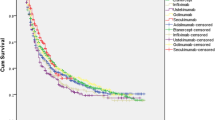
Biologic-naïve patients were consistently more persistent than their biologic-experienced counterparts at all time points assessed over 4 years of follow-up (Fig. 1). At 6 months, 87% of biologic-naïve and 80% of biologic-experienced patients were persistent; at 48 months, 42 and 31%, respectively, were persistent. The median (95% CI) time to discontinuation among biologic-experienced and biologic-naïve patients was significantly different: 23 (18, 29) vs 32 (27, 37) months (p = 0.0002 from log-rank test; Fig. 2).
Percentage of biologic-naïve and biologic-experienced patients with PsA who were persistent over time. The percentage (95% CI) of patients with PsA who were persistent at 6, 12, 24, 36, and 48 months in biologic-naïve (light gray bars) and biologic-experienced (dark gray bars) patients. CI confidence interval, PsA psoriatic arthritis, TNFi tumor necrosis factor inhibitor
Time to TNFi discontinuation in biologic-naïve and biologic-experienced patients. Time to discontinuation of TNFi in biologic-naïve (light gray curve) and biologic-experienced (dark gray curve) patients. Median (95% CI) time to discontinuation is indicated for each subgroup. CI confidence interval, TNFi tumor necrosis factor inhibitor
Predictors of TNFi nonpersistence
Multivariate Cox proportional hazard models were completed separately for biologic-naïve and biologic-experienced patients; models were adjusted for female gender, race, age, body mass index, smoking status, comorbidity, and concomitant prednisone use. In both biologic-naïve and biologic-experienced patients, activity based on the CDAI was associated with an increased risk of nonpersistence, although the magnitude of the association was greater in biologic-naïve patients: moderate CDAI (vs LDA) HR (95% CI) = 2.5 (1.5, 4.3) and 1.6 (1.1, 2.3) for biologic-naïve and biologic-experienced patients, respectively, and high CDAI (vs LDA) HR (95% CI) = 2.2 (1.1, 4.1) and 1.6 (1.0, 2.5) for biologic-naïve and biologic-experienced patients, respectively (Table 4). Shorter disease duration was also associated with better persistence in both biologic-naïve and biologic-experienced patients. In patients who were biologic-experienced, the risk of discontinuation was higher among patients with prior nbDMARD treatment compared with patients with no prior nbDMARD treatment; the risk of discontinuation was also greater among patients with greater skin involvement (i.e., a high [>20] vs low [≤20] score on the physician global skin assessment).
Discussion
Based on results from a nationwide cohort of patients with PsA, slightly more than half (52%) of the patients included in this study were persistent with their TNFi therapy at 24 months after initiation. Biologic-naïve patients had greater persistence compared with biologic-experienced patients. Increased disease activity was associated with a greater risk of nonpersistence in both groups. The magnitude of this association was greater in biologic-naïve patients compared with biologic-experienced patients.
Persistence is often associated with treatment efficacy [8–14]. The current study provides insight into the factors that influence persistence with a TNFi and how persistence differs based on prior exposure to biologics. Specifically, in patients who were biologic-naïve, disease duration and disease activity were associated with persistence. In addition to these factors, in patients who were biologic-experienced, prior medications and physician global skin assessment were also associated with persistence.
Most of the existing data on persistence and treatment outcomes describe patients with RA; there are fewer studies describing patients with PsA. Previous data indicate that patients with RA who fail their first biologic therapy experience worse efficacy on their second biologic [17–20]. Patients who are biologic-naïve experience better efficacy when they start a new biologic compared with patients who are biologic-experienced. The association between biologic experience and persistence is fairly well established in RA populations [5, 21, 22]; our study adds to the limited evidence suggesting that the pattern is similar in patients with PsA [23, 24]. Our overall rate of discontinuation was 46% including stopping, switching, or having a ≥1-year gap in treatment. In comparison, a study based on the DANBIO registry found that 39% of patients switched treatment and 17% of patients stopped treatment [24]. Of note, that study was focused on biologic-naïve patients only. Our findings are also consistent with another study that reported that, among patients who were biologic-naïve at TNFi initiation, there was no difference in persistence with TNFi between patients who initiated combination therapy (with methotrexate) and those who initiated monotherapy [25].
Physicians who are treating biologic-naïve patients may be more willing to switch them to a new drug when their disease activity increases because they have more remaining therapeutic options available to try. Such a scenario would be consistent with our findings. Similarly, physicians may be less willing to quickly switch biologic-experienced patients when their disease activity increases because those patients have fewer remaining therapeutic options available to try [26]. Both of these scenarios suggest careful consideration in choosing the initial biologic to increase the likelihood of a favorable outcome with the first drug used. Even with such careful consideration, some patients may experience increases in disease activity and may need to switch to a different TNFi. The findings from this study overall suggest the need for careful and continued monitoring to ensure that patients remain persistent in order to enhance their opportunity to obtain the greatest therapeutic benefit from any TNFi that they may be using. Monitoring may also help physicians intervene and switch patients to a different medication when necessary.
An important strength of this study is its use of data from a national cohort of patients receiving care in both academic and private practice settings. As such, this study provides insights into TNFi persistence in the real-world, routine care setting. Although one limitation associated with all real-world observational studies is that the patients enrolled in the registries may not be generalizable to patients in general practice, a recent study confirmed that Medicare patients represented in the Corrona registry are similar to the national Medicare population with regard to demographic characteristics, suggesting that data collected from patients in the Corrona registry may be generalizable to patients with RA in the USA [27]. As with most observational studies, there is the potential for selection bias in our study, especially given that the patients in our study entered the Corrona registry at the discretion of their treating rheumatologist. The PsA treatment landscape is evolving, with the availability of new agents possibly changing patterns of persistence. This issue was at least partially addressed by our decision to evaluate treatment-related covariates in a time-varying fashion for the models assessing predictors of persistence. Finally, we did not have access to data on reasons for TNFi discontinuation; consequently, the observed treatment nonpersistence could reflect either treatment success (i.e., achievement of LDA or remission) or failure (i.e., increased disease activity or worsening of symptoms).
Conclusion
The majority of patients with PsA assessed in this study were persistent with their TNFi therapy. When patients were stratified based on their previous biologic exposure, biologic-naïve patients had greater persistence compared with their biologic-experienced counterparts. While predictors of persistence differed slightly between biologic-naïve and biologic-experienced patients, disease activity was significant in both subgroups, although the magnitude of the association was a greater predictor for biologic-naïve compared with biologic-experienced patients. Understanding the factors associated with persistence in patients with PsA with different biologic experience may be important for making treatment decisions.
References
Gladman DD, Antoni C, Mease P et al (2005) Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis 64(Suppl 2):ii14–ii17
Lloyd P, Ryan C, Menter A (2012) Psoriatic arthritis: an update. Arthritis 2012:176298
Wilson FC, Icen M, Crowson CS et al (2009) Time trends in epidemiology and characteristics of psoriatic arthritis over 3 decades: a population-based study. J Rheumatol 36:361–367
Gottlieb A, Korman NJ, Gordon KB et al (2008) Guidelines of care for the management of psoriasis and psoriatic arthritis: section 2. Psoriatic arthritis: overview and guidelines of care for treatment with an emphasis on the biologics. J Am Acad Dermatol 58:851–864
Ogale S, Hitraya E, Henk HJ (2011) Patterns of biologic agent utilization among patients with rheumatoid arthritis: a retrospective cohort study. BMC Musculoskelet Disord 12:204
Ceponis A, Kavanaugh A (2010) Treatment of psoriatic arthritis with biological agents. Semin Cutan Med Surg 29:56–62
Gossec L, Smolen JS, Ramiro S et al (2016) European League Against Rheumatism (EULAR) recommendations for the management of psoriatic arthritis with pharmacological therapies: 2015 update. Ann Rheum Dis 75:499–510
Burudpakdee C, Khan ZM, Gala S et al (2015) Impact of patient programs on adherence and persistence in inflammatory and immunologic diseases: a meta-analysis. Patient Prefer Adherence 9:435–448
Contreras-Yanez I, Cabiedes J, Villa AR et al (2010a) Persistence on therapy is a major determinant of patient-, physician- and laboratory-reported outcomes in recent-onset rheumatoid arthritis patients. Clin Exp Rheumatol 28:748–751
Contreras-Yanez I, Pascual-Ramos V (2015) Window of opportunity to achieve major outcomes in early rheumatoid arthritis patients: how persistence with therapy matters. Arthritis Res Ther 17:177
Contreras-Yanez I, Ponce De Leon S, Cabiedes J et al (2010b) Inadequate therapy behavior is associated to disease flares in patients with rheumatoid arthritis who have achieved remission with disease-modifying antirheumatic drugs. Am J Med Sci 340:282–290
Fernandez-Nebro A, Irigoyen MV, Urena I et al (2007) Effectiveness, predictive response factors, and safety of anti-tumor necrosis factor (TNF) therapies in anti-TNF-naive rheumatoid arthritis. J Rheumatol 34:2334–2342
Kievit W, Fransen J, Adang EM et al (2011) Long-term effectiveness and safety of TNF-blocking agents in daily clinical practice: results from the Dutch Rheumatoid Arthritis Monitoring register. Rheumatology (Oxford) 50:196–203
Waimann CA, Marengo MF, de Achaval S et al (2013) Electronic monitoring of oral therapies in ethnically diverse and economically disadvantaged patients with rheumatoid arthritis: consequences of low adherence. Arthritis Rheum 65:1421–1429
Gelfand JM, Gladman DD, Mease PJ et al (2005) Epidemiology of psoriatic arthritis in the population of the United States. J Am Acad Dermatol 53:573
Liu JT, Yeh HM, Liu SY et al (2014) Psoriatic arthritis: epidemiology, diagnosis, and treatment. World J Orthop 5:537–543
Gomez-Reino JJ, Carmona L (2006) Switching TNF antagonists in patients with chronic arthritis: an observational study of 488 patients over a four-year period. Arthritis Res Ther 8:R29
Karlsson JA, Kristensen LE, Kapetanovic MC et al (2008) Treatment response to a second or third TNF-inhibitor in RA: results from the South Swedish Arthritis Treatment Group Register. Rheumatology (Oxford) 47:507–513
Navarro-Sarabia F, Ruiz-Montesinos D, Hernandez B et al (2009) DAS-28-based EULAR response and HAQ improvement in rheumatoid arthritis patients switching between TNF antagonists. BMC Musculoskelet Disord 10:91
Rendas-Baum R, Wallenstein GV, Koncz T et al (2011) Evaluating the efficacy of sequential biologic therapies for rheumatoid arthritis patients with an inadequate response to tumor necrosis factor-alpha inhibitors. Arthritis Res Ther 13:R25
Smolen JS, Aletaha D (2015) Rheumatoid arthritis therapy reappraisal: strategies, opportunities and challenges. Nat Rev Rheumatol 11:276–289
Tanaka Y, Kubo S, Yamanaka H et al (2014) Efficacy and safety of abatacept in routine care of patients with rheumatoid arthritis: Orencia® as biological intensive treatment for RA (ORBIT) study. Mod Rheumatol 24:754–762
Kavanaugh A, Mease PJ, Gomez-Reino JJ et al (2014) Treatment of psoriatic arthritis in a phase 3 randomised, placebo-controlled trial with apremilast, an oral phosphodiesterase 4 inhibitor. Ann Rheum Dis 73:1020–1026
Glintborg B, Ostergaard M, Krogh NS et al (2013) Clinical response, drug survival, and predictors thereof among 548 patients with psoriatic arthritis who switched tumor necrosis factor alpha inhibitor therapy: results from the Danish Nationwide DANBIO registry. Arthritis Rheum 65:1213–1223
Mease PJ, Collier DH, Saunders KC et al (2015) Comparative effectiveness of biologic monotherapy versus combination therapy for patients with psoriatic arthritis: results from the Corrona registry. RMD Open 1:e000181
Zhang J, Shan Y, Reed G et al (2011) Thresholds in disease activity for switching biologics in rheumatoid arthritis patients: experience from a large U.S. cohort. Arthritis Care Res (Hoboken) 63:1672–1679
Curtis JR, Chen L, Bharat A et al (2014) Linkage of a de-identified United States rheumatoid arthritis registry with administrative data to facilitate comparative effectiveness research. Arthritis Care Res (Hoboken) 66:1790–1798
Acknowledgments
Jessica Ma, Ph.D. of Amgen Inc. provided medical writing support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Institutional review board (IRB) approvals for this registry were obtained from local IRBs of participating academic sites and from a central IRB (New England IRB) for private practice sites. All patients were required to provide written informed consent prior to participation in the registry. This study has therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Conflicts of interest
LRH is an employee and shareholder of Corrona, LLC and an employee of the University of Massachusetts Medical School (UMMS), has a research grant through UMMS from Pfizer, and is a consultant for Roche; BSS and DHC are employees and shareholders of Amgen Inc.; SR is an employee of Corrona, LLC; AM is a former employee and shareholder of Amgen Inc.; SWW is a partner in Wade Outcomes Research and Consulting and a consultant for Amgen Inc.; WM is an employee of Corrona, LLC and Lamar University; JDG is an employee and shareholder of Corrona, LLC and consultant of Genentech, Eli Lilly, Janssen, Novartis, and Pfizer; and CJE is an employee of Corrona, LLC and advisory board member for Merck. All authors had full control of all primary data and agreed to allow the journal to review the data if requested. The data that support the findings of this study are available through Corrona, LLC, but restrictions apply to the availability of these data, which were used under a subscription agreement for the current study and so are not publicly available.
Funding
This study was sponsored by Corrona, LLC. The Corrona RA registry has been supported through contracted subscriptions in the last 2 years by AbbVie, Amgen, BMS, Crescendo, Genentech, Horizon Pharma USA, Janssen, Eli Lilly, Novartis, Pfizer, and UCB.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Harrold, L.R., Stolshek, B.S., Rebello, S. et al. Impact of prior biologic use on persistence of treatment in patients with psoriatic arthritis enrolled in the US Corrona registry. Clin Rheumatol 36, 895–901 (2017). https://doi.org/10.1007/s10067-017-3593-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-017-3593-x