Abstract
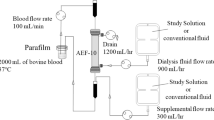
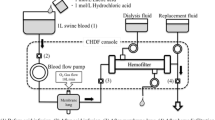
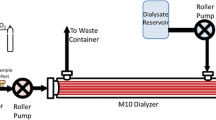
Administering nitrite has therapeutic effects on ischemic conditions wherein the enzymatic production of nitric oxide depends on oxygen. We developed a supplemental fluid containing nitric oxide (NO) and determined the clearance and supply between the pre- and post-dilution modes of continuous hemofiltration in vitro. Nitric oxide gas, 1000 mL or 2000 mL, at a concentration of 1000 ppm, was injected into 2020 mL of conventional supplemental fluid (experimental solution). The same volume of nitrogen gas was injected into the supplemental fluid (control solution). NO concentrations were measured using commercially available NO assay kit. Pre- or post-dilution continuous hemofiltration was performed using a control solution as supplemental fluid to determine the NO clearance. We determined the NO concentration of the outlet blood circuit to confirm the NO supply using the experimental solution as supplemental fluid. Also, using the bovine blood, white blood cell and platelet change rates and the dialysis membrane water flux during continuous hemodiafiltration were evaluated ex vivo as index of the biocompatibilities of a nitric oxide-containing solution. NO was not detected in the control solutions. The experimental solutions significantly increased in nitric oxide concentrations. NO clearance increased as the increase in supplemental and ultrafiltration flow rates using the control solution as supplemental fluid. However, using the experimental solution as supplemental fluid, nitric oxide supply showed a similar trend of NO clearance. Without any changes in biocompatibility using the supplemental fluid containing NO, it could maintain intravascular nitric oxide during continuous renal replacement therapy.



Similar content being viewed by others
References
Blood AB, Power GG. Nitrite: on the journey from toxin to therapy. Clin Pharmacokinet. 2015;54:221–3. https://doi.org/10.1007/s40262-014-0231-5.
Blood AB. The medicinal chemistry of nitrite as a source of nitric oxide signaling. Curr Top Med Chem. 2017;17:1758–68. https://doi.org/10.2174/1568026617666161116145046.
Calvert JW, Lefer DJ. Myocardial protection by nitrite. Cardiovasc Res. 2009;83:195–203. https://doi.org/10.1093/cvr/cvp079.
Dejam A, Hunter CJ, Schechter AN, Gladwin MT. Emerging role of nitrite in human biology. Blood Cells Mol Dis. 2004;32:423–9. https://doi.org/10.1016/j.bcmd.2004.02.002.
Bryan NS, Calvert JW, Elrod JW, Gundewar S, Ji SY, Lefer DJ. Dietary nitrite supplementation protects against myocardial ischemia-reperfusion injury. Proc Natl Acad Sci USA. 2007;104:19144–9. https://doi.org/10.1073/pnas.0706579104.
Lundberg JO, Gladwin MT, Ahluwalia A, Benjamin N, Bryan NS, Butler A, Cabrales P, Fago A, Feelisch M, Ford PC, Freeman BA, Frenneaux M, Friedman J, Kelm M, Kevil CG, Kim-Shapiro DB, Kozlov AV, Lancaster JR, Lefer DJ, McColl K, McCurry K, Patel RP, Petersson J, Rassaf T, Reutov VP, Richter-Addo GB, Schechter A, Shiva S, Tsuchiya K, van Faassen EE, Webb AJ, Zuckerbraun BS, Zweier JL, Weitzberg E. Nitrate and nitrite in biology, nutrition and therapeutics. Nat Chem Biol. 2009;5:865–9. https://doi.org/10.1038/nchembio.260.
Bryan NS, Torregrossa AC, Mian AI, Berkson DL, Westby CM, Moncrief JW. Acute effects of hemodialysis on nitrite and nitrate: potential cardiovascular implications in dialysis patients. Free Radic Biol Med. 2013;58:46–51. https://doi.org/10.1016/j.freeradbiomed.2013.01.020.
Heredia Martinez A, Rosa Diez G, Ferraris V, Coccia PA, Ferraris JR, Checa A, Wheelock CE, Lundberg JO, Weitzberg E, Carlström M, Krmar RT. Removal of nitrate and nitrite by hemodialysis in end-stage renal disease and by sustained low-efficiency dialysis in acute kidney injury. Nitric Oxide. 2020;98:33–40. https://doi.org/10.1016/j.niox.2020.02.004.
Chihara S, Masuda Y, Yamakage M. Experimental and clinical evaluation of predilution and postdilution continuous venovenous hemofiltration on clearance characteristics. ASAIO J. 2017;63:229–34. https://doi.org/10.1097/MAT.0000000000000468.
Tange Y, Yoshitake S, Watanabe W. Data on producing an infusion fluid that contains nitric oxide. Data Brief. 2020;28: 105011. https://doi.org/10.1016/j.dib.2019.105011.
Tange Y, Takesawa S, Yoshitake S. Asymmetric triacetate membrane keeps high water flux during ultrafiltration: in vitro study. J Artif Organs. 2017;20:399–402. https://doi.org/10.1007/s10047-017-0971-8.
Zuckerbraun BS, Shiva S, Ifedigbo E, Mathier MA, Mollen KP, Rao J, Bauer PM, Choi JJ, Curtis E, Choi AM, Gladwin MT. Nitrite potently inhibits hypoxic and inflammatory pulmonary arterial hypertension and smooth muscle proliferation via xanthine oxidoreductase-dependent nitric oxide generation. Circulation. 2010;121:98–109. https://doi.org/10.1161/CIRCULATIONAHA.109.891077.
Oliveira-Paula GH, Pinheiro LC, Tanus-Santos JE. Mechanisms impairing blood pressure responses to nitrite and nitrate. Nitric Oxide. 2019;85:35–43. https://doi.org/10.1016/j.niox.2019.01.015.
Parakaw T, Suknuntha K, Vivithanaporn P, Schlagenhauf A, Topanurak S, Fucharoen S, Pattanapanyasat K, Schechter A, Sibmooh N, Srihirun S. Platelet inhibition and increased phosphorylated vasodilator-stimulated phosphoprotein following sodium nitrite inhalation. Nitric Oxide. 2017;66:10–6. https://doi.org/10.1016/j.niox.2017.02.008.
Carlström M, Cananau C, Checa A, Wide K, Sartz L, Svensson A, Wheelock CE, Westphal S, Békássy Z, Bárány P, Lundberg JO, Hansson S, Weitzberg E, Krmar RT. Peritoneal dialysis impairs nitric oxide homeostasis and may predispose infants with low systolic blood pressure to cerebral ischemia. Nitric Oxide. 2016;58:1–9. https://doi.org/10.1016/j.niox.2016.05.005.
Carlström M, Wide K, Lundvall M, Cananau C, Svensson A, Lundberg JO, Bárány P, Krmar RT. Plasma nitrate/nitrite removal by peritoneal dialysis might predispose infants with low blood pressure to cerebral ischaemia. Clin Kidney J. 2015;8:215–8. https://doi.org/10.1093/ckj/sfv009.
Carlstrom M, Montenegro MF. Therapeutic value of stimulating the nitrate–nitrite–nitric oxide pathway to attenuate oxidative stress and restore nitric oxide bioavailability in cardiorenal disease. J Intern Med. 2019;285:2–18. https://doi.org/10.1111/joim.12818.
Brücken A, Bleilevens C, Berger P, Nolte K, Gaisa NT, Rossaint R, Marx G, Derwall M, Fries M. Effects of inhaled nitric oxide on outcome after prolonged cardiac arrest in mild therapeutic hypothermia treated rats. Sci Rep. 2018;8:6743. https://doi.org/10.1038/s41598-018-25213-1.
Morgan RW, Sutton RM, Karlsson M, Lautz AJ, Mavroudis CD, Landis WP, Lin Y, Jeong S, Craig N, Nadkarni VM, Kilbaugh TJ, Berg RA. Pulmonary vasodilator therapy in shock-associated cardiac arrest. Am J Respir Crit Care Med. 2018;197:905–12. https://doi.org/10.1164/rccm.201709-1818OC.
Lei C, Berra L, Rezoagli E, Yu B, Dong H, Yu S, Hou L, Chen M, Chen W, Wang H, Zheng Q, Shen J, Jin Z, Chen T, Zhao R, Christie E, Sabbisetti VS, Nordio F, Bonventre JV, Xiong L, Zapol WM. Nitric oxide decreases acute kidney injury and Stage 3 chronic kidney disease after cardiac surgery. Am J Respir Crit Care Med. 2018;198:1279–87. https://doi.org/10.1164/rccm.201710-2150OC.
Ho XL, Loke WM. Dietary plant sterols supplementation increases in vivo nitrite and nitrate production in healthy adults: a randomized, controlled study. J Food Sci. 2017;82:1750–6. https://doi.org/10.1111/1750-3841.13752.
Cantow K, Flemming B, Ladwig-Wiegard M, Persson PB, Seeliger E. Low dose nitrite improves reoxygenation following renal ischemia in rats. Sci Rep. 2017;7:14597. https://doi.org/10.1038/s41598-017-15058-5.
Münzel T, Daiber A. Inorganic nitrite and nitrate in cardiovascular therapy: a better alternative to organic nitrates as nitric oxide donors? Vascul Pharmacol. 2018;102:1–10. https://doi.org/10.1016/j.vph.2017.11.003.
Togo K, Yamamoto M, Ono T, Imai M, Akiyama K, Ebine K, Yamashita AC. Comparison of biocompatibility in polysulfone dialysis membranes with different sterilization. Hemodial Int. 2018;22:S10–4. https://doi.org/10.1111/hdi.12698.
Funding
This research has not received any specific grant from any funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
YT and SY contributed to the conception and design of the study, data analysis and interpretation, and drafting of the manuscript. WW and YT contributed to determining the sample concentrations. All authors are accountable for all aspects of the study; they worked on and approved the final version to be submitted.
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no proprietary or commercial interest for any product mentioned or any concept discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tange, Y., Watanabe, W. & Yoshitake, S. Nitric oxide delivery using nitric oxide-containing fluid in continuous hemofiltration: an in vitro study. J Artif Organs 25, 66–71 (2022). https://doi.org/10.1007/s10047-021-01284-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-021-01284-2