Abstract
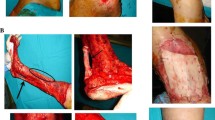
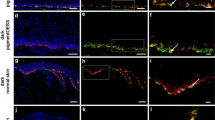
Giant congenital melanocytic nevi (GCMN) are defined as nevi greater than 20 cm in diameter. It is difficult to completely remove GCMN because of the lack of available skin grafts for covering the resultant defects. This study examined whether it is possible to produce reconstructed skin by combining epidermal and acellular dermal matrix (ADM) tissue derived from excised GCMN. GCMN skin samples were obtained with the informed consent of volunteer patients. The abilities of hypertonic saline (1 N NaCl), 0.05 % trypsin, 0.1 % SDS (sodium dodecyl sulfate), and phosphate buffered saline (PBS) to decellularize GCMN tissue were compared. The specimens were incubated in one of the test solutions at 37 °C for 48 h, before being washed with PBS at 4 °C for 14 days. Residual nuclei, residual DNA, nevus tissue viability, and the structural integrity of the basement membrane and capillaries were evaluated before treatment, and after 48 h’ treatment with or without 7 or 14 days’ washing. We tried to produce reconstructed skin by combining the resultant ADM with enzymatically separated GCMN epidermal tissue. The histological structure of the reconstructed skin was examined after it had been cultured for 5 days. In the SDS group, most cells had been removed after 48 h, and the DNA content of the ADM was significantly lower than in the other groups. As for viability, no significant difference was detected among the groups. The basement membrane and capillaries remained intact in all groups. After 5 days’ culturing, the epidermis had become attached to the ADM in all groups, except the SDS group. SDS displayed a superior decellularization ability compared with the other methods; however, it cannot be used to produce reconstructed skin because of its toxicity. In conclusion, we produced reconstructed skin that was devoid of nevus cells by combining GCMN epidermal tissue with GCMN-derived ADM produced with NaCl or trypsin. This is a promising treatment strategy for giant nevus.








Similar content being viewed by others
References
Arneja JS, Gosain AK. Giant congenital melanocytic nevi. Plast Reconstr Surg. 2009;124:1e–13e.
Mizuno H, Takeda A, Uchinuma E. Promising therapy for congenital giant pigmented nevi using acellular autograft nevi-dermal matrix. Ann Plast Surg. 1999;43:273–82.
Kopf AW, Bart RS, Hennessey P. Congenital nevocytic nevi and malignant melanomas. J Am Acad Dermatol. 1979;1:123–30.
Tannous ZS, Mihm MC Jr, Sober AJ, Duncan LM. Congenital melanocytic nevi: clinical and histopathologic features, risk of melanoma, and clinical management. J Am Acad Dermatol. 2005;52:197–203.
Wainwright DJ. Use of an acellular allograft dermal matrix (AlloDerm) in the management of full-thickness burns. Burns. 1995;21:243–8.
Livesey SA, Herndon DN, Hollyoak MA, Atkinson YH, Nag A. Transplanted acellular allograft dermal matrix. Potential as a template for the reconstruction of viable dermis. Transplantation. 1995;60:1–9.
Walter RJ, Matsuda T, Reyes HM, Walter JM, Hanumadass M. Characterization of acellular dermal matrices (ADMs) prepared by two different methods. Burns. 1998;24:104–13.
Takami Y, Matsuda T, Yoshitake M, Hanumadass M, Walter RJ. Dispase/detergent treated dermal matrix as a dermal substitute. Burns. 1996;22:182–90.
Takami Y, Matsuda T, Hanumadass M, Sakurai M, Water R. Acellular allogenic dermal matrix as a dermal substitute. Proc Int Conf Plastic Reconstruct Aesthet Surg. 1995;55:212–5.
Walter RJ, Jennings LJ, Matsuda T, Reyes HM, Hanumadass M. Dispase/Triton treated acellular dermal matrix as a dermal sustitute in rats. Curr Surg. 1997;54:371–4.
Chen RN, Ho HO, Tsai YT, Sheu MT. Process development of an acellular dermal matrix (ADM) for biomedical applications. Biomaterials. 2004;25:2679–86.
Funamoto S, Nam K, Kimura T, Murakoshi A, Hashimoto Y, Niwaya K, et al. The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials. 2010;31:3590–5.
Morimoto N, Takemoto S, Kanda N, Ayvazyan A, Taira MT, Suzuki S. The utilization of animal product-free media and autologous serum in an autologous dermal substitute culture. J Surg Res. 2011;171:339–46.
Qiagen. Protocol: Purification of total DNA from animal tissues (Spin-Column protocol). DNeasy blood and tissue handbook. 2006:28–30.
Kobayashi S, Kubo K, Matsui H, Torikai K, Kuroyanagi Y. Skin regeneration for giant pigmented nevus using autologous cultured dermal substitutes and epidermis separated from nevus skin. Ann Plast Surg. 2006;56:176–81.
Rompel R, Möser M, Petres J. Dermabrasion of congenital nevocellular nevi: experience in 215 patients. Dermatology. 1997;194:261–7.
Dave R, Mahaffey PJ. Combined early treatment of congenital melanocytic naevus with carbon dioxide and NdYag lasers. Br J Plast Surg. 2004;57:720–4.
Kono T, Ercocen AR, Kikuchi Y, Isago T, Honda T, Nozaki M. A giant melanocytic nevus treated with combined use of normal mode ruby laser and Q-switched alexandrite laser. J Dermatol. 2003;30:538–42.
Michel JL. Laser therapy of giant congenital melanocytic nevi. Eur J Dermatol. 2003;13:57–64.
Horner BM, El-Muttardi NS, Mayou BJ. Treatment of congenital melanocytic naevi with CO2 laser. Ann Plast Surg. 2005;55:276–80.
August PJ, Ferguson JE, Madan V. A study of the efficacy of carbon dioxide and pigment-specific lasers in the treatment of medium-sized congenital melanocytic naevi. Br J Dermatol. 2011;164:1037–42.
De Raeve LE, Roseeuw DI. Curettage of giant congenital melanocytic nevi in neonates: a decade later. Arch Dermatol. 2002;138:943–7.
Casanova D, Bardot J, Andrac-Meyer L, Magalon G. Early curettage of giant congenital naevi in children. Br J Dermatol. 1998;138:341–5.
Michel JL, Laborde-Milaa Roux V, Chavrier Y, Roux V, Metafiot H, Chalencon F, et al. Neonatal curettage of giant congenital nevi. Ann Dermatol Venereol. 2000;127:23–8.
Hopkins JD, Smith AW, Jackson IT. Adjunctive treatment of congenital pigmented nevi with phenol chemical peel. Plast Reconstr Surg. 2000;105:1–11.
Abai B, Thayer D, Glat PM. The use of a dermal regeneration template (Integra) for acute resurfacing and reconstruction of defects created by excision of giant hairy nevi. Plast Reconstr Surg. 2004;114:162–8.
Earle SA, Marshall DM. Management of giant congenital nevi with artificial skin substitutes in children. J Craniofac Surg. 2005;16:904–7.
Dantzer E, Braye FM. Reconstructive surgery using an artificial dermis (Integra): results with 39 grafts. Br J Plast Surg. 2001;54:659–64.
Moiemen N, Yarrow J, Hodgson E, Constantinides J, Chipp E, Oakley H, et al. Long-term clinical and histological analysis of Integra dermal regeneration template. Plast Reconstr Surg. 2011;127:1149–54.
Mastuda K, Suzuki S, Issiki N. Using two layers of artificial skin for giant pigmented nevi. J Jap Soc Plast Reconstr Surg. 1990;10:627–35.
Munster AM, Smith-Meek M, Shalom A. Acellular allograft dermal matrix: immediate or delayed epidermal coverage? Burns. 2001;27:150–3.
Eppley BL. Experimental assessment of the revascularization of acellular human dermis for soft-tissue augmentation. Plast Reconstr Surg. 2001;107:757–62.
Kishi K, Ninomiya R, Okabe K, Konno E, Katsube K, Imanishi N, et al. Treatment of giant congenital melanocytic nevi with enzymatically separated epidermal sheet grafting. J Plast Reconstr Aesthet Surg. 2010;63:914–20.
Woods T, Gratzer PF. Effectiveness of three extraction techniques in the development of a decellularized bone-anterior cruciate ligament-bone graft. Biomaterials. 2005;26:7339–49.
Rieder E, Kasimir MT, Silberhumer G, Seebacher G, Wolner E, Simon P, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004;127:399–405.
Voet D, Voet JG, Pratt CW. Fundamentals of biochemistry. New York: Wiley; 2002.
Gilbert TW, Sellaro TL, Badylak SF. Decellularization of tissues and organs. Biomaterials. 2006;27:3675–83.
Grauss RW, Hazekamp MG, Oppenhuizen F, van Munsteren CJ, Gittenberger-de Groot AC, DeRuiter MC. Histological evaluation of decellularised porcine aortic valves: matrix changes due to different decellularisation methods. Eur J Cardiothorac Surg. 2005;27:566–71.
Schenke-Layland K, Vasilevski O, Opitz F, König K, Riemann I, Halbhuber KJ, et al. Impact of decellularization of xenogeneic tissue on extracellular matrix integrity for tissue engineering of heart valves. J Struct Biol. 2003;143:201–8.
Dahl SL, Koh J, Prabhakar V, Niklason LE. Decellularized native and engineered arterial scaffolds for transplantation. Cell Transpl. 2003;12:659–66.
Goissis G, Suzigan S, Parreira DR, Maniglia JV, Braile DM, Raymundo S. Preparation and characterization of collagen-elastin matrices from blood vessels intended as small diameter vascular grafts. Artif Organs. 2000;24:217–23.
Martins-Green M, Bissel MF. Cell-extracellular matrix interactions in development. Semi Dev Biol. 1995;6:149–59.
Vyavahare N, Hirsch D, Lerner E, Baskin JZ, Schoen FJ, Bianco R, et al. Prevention of bioprosthetic heart valve calcification by ethanol preincubation Efficacy Mech Circ. 1997;95:479–88.
Gratzer PF, Harrison RD, Woods T. Matrix alteration and not residual sodium dodecyl sulfate cytotoxicity affects the cellular repopulation of a decellularized matrix. Tissue Eng. 2006;12:2975–83.
Wilson GJ, Courtman DW, Klement P, Lee JM, Yeger H. Acellular matrix: biomaterial approach for coronary artery bypass and heart valve replacement. Ann Thorac Surg. 1995;60:S353.
Cartmell JS, Dunn MG. Development of cell-seeded patellar tendon allografts for anterior cruciate ligament reconstruction. Tissue Eng. 2004;10:1065–75.
Acknowledgments
This study was supported by a Grant-in-Aid for Scientific Research.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Liem, P.H., Morimoto, N., Ito, R. et al. Autologous skin reconstruction by combining epidermis and acellular dermal matrix tissue derived from the skin of giant congenital melanocytic nevi. J Artif Organs 16, 332–342 (2013). https://doi.org/10.1007/s10047-013-0708-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-013-0708-2