Abstract
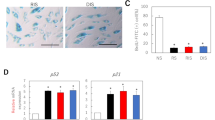
Fibroblasts cultured from venous ulcers demonstrate phenotypic characteristics of cellular senescence including slow growth, altered morphology, upregulation of fibronectin, and increased senescence-associated β-galactosidase activity. In senescent cells, arrest of cell replication is related to overexpression of p21 and underexpression of phosphorylated tumor-suppressor protein retinoblastoma (ppRb). The regulatory mechanisms for cell proliferation in venous ulcer fibroblasts are unknown. In this study, venous ulcer fibroblasts are examined for cell cycle protein expression and modulation by basic fibroblast growth factor (bFGF). Fibroblasts were isolated from the venous ulcer of the distal lower extremity (fb-D) of patients with chronic venous insufficiency. A control biopsy was obtained from the proximal ipsilateral thigh (fb-P). Paired cultures were plated at 100,000 cells/plate and the cells synchronized. After 24 hr, one culture set was treated with bFGF (20 ng/mL) and the other was kept in culture medium only (untreated). All cultures, treated and untreated, were lysed following 24 hr of incubation, and the lysate was used to perform immunoblot analysis for p21, ppRb, and cyclin D1. Immunoblot samples were standardized to protein content. In all patients analyzed (n = 4), at basal levels (untreated) fb-D demonstrated significant overexpression of p21 versus fb-P (p = 0.016). Treatment with bFGF resulted in significant downregulation of p21 levels for fb-D (p = 0.008) and fb-P (p = 0.037) compared to untreated fibroblasts. ppRb was underexpressed in fb-D versus fb-P (p = 0.069). Treatment with bFGF increased ppRb significantly in fb-D (p = 0.030) and in fb-P (p = 0.027) compared to untreated fibroblasts. No differences were observed in cyclin D1 with respect to basal levels in fb-P versus fb-D or in treated versus untreated groups. Venous ulcer fibroblasts show phenotypic similarity to senescent cells, with overexpression of p21 as well as down regulation of phosphorylated pRb. The aberrations seen in the cell cycle proteins in fb-D are similar to those seen in senescent cells; however, bFGF can modulate important cell cycle regulatory proteins, promoting a proliferative environment in fb-D that is not possible in a senescent cell. The role of bFGF may be useful in the clinical treatment of venous ulcer pathology.



Similar content being viewed by others
References
Callam MH, Harper D. Chronic ulcer of the leg: clinical history. Br J Med 1987;294:1389–1391
Pierce GF, Mustoe TA. Pharmacologic enhancement of wound healing. Annu Rev Med 1995;46:467–481
Nelzen O, Bergquist D, Lindhagen A. Venous and non-venous leg ulcers: clinical history and appearance in a population study: Br J Surg 1991;78:864–866
Stanley AC, Park HY, Phillips TJ, et al. Reduced growth of dermal fibroblasts from chronic venous ulcers can be stimulated with growth factors. J Vasc Surg 1997;26:994–1001
Mendez MV, Stanley AC, Park HY, et al. Fibroblasts cultured from venous ulcers display cellular characteristics of senescence: J Vasc Surg 1998;28:876–883
Campisi J. The biology of replicative senescence. Eur J Cancer 1997;33:703–709
Cristofalo VJ, Pignolo RJ. Molecular markers of senescence in fibroblast-like cultures. Exp Gerontol 1996;31:111–123
Dimri GP, Lee X, Basile G, et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 1995;92:9363–9367
Mendez MV, Stanley AC, Phillips TJ, et al. Fibroblasts cultured from distal lower extremities in patients with venous reflux display cellular characteristics of senescence. J Vasc Surg 1998;28:1040–1050
Stein GH, Dulic V. Molecular mechanisms for the senescent cell cycle arrest. J Inves Dermatol Symp Proc 1998;3:14–18
Stein GH, Beeson M, Gordon L. Failure to phosphorylate the retinoblastoma gene product in senescent human fibroblasts. Science 1990;249:666–669
Noda A, Ning Y, Venable SF, et al. Cloning of senescent cell-derived inhibitors of DNA synthesis using an expression screen. Exp Cell Res 1994;211:90–98
Sherr CJ, DePinho RA. Cellular senesence: mitotic clock or culture shock. Cell 2000;102:407–410
Cristofalo VJ, Phillips PD, Sorger T, et al. Alterations in the responsiveness of Senescent Cells to Growth Factors. J Gerentol 1989;44:55–62
Bergan JJ, Eklof B, Kistner RL, et al. Classification and grading of chronic venous disease in the lower limbs: a consensus statement. In: Gloviczki P, Yao JST, eds. Handbook of Venous Disorders: Guidelines of the American Venous Forum. New York: Chapman and Hall, 1996, pp 652–660
Simpson IA, Soanne O. A simple, rapid, and sensitive method for measuring protein concentration in subcellular membrane fractions prepared by sucrose density ultracentrifugation. Anal Biochem 1982;119:424–427
Raffeto JD, Mendez MV, Phillips TJ, et al. The effect of passage number on fibroblast cellular senescence in patients with chronic venous insufficiencey with and without ulcers. Am J Surg 1999;178:107–112
West MD, Pereira-Smith OM, Smith JR. Replicative senescence of human skin fibroblasts correlates with a loss of regulation and overexpression of collagenase activity. Exp Cell Res 1989;184:138–147
Seidman CS, Raffetto JD, Marien BJ, et al. bFGF induced alterations in cellular markers of senescence in growth rescued fibroblasts from chronic venous ulcer and venous reflux patients. Ann Vasc Surg 2003;17:239–44
Chen Q, Fischer A, Reagan JD, et al. Oxidative DNA damage and senescence of human diploid fibroblast cells. Proc Natl Acad Sci USA 1995;92:4337–4341
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Seidman, C., Raffetto, J.D., Overman, K.C. et al. Venous Ulcer Fibroblasts Respond to Basic Fibroblast Growth Factor at the Cell Cycle Protein Level. Ann Vasc Surg 20, 376–380 (2006). https://doi.org/10.1007/s10016-006-9036-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10016-006-9036-9