Abstract
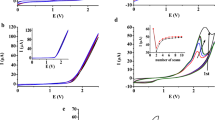
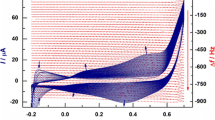
Methanol electrooxidation on smooth platinum electrodes bonded to solid polymer electrolytes was studied in water and acid solution by voltammetric measurements with different scanning rates. An enhancement of the oxidation rates was observed in these systems as compared to identical platinum electrodes in contact with liquid electrolytes. This electrocatalytic effect strongly depends on the measuring conditions and on the electrode potential. The reasons for the catalytic effects at different potentials are discussed.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 8 January 1997 / Accepted: 1 December 1997
Rights and permissions
About this article
Cite this article
Mayorova, N., Khazova, O. & Bagotzky, V. Methanol electrooxidation on platinum mesh electrodes bonded to solid polymer electrolytes. J Solid State Electrochem 2, 262–265 (1998). https://doi.org/10.1007/s100080050098
Issue Date:
DOI: https://doi.org/10.1007/s100080050098