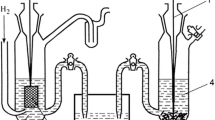
Abstract
Entropy is one of the less discussed and analyzed among the state functions studied in the thermodynamics of electrochemical systems. Few universities and post-graduate Electrochemistry courses and textbooks deal with this function’s basic aspects. Consequently, the importance of its practical applications needs to be thoroughly studied. Entropy changes are related to the reversible heat released or absorbed in electrochemical reactions. This reversible heat is also named molar electrochemical Peltier heat, which phenomenologically differs from the conventional Peltier heat used in physics and thermoelectric theory. In a simple and detailed manner, this manuscript presents fundamental concepts plus practical and experimental aspects for quantifying entropy changes and the associated molar electrochemical Peltier heat effects in a reversible and isothermal half-cell electrochemical reaction.
The differences between the conventional and molar electrochemical Peltier heat are also presented. Electrochemistry professors can use the material included here to encourage university and post-graduate students to further their knowledge and understanding of the electrochemical processes.





Similar content being viewed by others
References
Wedler G (1987) Lehrbuch der Physikalische Chemie, VCH Ed. Weinheim
Scholz F (2010) Thermodynamics of electrochemical reactions, Electroanalytical Methods, Berlin, Heidelberg: Springer Berlin Heidelberg. https://doi.org/10.1007/978-3-642-02915-8_2
Inzelt G (2015) Crossing the bridge between thermodynamics and electrochemistry. From the potential of the cell reaction to the electrode potential. ChemTexts. https://doi.org/10.1007/s40828-014-0002-9
Fang Z (2011) in Thermodynamics-physical chemistry of aqueous systems, edited by Moreno Pirajn, J. C. (InTech), Chap. 2. https://doi.org/10.5772/1434
Bouty M (1879) Sur un phénomène analogue au phénomène de Peltier. J Phys Theor Appl. https://doi.org/10.1051/jphystap:018790080034101
Schuster R (2017) Electrochemical microcalorimetry at single electrodes. Curr Opin Electrochem. https://doi.org/10.1016/j.coelec.2017.01.007
Barragan JA, Alemán JR, Peregrina-Lucano AA, Sánchez-Amaya M, Rivero P, Larios-Duran ER (2021) Leaching metals from e-waste: from its thermodynamic analysis and design to its implementation and optimization. ACS Omega. https://doi.org/10.1021/acsomega.1c00724
Bandhauer TM, Garimella S, Fuller TF (2011) A critical review of thermal issues in lithium-ion batteries. J Electrochem Soc doi 10(1149/1):3515880
Fang Z (2011) Some basic matters on the heat effects at electrode-electrolyte interfaces. Thermochim Acta. https://doi.org/10.1016/j.tca.2011.01.017
Vetter KJ (1967) Electrochemical kinetics: theoretical and experimental aspects. Academic Press, New York
Berry RS, Rice ST, Ross J (1980) Physical Chemistry. John Wiley & Sons, New York
Sun P, Kumar KR, Lyu M, Wang Z, Xiang J, Zhang W (2021) Generic Seebeck effect from spin entropy. The Innovation. https://doi.org/10.1016/j.xinn.2021.100101
Boudeville P (1994) Thermometric determination of electrochemical Peltier heat (thermal effect associated with electron transfer) of some redox couples. Inorganica Chim. Acta. https://doi.org/10.1016/0020-1693(94)04072-9
Agar JN (1963) in Advances in electrochemistry and electrochemical engineering, edited by Delahay P (Interscience), Vol. 5, Chap. 2. New-York
Lange E, Göhr H (1962) Thermodynamische elektrochemie. Alfred Hüthig Verlag, Heidelberg, Dr
Ozeki T, Watanabe I, Ikeda S (1983) Analysis of copper(I) ion in chloride solution with cyclic-voltammo-thermometry. J Electroanal Chem Interfacial Electrochem. https://doi.org/10.1016/S0022-0728(83)80031-X
Ozeki T, Ogawa N, Aikawa K, Watanabe I, Ikeda S (1983) Thermal analysis of electrochemical reactions: influence of electrolytes on Peltier heat for Cu(0)/Cu(II) and Ag(0)/Ag(I) redox systems. J Electroanal Chem Interfacial Electrochem. https://doi.org/10.1016/S0022-0728(83)80293-9
Boudeville P, Tallec A (1988) Electrochemistry and calorimetry coupling: IV. Determination of electrochemical peltier heat. Thermochim Acta. https://doi.org/10.1016/0040-6031(88)87268-X
Bard AJ, Inzelt G, Scholz F (2008) Electrochemical Dictionary. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-540-74598-3
Atkins P (1990) Physikalische Chemie, VCH Ed. Weinheim
Sanchez-Amaya M, Scholz F, Bárcena-Soto M, et al. (2022) Review—the thermistor-electrode as a temperature sensor to obtain thermodynamic data of electrochemical systems. ECS Advances. 10.1149/ 2754–2734/ac77a1
Bárcena-Soto M, Kubsch G, Scholz F (2002) Cyclic voltammetry of immobilized microparticles with in situ calorimetry: part I: the thermistor electrode. J Electroanal Chem. https://doi.org/10.1016/S0022-0728(02)00892-6
Forker W (1966) «Elektrochemische Kinetik,» Akademie-Verlag. Berlin
Ozeki T, Watanabe I, Ikeda S (1979) The application of the thermistor- electrode to Peltier heat measurement. J Electroanal Chem Interfacial Electrochem. https://doi.org/10.1016/S0022-0728(79)80308-3
Kuhn AT, Shams El Din AS (1983) Thermometric and calorimetric methods in electrochemical and corrosion studies. Surface Technology. https://doi.org/10.1016/0376-4583(83)90077-8
Sanchez-Amaya M, Bárcena-Soto M, Rodríguez-López A, Antaño-López R, Larios-Durán ER (2020) Sinusoidal temperature variation response associated with electrochemical Peltier heat as a transfer function approach. Electrochem Commun. https://doi.org/10.1016/J.ELECOM.2020.106769
Sánchez-Amaya M, Bárcena-Soto M, Antaño-López R, Rodríguez- López A, Gutiérrez-Becerra A, Larios-Durán ER (2021) Frequency responses of molar electrochemical Peltier heat and entropy changes analyzed as thermometric transfer functions. J Electrochem Soc. https://doi.org/10.1149/1945-7111/AC38F3
Hernández-Rizo SG, Larios-Durán ER, Bárcena-Soto M (2023) Frequency response of Gibbs free energy and enthalpy changes of electrochemical systems analyzed as thermometric transfer functions. J Solid State Electrochem. https://doi.org/10.1007/s10008-023-05553-3
Dean JA (1999) Lange’s Handbook of Chemistry, 15th edn. McGraw-Hill, New York
Fang Z, Wang S, Zhang Z, Qiu G (2008) The electrochemical Peltier heat of the standard hydrogen electrode reaction. Thermochim Acta. https://doi.org/10.1016/j.tca.2008.04.002
Acknowledgements
The authors thank CONAHCyT for the financial support to the project CF-2096004.
Funding
CONAHCyT supported this work through project CF-2096004.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Larios-Durán, E.R., Bárcena-Soto, M. Are the entropy changes important in an electrochemical process?. J Solid State Electrochem 28, 995–1006 (2024). https://doi.org/10.1007/s10008-023-05787-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-023-05787-1