Abstract
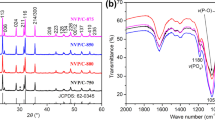
Nanocomposite materials, Na3V2(PO4)3/C and Na3V2(PO4)3/C/Ag, were synthesized by a modified Pechini method. Their properties were characterized with the use of the X-ray diffraction analysis, scanning electron microscopy, transmission electron microscopy, thermogravimetric analysis, elemental analysis, Raman spectroscopy, impedance spectroscopy, and charge-discharge tests as cathode materials for sodium-ion batteries. The discharge capacity of Na3V2(PO4)3/C obtained at 600 °C was 116.1 and 75 mAh g−1 at a current density of 11 (0.1 C) and 110 mA g−1 (1 С) in the potential range of 2.7–3.8 V. The high capacity values for fast charge/discharge were achieved as a result of heat treatment by two steps and incorporation of appropriate amount of silver particles into Na3V2(PO4)3/C nanocomposite. The discharge capacities of thus obtained Na3V2(PO4)3/C with 0.2 wt% of Ag were 117.2, 112.5, and 83.5 mAh g−1 at the current densities of 11, 110, and 880 mA g−1. This experimental evidence reveals the great potential of NVP/C/Ag synthesized by the modified Pechini method as cathode materials for the production of sodium-ion batteries.








Similar content being viewed by others
References
Palomares V, Serras P, Villaluenga I, Hueso KB, Carretero-González J, Rojo T (2012) Na-ion batteries, recent advances and present challenges to become low cost energy storage systems. Energy Environ Sci 5:5884–5901
Pan H, Hu YS, Chen L (2013) Room-temperature stationary sodium-ion batteries for large-scale electric energy storage. Energy Environ Sci 6:2338–2360
Slater MD, Kim D, Lee E, Johnson CS (2013) Sodium-ion batteries. Adv Funct Mater 23:947–958
Kim SW, Seo DH, Ma X, Ceder G, Kang K (2012) Electrode materials for rechargeable sodium-ion batteries: potential alternatives to current lithium-ion batteries. Adv. Energy Mater 2:710–721
Sudworth JL (1984) The sodium/sulphur battery. J Power Sources 11:143–154
Dustmann CH (2004) Advances in ZEBRA batteries. J Power Sources 127:85–92
Yabuuchi N, Kubota K, Dahbi M, Komaba S (2014) Research development on sodium-ion batteries. Chem Rev 114:11636–11682
Li H, Bai Y, Wu F, Li Y, Wu C (2015) Budding willow branches shaped Na3V2(PO4)3/C nanofibers synthesized via an electrospinning technique and used as cathode material for sodium ion batteries. J Power Sources 273:784–792
Lim SY, Kim H, Shakoor RA, Jung Y, Choi JW (2012) Electrochemical and thermal properties of NASICON structured Na3V2(PO4)3 as a sodium rechargeable battery cathode: a combined experimental and theoretical study. J Electrochem Soc 159:A1393–A1397
Song W, Ji X, Pan C, Zhu Y, Chen Q, Banks CE (2013) A Na3V2(PO4)3 cathode material for use in hybrid lithium ion batteries. Phys Chem Chem Phys 15:14357–14363
Song W, Ji X, Yao Y, Zhu H, Chen Q, Sun Q, Banks CE (2014) A promising Na3V2(PO4)3 cathode for use in the construction of high energy batteries. Phys Chem Chem Phys 16:3055–3061
Song W, Ji X, Wu Z, Zhu Y, Yang Y, Chen J, Jing M, Li F, Banks CE (2014) First exploration of Na-ion migration pathways in the NASICON structure Na3V2 (PO4)3. J Mater Chem A 2:5358–5362
Jung YH, Lim Ch H, Kim DK (2013) Graphene-supported Na3V2(PO4)3 as a high rate cathode material for sodium-ion batteries. J Mater Chem A 1:11350–11354
Goodenough JB, Hong HYP, Kafalas JA (1976) Fast Na+-ion transport in skeleton structures. Mater Res Bull 11:203–220
Saravanan K, Mason CW, Rudola A, Wong KH, Balaya P (2013) The first report on excellent cycling stability and superior rate capability of Na3V2 (PO4)3 for sodium ion batteries. Adv Energy Mater 3:444–450
Zhu X, Fang Y, Ai X, Yang H, Cao Y (2015) Na3V2 (PO4)3/C nanocomposite synthesized via pre-reduction process as high-performance cathode material for sodium-ion batteries. J Alloys Compd 646:170–174
Plashnitsa LS, Kobayashi E, Noguchi Y, Okada S (2010) Yamaki JI. Performance of NASICON Symmetric Cell with Ionic Liquid Electrolyte J Electrochem Soc 157:A536–A543
Si L, Yuan Zh HL, Zhu Y, Qian Y (2014) Uniform and continuous carbon coated sodium vanadium phosphate cathode materials for sodium-ion battery. J Power Sources 272:880–885
Zhu X, Fang Y, Ai X, Yang H, Cao Y (2015) Na3V2 (PO4)/C nanocomposite synthesized via pre-reduction process as high-performance cathode material for sodium-ion batteries. J Alloys Compd 646:170–174
Li S, Dong YF, Xu L, Xu X, He L, Mai LQ (2014) Batteries: effect of carbon matrix dimensions on the electrochemical properties of Na3V2 (PO4)3 nanograins for high-performance symmetric sodium-ion batteries. Adv Mater 26:3545–3553
Aragon MJ, Lavela P, Ortiz GF, Tirado JL (2015) Effect of iron substitution in the electrochemical performance of Na3V2(PO4)3 as cathode for Na-ion batteries. J Electrochem Soc 162:A3077–A3083
Liu J, Tang K, Song KP, Aken PAV, Yu Y, Maier J (2014) Electrospun Na3V2 (PO4)3/C nanofibers as stable cathode materials for sodium-ion batteries. Nanoscale 6:5081–5086
Li GQ, Jiang DL, Wang H, Lan XZ, Zhong HH, Jiang Y (2014) Glucose-assisted synthesis of Na3V2 (PO4)3/C composite as an electrode material for high-performance sodium-ion batteries. J Power Sources 265:325–334
Shen W, Wang C, Liu HM, Yang WS (2013) Towards highly stable storage of sodium ions: a porous Na3V2(PO4)3/C cathode material for sodium-ion batteries. Chem Eur J 19:14712–14718
Zhu CB, Song KP, Aken PAV, Yu Y, Maier J (2014) Carbon-coated Na3V2 (PO4)3 embedded in porous carbon matrix: an ultrafast Na-storage cathode with the potential of outperforming Li cathodes. Nano Lett 14:2175–2180
Chen M, Kou K, Tu M, Hu J, Yang B. g (2015) Fabrication of multi-walled carbon nanotubes modified Na3V2 (PO4)3/C and its application to high-rate lithium-ion batteries cathode Solid State Ionics 274:24–28
Chu Zh, Yue C (2016) Core–shell structured Na3V2 (PO4)3/C nanocrystals embedded in multi-walled carbon nanotubes: a high-performance cathode for sodium-ion batteries Solid State Ionics 287:36–41.
Wang KX, Li XH, Chen JS (2015) Surface and interface engineering of electrode materials for lithium-ion batteries. Adv Mater 27:527–545
Gryzlov D, Novikova S, Kulova T, Skundin A, Yaroslavtsev A (2016) Behavior of LiFePO4/CPVDF/Ag-based cathode materials obtained using polyvinylidene fluoride as the carbon source. Mater Des 104:95–101
Jian Z, Zhao L, Pan H, Hu YS, Li H, Chen W, Chen L (2012) Carbon coated Na3V2 (PO4)3 as novel electrode material for sodium ion batteries. Electrochem Commun 14:86–89
Wang H, Jiang D, Zhang Y, Li G, Lan X, Zhong H, Zhang Z, Jiang Y (2015) Self-combustion synthesis of Na3V2 (PO4)3 nanoparticles coated with carbon shell as cathode materials for sodium-ion batteries. Electrochim Acta 155:23–28
Zhang Y, Zhao H, Du Y (2016) Symmetric full cell assembled by self-supporting Na3V2 (PO4)3 bipolar electrodes for superior sodium energy storage. J Mater Chem A 4:7155–7159
Fang J, Wang S, Li Z, Chen H, Xia L, Ding L, Wang H (2016) Porous Na3V2 (PO4)3@C nanoparticles enwrapped in three-dimensional graphene for high performance sodium-ion batteries. J Mater Chem A 4:1180–1185
Tao S, Cui P, Huang W, Yu Z, Wang X, Liu D, Song L, Chu W, Wei S (2016) Sol-gel design strategy for embedded Na3V2 (PO4)3 particles into carbon matrices for high-performance sodium-ion batteries. Carbon 96:1028–1033
Wang D, Chen N, Li M, Wang C, Ehrenberg H, Bie X, Wei Y, Chen G, Du F (2015) Na3V2 (PO4)/C composite as the intercalation-type anode material for sodium-ion batteries with superior rate capability and long-cycle life. J Mater Chem A 3:8636–8642
Vidano RP, Fishbach DB (1981) Observation of Raman band shifting with excitation wavelength for carbons and graphites. Solid State Comm 39:341–344
Xie W, Zhu X, Yi S, Kuang J, Cheng H, Tang W, Deng Y (2016) Electromagnetic absorption properties of natural microcrystalline graphite. Mater Des 90:38–46
Wilcox JD, Doeff MM, Marcinek M, Kostecki R (2007) Factors influencing the quality of carbon coatings on LiFePO4. J Electrochem Soc 154:A389–A395
Bhuvaneswari MS, Bramnik NN, Ensling D, Ehrenberg H, Jaegermann W (2008) Synthesis and characterization of carbon nano fiber/LiFePO4 composites for Li-ion batteries. J Power Sources 180:553–560
Doeff MM, Hu Y, F. McLarnon, Kostecki R (2003) Effect of surface carbon structure on the electrochemical performance of LiFePO4. Electrochem Solid-State Lett 6:A207-A209
Bonhomme F, Lassegues JC, Servant L (2001) Raman spectroelectrochemistry of a carbon supercapacitor. J Electrochem Soc 148:E450–E458
Mariappan CR, Galven C, Crosnier-Lopez MP, Le Berre F, Bohnke O (2006) Synthesis of nanostructured LiTi2(PO4)3 powder by a Pechini-type polymerizable complex method. J Solid State Chem 179:450–456
Ejehi F, Marashi SPH, Ghaani MR, Haghshenas DF (2012) The synthesis of NaSICON-type ZrNb (PO4)3 structure by the use of Pechini method. Ceram Int 38:6857–6863
Il’in AB, Novikova SA, Sukhanov MV, Ermilova MM, Orekhova NV, Yaroslavtsev AB (2012) Catalytic activity of NASICON-type phosphates for ethanol dehydration and dehydrogenation. Inorg Mater 48:397–401
Vujković M, Stojković I, Cvjetićanin N, Mentus S (2013) Gel-combustion synthesis of LiFePO4/C composite with improved capacity retention in aerated aqueous electrolyte solution. Electrochim Acta 92:248–256
Li P, Shao L, Wang P, Zheng X, Yu H, Qian Sh, Shui M, Long N, Shu J (2015) Lithium sodium vanadium phosphate and its phase transition as cathode material for lithium ion batteries. Electrochim Acta 180:120–128
Acknowledgments
This work was financially supported by the Russian Foundation for Basic Research and Moscow Government (project no. 15-38-70042). In this work, we used the equipment of the Joint Research Center of IGIC RAS.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOC 76 kb)
Rights and permissions
About this article
Cite this article
Chekannikov, A., Kapaev, R., Novikova, S. et al. Na3V2(PO4)3/C/Ag nanocomposite materials for Na-ion batteries obtained by the modified Pechini method. J Solid State Electrochem 21, 1615–1624 (2017). https://doi.org/10.1007/s10008-017-3524-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3524-4