Abstract
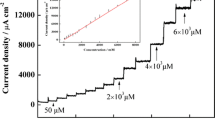
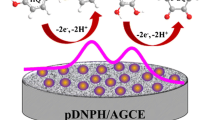
In an attempt to increase the stability and efficiency of hemin-modified electrodes, the present work reports the preparation of a new modified glassy carbon electrode obtained by immobilization of hemin (Hm) on the electrode surface together with a new N-substituted melamine (2,4,6-triamino-1,3,5-triazine) based G-2 dendrimer comprising p-aminophenol as peripheral unit (Den) or with one of its analogues, a melamine G-0 dimer (Dim). Basic structural features, able to determine intimate relationships between Hm and Dim (or Den) at room temperature in solid state, were evidenced with the use of vibrational analysis carried out by FT-IR. This method revealed contacts between Hm and Dim or Den respectively as H-bond interactions, proton-interchange, and π-π stacking interactions. The new modified electrodes were characterized by cyclic voltammetry and electrochemical impedance spectroscopy and tested for amperometric detection of H2O2. In this purpose, GC/Hm-Dim electrode exhibited better catalytic properties than GC/Hm-Den electrode, but lower stability.










Similar content being viewed by others
Notes
According to the definition from Ref.[25], rigid is “fixed in a single conformation either by geometric constraints or because of an overwhelmingly one-sided conformational equilibrium.”
References
Chen J, Zhao L, Bai H, Shi G (2011) J Electroanal Chem 657:34–38
Sosna MJ, Fapyane D, Ferapontova EE (2014) J Electroanal Chem 728:18–25
Brusova Z, Magner E (2009) Bioelechem 76(1–2):63–69
Bruice TC (1991) Acc Chem Res 24:243–249
Ni Y, Wang P, Song H, Lin X, Kokot S (2014) Anal Chim Acta 821:34–40
Santos RM, Rodrigues MS, Laranjinha J, Barbosa RM (2013) Biosens Bioelectron 44:152–159
Valentini F, Cristofanelli L, Carbone M, Palleschi G (2012) Electrochim Acta 63:37–46
Song H, Ni Y, Kokot S (2013) Anal Chim Acta 788:24–31
Wong A, Materon EM, Del Pilar Taboada Sotomayor M (2014) Electrochim Acta 146:830–837
Zeng F, Zimmerman SC (1997) Chem Rev 97:1681–1712
Balzani V, Ceroni P, Giansante C, Vicinelli V, Klarner FG, Verhaelen C, Vogtle F, Hahn U (2005) Angew Chem Int Ed 44:4574–4578
Newkome GR, Woosley BD, He E, Moorefield CN, Guther R, Baker GR, Escamilla GH, Merrill J, Luftmann H (1996) Chem Commun 24:2737–2738
Jockusch S, Turro NJ, Tomalia DA (1995) Macromolecules 28:7416–7418
Alonso B, Moran M, Casado CM, Lobete F, Losada J, Cuadrado I (1995) Chem Mater 7:1440–1442
Bustos Bustos E, Chapman TW, Rodriguez-Valadez F, Godinez LA (2006) Electroanal 18:2092–2098
Lates V, Gligor D, Darabantu M, Muresan LM (2007) J Appl Electrochem 37:631–636
Morar C, Turdean G, Bende A, Lameiras P, Antheaume C, Muresan LM, Darabantu M (2016) Manuscript under review
Weigend F, Ahlrichs R (2005) Phys Chem 7:3297–3305
Ghiviriga I, Oniciu DC (2002) Chem Commun 22:2718–2719
Drakenberg T, Forsen S (1971) Chem Commun 21:1404–1405
Mirvish SS, Gannett P, Babcook DM, Williamson D, Chen SC, Weisenburger DD (1991) J Agric Food Chem 39:1205–1210
Willner I, Rosengaus J, Eichen YJ (1993) Phys Org Chem 6:29–43
Katritzky AR, Ghiviriga I, Oniciu DC, Barkock A (1995) J Chem Soc Perkin Trans 2(4):785–792
Katritzky AR, Ghiviriga I, Steel PG, Oniciu DC (1996) J Chem Soc Perkin Trans 2(3):443–447
Eliel EL, Wilen SH (1994) Stereochemistry of the organic compounds. John Wiley & Sons, New York, pp 642–1191
Parker FS (1971) Biology and medicine: applications of infrared spectroscopy in biochemistry. Plenum Press, New York, p 351
Tom RT, Pradeep T (2005) Langmuir 21:11896–11902
Wood BR, Langford SJ, Cooke BM, Lim J, Glenister KK, Duriska M, Unthank JK, McNaughton D (2004) J Am Chem Soc 126:9233–9239
Hasinoff BB, Dunford HB, Horne DG (1969) Can J Chem 47:3225–3232
Wu DG, Cahen D, Graf P, Naaman R, Nitzan A, Shvarts D (2001) Chem Eur J 7:1743–1749
Schappacher M, Deffieux A (2004) Polymer 45:4633–4639
Wang Q, Yang Z, Zhang X, Xiao X, Chang CK, Xu B (2007) Angew Chem Int Ed 46:4285–4289
Luo F, Lin Y, Zheng L, Lin X, Chi Y (2015) Appl Mater Interfaces 7:11322–11329
Xie S, Ye J, Yuan Y, Chai Y, Yuan R (2015) Nanoscale 7:18232–18238
Hunter AA, Sanders JKM (1990) Chem Rew 112:5525–5534
Toader AM, Volanschi E, Lazarescu MF, Lazarescu V (2010) Electrochim Acta 56:863–866
Laviron E (1979) J Electroanal Chem 101:19–28
Ye JS, Wen Y, Zhang W, Cui HF, Gan LM, Xu GQ, Sheu FS (2004) J Electroanal Chem 562:241–246
Ma Q, Ai S, Yin H, Chen Q, Tang T (2010) Electrochim Acta 55:687–6694
Huang W, Hao Q, Lei W, Wu L, Xia X (2014) Mater Res Express. doi:10.1088/2053-1591/1/4/045601
Zuo G, Liu X, Yang J, Li X, Lu X (2007) J Electroanal Chem 605:81–88
Hirschorn B, Orazem ME, Tribollet B, Vivier V, Frateur I, Musiani M (2010) Electrochim Acta 55:6218–6227
Chen G, Sun H, Hou S (2016) Anal Biochem 502:43–49
Acknowledgments
The financial support from a grant provided by the Research Council Romania (Project PN-II-ID-PCE-2011-3-0128) is gratefully acknowledged. A.B. acknowledges the financial support from the Romanian National Authority for Scientific Research and Innovation (ANCSI) through the Core Program 2015.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deac, A.R., Morar, C., Turdean, G.L. et al. Glassy carbon electrode modified with hemin and new melamine compounds for H2O2 amperometric detection. J Solid State Electrochem 20, 3071–3081 (2016). https://doi.org/10.1007/s10008-016-3298-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-016-3298-0