Abstract
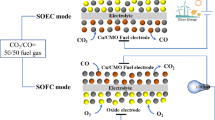
Fluorite-type heterogeneous catalyst ceria is a mixed conductor and widely used as a hydrocarbon-fueled solid oxide fuel cell anode because of its advantage of anti-carbon deposition, redox stability, and thermal compatibility. However, the electrocatalytic activity of a ceria cathode is limited for the catalysis of electrochemical oxidation or reduction reactions. In this work, catalytic-active iron and nickel catalysts are loaded onto a ceria cathode via an infiltration method to enhance electrode performance. Direct electrolysis of carbon dioxide is performed on ceria cathodes loaded with iron and nickel catalysts in solid oxide electrolyzers, respectively. The polarization resistance of symmetrical cells and electrolysis cells loaded with nickel and iron catalysts is largely improved in comparison with the bare ceria. The current efficiencies for carbon dioxide electrolysis for the iron- and nickel-loaded cathodes are 76 and 80 % at 2.0 V and 800 °C, respectively, approximately 25 % higher than that for the bare ceria cathode.









Similar content being viewed by others
References
Xie K, Zhang YQ, Meng GY, Irvine JTS (2011) Energy Environ Sci 4:2218–2222
Sista S, Hong Z, Chen L, Yang Y (2011) Energy Environ Sci 4:1606–1620
Hauch A, Ebbesen SD, Jensen SH, Mogensen M (2008) J Mater Chem 18:2331–2340
Bidrawn F, Kim G, Corre G, Irvine JTS, Vohs JM, Gorte RJ (2008) Electrochem Solid State Lett 11:B167–B170
Jensen SH, Larsen PH, Mogensen M (2007) Int J Hydrog Energy 32:3253–3257
Brisse A, Schefold J, Zahid M (2008) Int J Hydrog Energy 33:5375–5382
Zhan ZL, Zhao L (2010) J Power Sources 195:7250–7254
Ebbesen SD, Mogensen M (2009) J Power Sources 193:349–358
Hansen KV, Norrman K, Mogensen M (2004) J Electrochem Soc 151:A1436–A1444
Pihlatie M, Kaiser A, Mogensen M (2009) Solid State Ionics 180:1100–1112
Yang XD, Irvine JTS (2008) J Mater Chem 18:2349–2354
Wu GJ, Xie K, Wu YC, Yao WT, Zhou J (2013) J Power Sources 232:187–192
Alzate-Restrepo A, Hill JM (2010) J Power Sources 195:1344–1351
Weber A, Sauer B, Müller AC, Herbstritt D, Ivers-Tiffée E (2002) Solid State Ionics 152–153:543–550
Gan Y, Zhang J, Li SS, Xie K, Irvine JTS (2012) J Electrochem Soc 159:F763–F767
Tao SW, Irvine JTS (2003) Nat Mater 2:320–323
Tao SW, Irvine JTS, Kilner JA (2005) Adv Mater 17:1734–1737
Bastidas DM, Tao SW, Irvine JTS (2006) J Mater Chem 16:1603–1605
Xu SS, Li SS, Yao WT, Dong DH, Xie K (2003) J Power Sources 230:115–121
Tsekouras G, Neagu D, Irvine JTS (2013) Energy Environ Sci 6:256–266
Li YX, Gan Y, Li SS, Wang Y, Xiang HF, Xie K (2012) Phys Chem Chem Phys 14:15547–15553
Guzman J, Carrettin S, Corma A (2005) J Am Chem Soc 127:3286–3287
Chiang YM, Lavik EB, Kosacki I, Tuller HL, Ying JY (1996) Appl Phys Lett 69:185–187
Wang XQ, Rodriguez JA, Hanson JC, Gamarra D, Martinez-Arias A, Fernandez-Garcia M (2006) J Phys Chem B 110:428–434
Machida M, Uto M, Kurogi D, Kijima T (2000) Chem Mater 12:3158–3164
Chueh WC, Hao Y, Jung WC, Haile SM (2012) Nat Mater 11:155–161
Murray EP, Tsai T, Barnett SA (1999) Nature 400:649–651
Park S, Vohs JM, Gorte RJ (2000) Nature 404:265–267
Zhang XG, Robertson M, Deces-Petit C, Qu W, Kesler O, Maric R, Ghosh D (2007) J Power Sources 164:668–677
Zhao L, Shen JC, He BB, Chen FL, Xia CR (2011) Int J Hydrog Energy 36:3658–3665
Burroughs P, Hamnett A, Orchard AF, Thornton G (1976) J Chem Soc Dalton Trans 17:1686–1698
Francisco MSP, Mastelaro VR (2001) J Phys Chem B 105:10515–10522
Reddy BM, Khan A (2002) J Phys Chem B 106:10964–10972
Shah M, Barnett SA (2008) Solid State Ionics 179:2059–2064
Ruiz-Trejo E, Irvine JTS (2013) Solid State Ionics 252:157–164
Park JH, Blumenthal RN (1989) J Electrochem Soc 136:2867–2876
Schefold J, Brisse A, Zahid M (2009) J Electrochem Soc 156:B897–B904
Acknowledgments
This work was financially supported by the Natural Science Foundation of China (NSFC), No. 21303037, the China Postdoctoral Science Foundation, No. 2013 M53150, the Ministry of Education of Overseas Returnees Fund, No. 20131792, and the Fundamental Research Funds for the Central Universities, No. 2012HGZY0001.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 130 kb)
Rights and permissions
About this article
Cite this article
Qi, W., Xie, K., Qin, Q. et al. Efficient carbon dioxide electrolysis based on ceria cathode loaded with metal catalysts. J Solid State Electrochem 18, 3415–3425 (2014). https://doi.org/10.1007/s10008-014-2572-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-014-2572-2