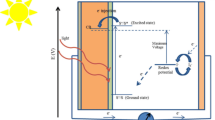
Abstract
In this work, we have synthesized Zinc oxide (ZnO) tripods and used its thin film as photoanode in dye-sensitized solar cells. SEM micrographs of the as-prepared sample of ZnO confirmed tripod-like morphology consisting of three cylindrical arms with well-defined ends, joined at a common core. The prepared sample of ZnO tripods was further characterized by EDX, XRD, UV-VIS, and FTIR. The dye N719-sensitized solar cell fabricated with photoanode of ZnO prepared in this work provided the open-circuit photo voltage (V oc) = 0.558 V, short-circuit photocurrent (J sc) = 6.368 mA cm-2, fill factor (FF) = 0.50, and total conversion efficiency (η) = 0.88 % under full light illumination (intensity 200 mW cm−2). When cell was illuminated by visible light (150 mW/cm2), V oc = 0.546 V, J sc = 4.437 mA/cm2, FF = 0.54, and η = 0.88 % were obtained.









Similar content being viewed by others
References
Tang ZK, Wong GKL, Yu P, Kawasaki M, Ohtomo A, Koinuma H, Segawa Y (1998) Room-temperature ultraviolet laser emission from self-assembled ZnO microcrystallite thin films. Appl Phys Lett 72:3270–3272
Ryu MK, Lee SH, Jang MS, Panin GN, Kang TN (2002) Postgrowth annealing effect on structural and optical properties of ZnO films grown on GaAs substrates by the radio frequency magnetron sputtering technique. J Appl Phys 92:154–159
Bahadur L, Srivastava P (2003) Efficient photon-to-electron conversion with rhodamine 6 G-sensitized nanocrystalline n-ZnO thin film electrodes in acetonitrile solution. Sol Energy Mater Sol Cell 79:235–248
Kushwaha S, Bahadur L (2011) Characterization of some metal-free organic dyes as photosensitizer for nanocrystalline ZnO-based dye sensitized solar cells. Int J Hydrogen Energy 36:1162–1167
Kushwaha S, Bahadur L (2011) Characterization of synthetic Ni(II)—xylenol complex as a photosensitizer for wide-band gap ZnO semiconductor electrodes. Int J Photoenergy. doi:10.1155/2011/980560
Bahadur L, Kushwaha S (2012) Highly efficient nanocrystalline ZnO thin films prepared by a novel method and their application in dye-sensitized solar cells. Appl Phys A: Mater Sci Process 109:655–663
Kushwaha S, Bahadur L (2012) Natural alkannin and anthocyanin as photosensitizers for dye-sensitized solar cells. Proc SCES (2012) Student conf, held at Allahabad, Uttar Pradesh, doi:10.1109/SCES.2012.6199084
Liu Y, Gorla CR, Liang S, Emanetoglu N, Lu Y, Shen H, Wraback M (2000) Ultraviolet detectors based on epitaxial ZnO films grown by MOCVD. J Electron Mater 29:69–74
Naseri N, Yousefi M, Moshfegh AZ (2011) A comparative study on photoelectrochemical activity of ZnO/TiO2 and TiO2/ZnO nanolayer systems under visible irradiation. Sol Energy 85:1972–1978
Lu L, Li R, Fan K, Peng T (2010) Effects of annealing conditions on the photoelectrochemical properties of dye-sensitized solar cells made with ZnO nanoparticles. Sol Energy 84:844–853
Chen H, Li W, Liu H, Zhu L (2010) A suitable deposition method of CdS for high performance CdS-sensitized ZnO electrodes: sequential chemical bath deposition. Sol Energy 84:1201–1207
Pawar RC, Shaikh JS, Babar AA, Dhere PM, Patil PS (2011) Aqueous chemical growth of ZnO disks, rods, spindles, and flowers: pH dependency and photoelectrochemical properties. Sol Energy 85:1119–1127
Hames Y, Alpaslan Z, Kösemen A, San SE, Yerli Y (2010) Electrochemically grown ZnO nanorods for hybrid solar cell applications. Sol Energy 84:426–431
Huang HM, Wu Y, Feick H, Tran N, Weber E, Yang P (2001) Catalytic growth of zinc oxide nanowires by vapor transport. Adv Mater 13:113–146
Choi JH, Tabata H, Kawai T (2001) Fabrication and optoelectronic properties of a transparent ZnO homostructural light-emitting diode. J Cryst Growth 226:493–500
Liu CH, Zapien JA, Yao Y, Meng XM, Lee CS, Lee FSS (2003) High-density, ordered ultraviolet light-emitting ZnO nanowire arrays. Adv Mater 15:838–841
Lyu SC, Zhang Y, Ruh H, Lee HJ, Shim HW, Suh EK, Lee CJ (2002) Low temperature growth and photoluminescence of well-aligned zinc oxide nanowires. Chem Phys Lett 363:134–138
Yao BD, Chen YF, Wang N (2002) Formation of ZnO nanostructures by a simple way of thermal evaporation. Appl Phys Lett 81:757–759
Wu JJ, Liu SC (2002) Catalyst-free growth and characterization of ZnO nanorods. J Phys Chem B 106:9546–9551
Zhang XT, Liu YC, Zhang LG, Zhang JY, Lu YM, Shen DZ, Xu W, Zhong GZ, Fan XW, Kong XG (2002) Structure and optically pumped lasing from nanocrystalline ZnO thin films prepared by thermal oxidation of ZnS thin films. J Appl Phys 92:3293–3298
Kim SW, Fujita S, Fujita S (2002) Self-organized ZnO quantum dots on SiO2/Si substrates by metalorganic chemical vapor deposition. Appl Phys Lett 81:5036–5065
Vayssieres L (2003) Growth of arrayed nanorods and nanowires of ZnO from aqueous solutions. Adv Mater 15:464–466
Hirano S, Kato K (1988) Formation of LiNbO3 films by hydrolysis of metal alkoxides. J Non-Cryst Solids 10:538–541
Arfsten NJ (1984) Sol-gel derived transparent IR-reflecting ITO semiconductor coatings and future applications. J Non-Cryst Solids 63:243–249
Hu JT, Odom TW, Lieber CM (1999) Chemistry and physics in one dimension: synthesis and properties of nanowires and nanotubes. Acc Chem Res 32:435–439
Xia Y, Yang P, Sun Y, Wu Y, Mayers B, Gates B, Yin Y, Kim F, Yan H (2003) One-dimensional nanostructures: synthesis, characterization, and applications. Adv Mater 15:353–389
Wu Y, Yang P (2000) Germanium nanowire growth via simple vapor transport. Chem Mater 12:605–607
Chen CC, Yeh CC (2000) Large-scale catalytic synthesis of crystalline gallium nitride nanowires. Adv Mater 12:738–741
Bai ZB, Yu DP, Zhang HZ, Ding Y, Gai XZ, Hang QL, Xiong GC, Feng SQ (1999) Nano-scale GeO2 wires synthesized by physical evaporation. Chem Phys Lett 303:311–314
Yazawa M, Koguchi M, Muto A, Ozawa M, Hiruma K (1992) Effect of one monolayer of surface gold atoms on the epitaxial growth of In as nanowhiskers. Appl Phys Lett 61:2051–2053
Choi YC, Kim WS, Park YS, Lee SM, Bae DJ, Lee YH, Park G-S, Choi WB, Lee NS, Kim JM (2000) Catalytic growth of β-Ga2O3 nanowires by arc discharge. Adv Mater 12:746–749
Duan XF, Leiber CM (2000) General synthesis of compound semiconductor nanowires. Adv Mater 12:298–302
Chen Z, Shan Z, Cao M, Lun L, Mao S (2004) ZnO nanotetrapods. Nanotechnology 15:365–367
Yan H, He R, Pham J, Yang P (2003) Morphogenesis of one-dimensional ZnO nano- and microcrystals. Adv Mater 15:402–405
Liu F, Cao PJ, Zhang HR, Li JQ, Gao HJ (2004) Controlled self-assembled nanoaeroplanes, nanocombs, and tetrapod-like networks of zinc oxide. Nanotechnology 15:949–952
Zhong Y, Djurisic AB, Hsu YF, Wong KS, Brauer G, Ling CC, Chan WK (2008) Exceptionally long exciton photoluminescence lifetime in ZnO tetrapods. J Phys Chem C 112:16286–16295
Chen W, Zhang H, Hsing IM, Yang S (2009) A new photoanode architecture of dye sensitized solar cell based on ZnO nanotetrapods with no need for calcinations. Electrochem Commun 11:1057–1060
Chen W, Yang S (2011) Dye-sensitized solar cells based on ZnO nanotetrapods. Front Optoelectron China 1:24–44
Liu Y, Chen Z, Kang Z, Bello I, Fan X, Shafiq I, Zhang W, Lee ST (2008) Self-catalytic synthesis of ZnO tetrapods, nanotetraspikes, and nanowires in air at atmospheric pressure. J Phys Chem C 112:9214–9218
Bahadur L, Rao TN (1995) Photoelectrochemical investigations on particulate ZnO thin film electrodes in non-aqueous solvents. J Photochem Photobiol A Chem 3:233–240
Acknowledgments
Financial support from the Ministry of New and Renewable Energy (MNRE, New Delhi, India) and Council of Scientific and Industrial Research (CSIR, New Delhi) is gratefully acknowledged. Thanks are also due to Indian Institute of Technology, Banaras Hindu University, Varanasi, India for providing their facilities (XRD and SEM) during this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahadur, L., Kushwaha, S. Structural and optical properties of tripod-like ZnO thin film and its application in dye-sensitized solar cell. J Solid State Electrochem 17, 2001–2008 (2013). https://doi.org/10.1007/s10008-013-2053-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-013-2053-z