Abstract
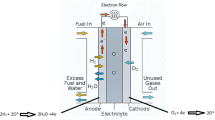
The effects of Co alloying to Pt catalyst and Nafion pretreatment by NaClO4 solution on the rate-determining step (RDS) of oxygen reduction at Nafion-impregnated Pt-dispersed carbon (Pt/C) electrode were investigated as a function of the potential step ΔE employing potentiostatic current transient (PCT) technique. For this purpose, the cathodic PCTs were measured on the pure Nafion-impregnated and partially Na+-doped Nafion-impregnated Pt/C and PtCo/C electrodes in an oxygen-saturated 1 M H2SO4 solution and analyzed. From the shape of the cathodic PCTs and the dependence of the instantaneous current on the value of ΔE, it was confirmed that oxygen reduction at the pure Nafion-impregnated electrodes is controlled by charge transfer at the electrode surface mixed with oxygen diffusion in the solution below the transition potential step |ΔE tr| in absolute value, whereas oxygen reduction is purely governed by oxygen diffusion above |ΔE tr|. On the other hand, the RDS of oxygen reduction at the partially Na+-doped Nafion-impregnated electrodes below |ΔE tr| is charge transfer coupled with proton migration, whereas above |ΔE tr|, it becomes proton migration in the Nafion electrolyte instead of oxygen diffusion. Consequently, it is expected in real fuel cell system that the cell performance is improved by Co alloying since the electrode reaches the maximum diffusion (migration) current even at small value of |ΔE|, whereas the cell performance is aggravated by Nafion pretreatment due to the decrease in the maximum diffusion (migration) current.









Similar content being viewed by others
References
De Souza A, Gonzalez ER (2003) J Solid State Electrochem 7:651. doi:10.1007/s10008-003-0363-2
Beard BC, Ross PN (1986) J Electrochem Soc 133:1839. doi:10.1149/1.2109033
Paffett MT, Berry GJ, Gottesfeld S (1988) J Electrochem Soc 135:1431. doi:10.1149/1.2096016
Beard BC, Ross PN (1990) J Electrochem Soc 137:3368. doi:10.1149/1.2086223
Mukerjee S, Srinivasan S (1993) J Electroanal Chem 357:201. doi:10.1016/0022-0728(93)80380-Z
Watanabe M, Tsurumi K, Mizukami T, Nakamura T, Stonehart P (1994) J Electrochem Soc 141:2659. doi:10.1149/1.2059162
Toda T, Igarashi H, Uchida H, Watanabe M (1999) J Electrochem Soc 146:3750. doi:10.1149/1.1392544
Min M, Cho J, Cho K, Kim H (2000) Electrochim Acta 45:4211. doi:10.1016/S0013-4686(00)00553-3
Antolini E, Passos RR, Ticianelli EA (2002) Electrochim Acta 48:263. doi:10.1016/S0013-4686(02)00644-8
Paulus UA, Scherer GG, Wokaun A, Schmidt TJ, Stamenkovic V, Radmilovic V, Markovic NM, Ross PN (2002) J Phys Chem B 106:4181. doi:10.1021/jp013442l
Antolini E, Salgado JRC, Giz MJ, Gonzalez ER (2005) Int J Hydrogen Energy 30:1213. doi:10.1016/j.ijhydene.2005.05.001
Remita H, Siril PF, Mbomekalle IM, Keita B, Nadjo L (2006) J Solid State Electrochem 10:506. doi:10.1007/s10008-005-0005-y
Li X, Colon-Mercado HR, Wu G, Lee JW, Popov BN (2007) Electrochem Solid-State Lett 10:B201. doi:10.1149/1.2777009
Koh S, Yu C, Mani P, Srivastava R, Strasser P (2007) J Power Sources 172:50. doi:10.1016/j.jpowsour.2007.01.002
Luna AMC, Bonesi A, Triaca WE, Baglio V, Antonucci V, Arico AS (2008) J Solid State Electrochem 12:643. doi:10.1007/s10008-007-0334-0
Lee MH, Wang PS, Do JS (2008) J Solid State Electrochem 12:879. doi:10.1007/s10008-007-0477-z
Gasteiger HA, Kocha SS, Sompalli B, Wagner FT (2005) Appl Catal B-Environ 56:9
O’Hayre RP, Cha SW, Colella W, Prinz FB (2006) Fuel Cell Fundamentals. Wiley, Hoboken, pp 112–121
Scharifker BR, Zelenay P, JO’M Bockris (1987) J Electrochem Soc 134:2714. doi:10.1149/1.2100276
Zecevic SK, Wainright JS, Litt MH, Gojkovic SL, Savinell RF (1997) J Electrochem Soc 144:2973. doi:10.1149/1.1837946
Eikerling M, Kornyshev AA (1999) J Electroanal Chem 475:107. doi:10.1016/S0022-0728(99)00335-6
Lee SJ, Pyun SI (2007) Electrochim Acta 52:6525. doi:10.1016/j.electacta.2007.04.081
Lee SK, Pyun SI, Lee SJ, Jung KN (2007) Electrochim Acta 53:740. doi:10.1016/j.electacta.2007.07.042
Lee JW, Pyun SI (2005) Electrochim Acta 50:1777. doi:10.1016/j.electacta.2004.08.046
Lee SJ (2008) Ph.D. Thesis, Chapter 3, Korea Advanced Institute of Science and Technology, Daejeon
Ayad A, Naimi Y, Bouet J, Fauvarque JF (2004) J Power Sources 130:50. doi:10.1016/j.jpowsour.2003.11.064
Biegler T, Rand DAJ, Woods R (1971) J Electroanal Chem 29:269. doi:10.1016/S0022-0728(71)80089-X
Hoare JP (1974) In: Bard AJ (ed) Encyclopedia of the Electrochemistry of the Elements, vol II. Marcel Dekker, New York, p 191
Tarasevich MR, Sadkovski A, Yeager E (1983) In: Conway BE, Bockris JO’M, Khan SVM, White RE (eds) Comprehensive Treatise of Electrochemistry, Vol. 7. Plenum, New York, p 301
Yaeger E (1984) Electrochim Acta 29:1527. doi:10.1016/0013-4686(84)85006-9
O’Sullivan EJM, Calvo EJ (1987) In: Compton RG (ed) Comprehensive Chemical Kinetics, vol 27. Elsevier, Amsterdam, p 247
Pletcher D, Sotiropoulos S (1993) J Electroanal Chem 356:109. doi:10.1016/0022-0728(93)80514-I
Floriano JB, Ticianelli EA, Gonzalez ER (1994) J Electroanal Chem 367:157. doi:10.1016/0022-0728(93)03007-C
Mello RMQ, Ticianelli EA (1997) Electrochim Acta 42:1031. doi:10.1016/S0013-4686(96)00282-4
Wen CJ, Boukamp BA, Huggins RA, Weppner W (1979) J Electrochem Soc 126:2258. doi:10.1149/1.2128939
Perez J, Gonzalez ER, Ticianelli EA (1998) Electrochim Acta 44:1329. doi:10.1016/S0013-4686(98)00255-2
Jiang J, Yi B (2005) J Electroanal Chem 577:107. doi:10.1016/j.jelechem.2004.11.022
Han JN, Seo M, Pyun SI (2001) J Electroanal Chem 499:152. doi:10.1016/S0022-0728(00)00506-4
Paulus UA, Wokaun A, Scherer GG, Schmidt TJ, Stamenkovic V, Markovic NM, Ross PN (2002) Electrochim Acta 47:3787. doi:10.1016/S0013-4686(02)00349-3
Bard AJ (2000) Electrochemical Methods, 2nd edn. Wiley, New York, p 163
Montella C (2002) J Electroanal Chem 518:61. doi:10.1016/S0022-0728(01)00691-X
Kim JS, Pyun SI, Lee JW, Song RH (2007) J Solid State Electrochem 11:117. doi:10.1007/s10008-005-0080-0
Kim JS, Pyun SI (2008) J Electrochem Soc 155:B8. doi:10.1149/1.2799084
Lee SJ, Pyun SI (2008) J Electrochem Soc 155:B1274. doi:10.1149/1.2987944
Lee JW, Pyun SI, Filipek S (2003) Electrochim Acta 48:1603. doi:10.1016/S0013-4686(03)00085-9
Lee SJ, Pyun SI, Lee JW (2005) Electrochim Acta 50:1121. doi:10.1016/j.electacta.2004.08.009
Acknowledgements
This study was supported by a grant from the Center for Advanced Materials Processing (CAMP) of the twenty-first century Frontier R&D Program funded by the Ministry of Commerce, Industry and Energy (MOCIE), Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SJ., Pyun, SI. Kinetics of mixed-controlled oxygen reduction at nafion-impregnated Pt-alloy-dispersed carbon electrode by analysis of cathodic current transients. J Solid State Electrochem 14, 775–786 (2010). https://doi.org/10.1007/s10008-009-0854-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-009-0854-x