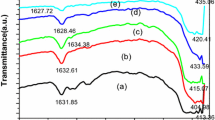
Abstract
Ruthenium (III) trichlorid solid crystals have been mechanically attached to gold surfaces and studied by cyclic electrochemical quartz crystal microbalance measurements in the presence of aqueous solutions of different concentrations containing M+Cl−, where M+=H+, Li+, Na+, K+, Rb+, Cs+. The RuCl3 and the complexes formed during the electrochemical transformations show two or more reduction and reoxidation pairs of waves, depending on the experimental conditions (concentration, scan rate, and potential range). The voltammetric peaks are shifted into the direction of higher potentials with increasing electrolyte concentrations except at very high concentrations when the peaks belong to the first reduction/reoxidation processes move oppositely. The mass change was reversible, during reduction mass increase, while during oxidation mass decrease occurred at medium electrolyte concentrations in two, more or less distinct steps. At high or low concentrations the mass excursions are more complex involving different mass increase/decrease regions as a function of potential which vary with the potential range of the measurements. The peak potentials and the electrochemical activity strongly depend on the nature of the cations and pH. It is related to the formation of complexes in different compositions. The mass change decreases with increasing electrolyte concentrations attesting the important role of the water activity and the transport of solvent molecules. It was concluded that in dilute solutions during the first reduction step M+ ions enter the surface layer. The strongly hydrated Li+ ions transfer water molecules into the microcrystals, while simultaneously with the incorporation of K+, Rb+, and Cs+ ions H2O molecules leave the surface layer. The opposite transport of ions and solvent molecules occur during oxidation. In the course of further reduction the incorporation of all ions studied except that of Cs+ ions is accompanied with water sorption. The number of sorbed water molecules is proportional to the hydration number of these ions. A reaction scheme is proposed in which M+ m-3[RuIIICl m (H2O) n ]3-m · xH2O (m≥3) and [RuIIICl m (H2O) n ]3-m (Cl−)3-m · xH2O (m≤3) type complexes are reduced to the respective – or depending on the electrolyte concentration higher or lower – Ru(II)chloro complexes resulting in mixed valence compounds (phases). Taking into account the layered structure of RuCl3 the electrochemical reduction can be explained as an intercalation reaction in that mixed valence intercalation phases with a general formula M x +(H2O) y [RuCl3]x− are formed from RuCl3·x H2O. The reduction/reoxidation waves are related to the redox transformations of Ru(III) to Ru(II) sites, while the composition of the polynuclear complexes and the structure of microcrystals change.














Similar content being viewed by others
References
Cotton FA, Wilkinson G, Murillo CA, Bochman M (1999) Advanced inorganic chemistry. Wiley, New York, pp 1010–1039
Livingstone SE (1973) In: Bailar JC, Emeléus MJ, Nyholm R, Trotman-Dickenson AF (eds) Comprehensive Inorganic Chemistry, vol 3. Pergamon Press, Oxford, pp 1163–1370
Chandret B, Sabo-Etienne S (1994) In: King RB (ed) Encyclopedia of inorganic chemistry, vol 7. Wiley, Chichester
Appelbaum L, Heinrichs C, Demtschuk J, Michman M, Oron M, Schäfer HJ, Schumann H, Organomet J (1999) Chem 592:240
Trasatti S (2000) Electrochim Acta 45:2377
Latimer WM (1952) The oxidation states of the elements and their potentials in aqueous solutions. Prentice-Hall, Englewood Cliffs, p 228
Llopis JF, Tordesillas IM (1976) In: Bard AJ (ed) Encyclopedia of electrochemistry, vol 6. Marcel Dekker, New York, p 277
Colom F (1985) In: Bard AJ, Parsons R, Jordan J (eds) Standard potentials in aqueous solution. Marcel Dekker, New York, p 413
De Benedetto GE, Guascito MR, Ciriello R, Cataldi TRI (2000) Anal Chim Acta 410:143
Kasem K, Steldt FR, Miller TJ, Zimmerman AN (2003) Microporous Mesoporous Mat 66:133
Chen SM, Hsueh SH (2004) J Electroanal Chem 566:291
Kulesza PJ (1987) J Electroanal Chem 220:295
Scholz F, Meyer B (1998) In: Bard AJ, Rubinstein I (eds) Electroanalytical Chemistry, vol 20. Marcel Dekker, New York, p 1
Grygar T, Marken F, Schröder U, Scholz F (2002) Coll Czech Chem Commun 67:163
Fiedler DA, Scholz F (2002) In: Scholz F (ed) Electroanalytical Methods Ch II 8. Springer, Berlin Heidelberg New York, pp 201–222
Bond AM, Marken F, Williams CT, Beattie DA, Keyes TE, Forster RJ, Vos JG (2000) J Phys Chem 104:1977
Ramaray R, Kabbe C, Scholz F (2000) Electrochem Commun 2:190
Inzelt G, Puskás Z (2004) Electrochem Commun 6:805
Pollini I (1994) Phys Rev B 50:4
Pollini I (1996) Phys Rev B 53:19
Mott NF (1961) Philos Mag 6:287
Wilson JA (1985) In: Edwards PP, Rao CN (eds) The Metallic and Nonmetallic States of Matter. Taylor and Francis, London, pp 215–260
Fehér K, Inzelt G (2002) Electrochim Acta 47:3551
Inzelt G (2003) J Solid State Electrochem 7:503
Inzelt G, Puskás Z (2004) Electrochim Acta 49:1969
Varga I, Bohlen A, von Klockenkämper R, Záray G (2000) Microchem J 67:265
Varga I, Rierpl E, Tusai A (1999) J Anal At Spectrom 14:881
Vittal L, Jayalakshim M, Gomathi H, Prahakara Rao G (1999) J Electrochem Soc 146:786
Schneemeyer LF, Spengler SE, Murphy DW (1985) Inorg Chem 24:3044
Bácskai J, Martinusz K, Czirók E, Inzelt G, Kulesza PJ, Malik MA (1995) 385:241
Retter U, Widmann A, Siegler K, Kahlert H (2003) J Electroanal Chem 546:87
Chen SM (2002) J Electroanal Chem 521:29
Pournaghi-Azar MH, Dastangoo H (2002) J Electroanal Chem 523:26
Engel D, Grabner EW (1985) Ber Bunsenges Phys Chem 89:982
Chen SM, Chan CM (2003) J Electroanal Chem 543:161
Cui X, Hong L, Lin X (2002) J Electroanal Chem 526:115
Malik MA, Horányi G, Kulesza PJ, Inzelt G, Kertész V, Schmidt R, Czirók E (1998) J Electroanal Chem 452:57
Schöllhorn R, Steffen R, Wagner K (1983) Angew Chem 95:559
Steffen R, Schöllhorn R (1986) Solid State Ionics 22:31
Evans CD, Chambers JQ (1994) Chem Mater 6:454
Hepel M, Janusz W (2000) Electrochim Acta 45:3785
Scholz F, Lovric M, Stojek Z (1997) J Solid State Electrochem 1:134
Suárez MF, Bond AM, Compton RG (1999) J Solid State Electrochem 4:24
Puskás Z, Inzelt G (2004) J Solid State Electrochem 8:828
Robinson RA, Stokes RH (1959) Electrolyte solutions. Butterworths, London, pp 491–504
Handbook of Chemistry and Physics (1977) Weast RC (ed) CRC Press, Cleveland, Ohio, p D-234
Taqui Khan MM, Ramachandraiah G, Prakash Rao A (1986) Inorg Chem 25:665
Trasatti S, Kurzweil P (1994) Platinum Met Rev 38:46
Wang JX, Marinkovic NS, Zajonz H, Ocko BM, Adzic RR (2001) J Phys Chem B 105:2809
Vericat C, Wakisaka M, Haasch R, Bagus PS, Wieckowski A (2004) J Solid State Electrochem 8:794
Wang L, Brazis P, Rocci M, Kannewurf CR, Kanatzidis MG (1998) Chem Mater 10:3298
Wang L, Rocci-Lane M, Brazis P, Kannewurf CR, Kim YI, Lee W, Choy JH, Kanatzidis MG (2000) J Am Chem Soc 122:6629
Levi MD, Aurbach D (1997) J Electroanal Chem 421:79
Levi MD, Levi EA, Aurbach D (1997) J Electroanal Chem 421:89
Levi MD, Aurbach D (1997) Electrochim Acta 45:167
Ohmori T, El-Deab MS, Osawa M (1999) J Electroanal Chem 470:46
Acknowledgements
Financial support by the National Scientific Research Fund (OTKA T046987) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Inzelt, G., Puskás, Z., Németh, K. et al. Electrochemically induced transformations of ruthenium(III) trichloride microcrystals in salt solutions. J Solid State Electrochem 9, 823–835 (2005). https://doi.org/10.1007/s10008-005-0019-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-005-0019-5