Abstract
Context
Polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs) are two families of persistent organic pollutants that are dangerous as they remain in the atmosphere for long periods and are toxic for humans and animals. They are found all over the world, including the penguins of Antarctica. One of the mechanisms that explains the toxicity of these compounds is related to oxidative stress. The main idea of this theoretical research is to use conceptual density functional theory as a theory of chemical reactivity to analyze the oxidative stress that PCBs and PBDEs can produce. The electron transfer properties as well as the interaction with DNA nitrogenous bases of nine PCBs and ten PBDEs found in Antarctic penguins are investigated. From this study, it can be concluded that compounds with more chlorine or bromine atoms are more oxidizing and produce more oxidative stress. These molecules also interact directly with the nitrogenous bases of DNA, forming hydrogen bonds, and this may be an explanation for the toxicity. Since quinone-type metabolites of PCBs and PBDEs can cause neurotoxicity, examples of quinones are also investigated. Condensed Fukui functions are included to analyze local reactivity. These results are important as the reactivity of these compounds helps to explain the toxicity of PCBs and PBDEs.
Methods
All DFT computations were performed using Gaussian16 at M06-2x/6–311 + g(2d,p) level of theory without symmetry constraints. Electro-donating (ω-) and electro-accepting (ω +) powers were used as global response functions and condensed Fukui functions as local parameters of reactivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Persistent organic pollutants are chemical compounds that remain intact in the environment for long periods [1]. They are widely distributed throughout the world, can accumulate in the fatty tissues of animals, and are toxic and dangerous to humans and wildlife. They are listed in the Stockholm Convention [2] which is a multilateral environmental agreement to “protect human health and the environment from these pollutants.” There are 152 countries that sign this agreement, and the idea is to work together in order to achieve a future free of persistent organic pollutants. In the meantime, it is necessary to know what we can expect from these compounds in relation to human and animal health.
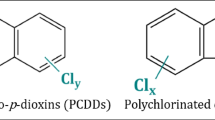
Polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs) are two families of persistent organic pollutants found in two penguin species in Antarctica [3]. PCBs have two fused benzene rings, with different chlorine atoms replacing the hydrogens. The number and position of chlorine atoms result in 209 different congeners [4]. PCBs have been used as flame retardant products and plasticizers [5]. PBDEs have two benzene rings connected by one oxygen atom. They are organo-brominated compounds in which different bromine atoms replace the hydrogens. The number and position of bromine atoms give rise to different congeners. They are widely used since the 1970s in paints, plastics, textiles, rugs, building materials, airplanes, and automobiles since the 1970s [6, 7]. Constitute up to 30% of these products by weight. Both families of compounds reach the environment and remain for a long time. Degradation takes more than 40 years [1]. Human contact comes from ingestion of contaminated food or air. Exposure to PCBs and PBDEs is associated with neurotoxicity, endocrine dysfunction, and reproductive disorders [8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35]. These compounds are chemically and enzymatically oxidized to active quinone-type derivatives that are also very toxic [36,37,38,39]. Quinones are Michael acceptors that can react with glutathione (GSH), proteins, and DNA [37, 38] inducing damage. They also have redox properties that increases oxidative stress [39].
Persistent organic pollutants studied in this investigation are those reported in Fig. 1. Within the PCBs, the most toxic are those with meta-chlorine and para-chlorine substituents [5]. Toxicological studies have shown that non-coplanar PCBs have high reactivity and toxicity [12] with PCB-77 being one of the most toxic [4]. Studies have reported that PCBs increase oxidative stress in the brain-producing apoptosis. This loss of neurons leads to various neurodegenerative diseases [10, 11, 16]. Among PBDEs, the congeners that induce oxidative DNA damage are PBDE-47 and PBDE-209. Within these, PDBE-47 is the most potent [13, 14]. Both compounds were reported to possibly damage DNA through more than one mechanism. Due to its proven toxicity, the production of PCBs and PBDEs has been prohibited for more than 5 years, but the problem is that those that have already been produced remain in the environment.
In Antarctica, Pygoscelis penguins are the most abundant vertebrates and are considered good bioindicators of different pollutants [3, 40,41,42,43]. In a previous work [3], the levels of PCBs and PBDEs were reported in samples from chinstrap penguin (Pygoscelis antarcticus) and gentoo penguin (Pygoscelis papua). The most abundant contaminants they found are those presented in Fig. 1. Although penguins are found in Antarctica, an area with a low population and far from civilization, we can find these contaminants. PCBs and PBDEs are not produced or used in this area, but they arrive through air and water. It is important to study these pollutants since they can apparently travel all over the world. Despite all the reported information on the effects on human and animal health produced of PCBs and PBDEs, there are no theoretical studies on the reactivity of these compounds and the oxidative stress that they can produce. There are theoretical investigations concerning the reactivity of PCB-77 [5, 44] that indicate the reaction of these compounds with ·OH during degradation and also regarding the interaction with nucleic DNA nitrogen bases. There are also reports that link oxidative stress to cancer and the development of brain disorders [45,46,47], but there are not a theoretical investigation concerning the charge transfer process of these compounds. For this reason, the main idea is to analyze the electron transfer process of PCBs and PBDEs. Examples of quinone-type metabolites are also included. Electron donor and acceptor properties of compounds shown in Fig. 1 and quinone-type derivatives of PCB-77 and PBDE-49 are investigated using density functional theory. The interaction of these compounds with DNA nitrogen bases is also analyzed. Quinones-type derivatives of PCB-77 and PBDE-49 were selected since the synthesis of these two quinones was previously reported [36,37,38]. The criterion for selecting these compounds is that they were found in Antarctic penguins and have been published as toxic. This research provides information on the reactivity of these compounds that may be useful to analyze their potential toxicity in animals and plants.
Computational details
Gaussian16 was used for all electronic calculations [48]. Geometry optimizations of initial geometries were obtained at M06-2x/6–311 + g(2d,p) level of theory without symmetry constraints [49,50,51]. These exchange correlation functionals were used before with success. This is a global hybrid functional with 54% HF exchange, and it is the best within the 06 functionals for main group thermochemistry, kinetics, and non-covalent interactions. To search for the global minimum structures, the structures published in PubChem were used as initial geometries. In some examples, we calculated different conformations, and the energy differences between optimized structures were very small. Furthermore, global reactivity descriptors were similar for each conformation. All optimized structures reported here are global minima and do not have negative harmonic frequencies.
Conceptual density functional theory is a chemical reactivity theory founded on density functional theory-based concepts [52,53,54,55,56,57,58]. Within this theory, there are global response functions such as the electro-donating (ω-) and electro-accepting (ω +) powers, previously reported by Gázquez and co-workers [53, 54]. The capacity to donate electrons (ω-) or accept electrons (ω +) is defined as follows:
I and A are vertical ionization energy and vertical electron affinity, respectively. They are obtained as follows:
Low values of ω- indicate good electron donor molecules. High values of ω + are for good electron acceptor molecules. With these parameters, it is possible to determine the electron donor–acceptor map (DAM, see Fig. 2) [59]. Systems located down to the left are considered good electron donors, while those situated up to the right are good electron acceptor. These chemical descriptors have been used successfully in different chemical systems [59,60,61,62,63,64,65,66].
Binding energies (BE) were obtained as follows:
X represents PCBs and PBDEs or their quinone derivatives, and Y represents guanine, deoxyadenosine, or GSH. Basis set superposition error (BSSE) was obtained according to the Counterpoise method of Boys and Bernardi [67, 68].
Condensed Fukui functions are calculated by the molecular fragment response approach using finite differences between the atomic charges of the systems [66,67,68]. The equations to calculate the condensed Fukui functions are the following:
Q are Mulliken charges of atom k. The larger the absolute value of f, the more reactive the atom will be.
Results and discussion
To investigate the potential oxidant capacity of PCBs and PBDEs, the analysis of the electron donor–acceptor properties was realized using the DAM for each system. Figures 3 and 4 present the corresponding DAMs. The first thing to note is that the electron acceptor capacity increases with the number of Cl or Br atoms. The best electron acceptors are those located up to the right, and they are molecules with seven Cl atoms or ten Br atoms. The best electron donors are those with three Cl or Br atoms. This is consistent with the electronegativity of Cl and Br. They are more electronegative than H or C, and therefore, the presence of these halogens improves the electron accepting capacity. PCB-77, one of the most toxic compounds, is found in the middle of the DAM. It is not the best electron donor, nor the worst, and the same can be said for its electron acceptor capacity. Therefore, the greatest toxicity of this compound is not related to the ability to increase oxidative stress through the electron transfer mechanism. It was recently demonstrated [16] that PCB-28 stimulates oxidative stress and induced toxicity on plants. Results reported in Fig. 3 show that this compound is the best electron donor and therefore the best reductant molecule among all the PCBs under study. It donates an electron and is oxidized, and this could be related to oxidative stress. PCB-180 is the best electron acceptor and, therefore, the best oxidant.
Results of Fig. 4 indicate that PBDE-28 is the best electron donor. One of the most toxic compounds is PBDE-47, and it is in the middle of the DAM; it is not the best or the worse, electron donor or acceptor. Also in this case, there is no correlation between the electron transfer capacity and the potential toxicity. PBDE-209 is a potent oxidant that produces oxidative stress, and this is in agreement with the results of Fig. 4 since it is the best electron acceptor. Being a good electron acceptor implies that the molecule oxidized other compounds. PBDE-209 accepts electrons that another molecule loses.
To analyze the possibility to donate or accept electrons, it is important to compare with the electron transfer capacity of DNA nitrogen basis. To this end, Fig. 5 presents the DAM including nitrogen bases. All biomolecules are located down to the left with respect to PCBs and PBDEs. Electrons will be transfer from molecules located down to the left (good donors) to those ubicated up to the right (good acceptors). Therefore, nitrogen basis will donate electrons to the persistent organic pollutants. This implies that these biomolecules will be oxidized by these compounds. PBDE-209 may be the most toxic since it is the best electron acceptor, and PCB-28, that is the worse electron acceptor, should be the less toxic.
Figure 5 includes results for melatonin. Melatonin is an antioxidant that was reported as potential antidote for PBDE-47 poisoning [16, 69]. Apparently, melatonin is able to protect cells from oxidative DNA damage induced by PBDE-47. Analyzing the capacity to donate or accept electrons of melatonin, the values in Fig. 5 indicate that it is the best electron donor and the worse electron acceptor of all molecules included. It is a good antioxidant molecule since it is the best electron donor. Melatonin prevents the oxidation of nitrogenous bases because it is oxidized first since it is a better electron donor. Oxidation is the loss of electrons; melatonin donates electrons and with that it is oxidized. These results explain the ability of melatonin to prevent oxidation and DNA damage.
To investigate the potential toxicity of quinone-type derivatives, two examples were selected: PBC-77-O and PBDE-79-O. The DAM of these compounds is presented in Fig. 6. A schematic representation of these molecules is also included. It was reported that quinone-type derivatives are more toxic for two reasons: they have redox properties that increases oxidative stress and can react with GSH and DNA [39]. Redox properties can be analyzed with the DAM reported in Fig. 6.
It is clear that quinone-type derivatives are the best electron acceptor molecules, and this explains the redox toxicity. They increase the oxidative stress accepting electrons from other molecules. Melatonin in this case also protects against the oxidative stress that quinone-type derivatives may produce. The results of Fig. 6 explain the greater toxicity of these compounds.
Throughout all the research reported until now, it has become completely clear that DNA oxidation is an important consequence of life in an oxygen-rich atmosphere. Survival in the presence of oxygen is possible thanks to DNA repair enzymes. However, when further oxidation occurs due to the presence of contaminants such as these persistent organic molecules, the enzymes are not able to repair the DNA, and mutations can occur. PCB-28, PCB-118, PCB-153, PBDE-153, and PBDE-209 are the most abundant compounds found in Antarctic penguins [3]. It was previously reported [70] the genetic damage in different species of Antarctic penguins by microscopic observation of erythrocytic nuclear abnormalities. The information reported provided the reference data. It will be for future work to investigate the genomic damage related to the presence of PCBs and PBDEs.
Substances that are genotoxic may bind directly to DNA or act indirectly leading to DNA damage by affecting enzymes involved in DNA replication, thereby causing mutations which may or may not lead to cancer or birth defects (inheritable damage). To investigate the direct interaction of these persistent organic pollutants, three examples of each family were selected: the best electron donor, the best electron acceptor, and the most toxic according with the reported data. Guanine was used as an example of nitrogen bases. Deoxyadenosine was also used since it has been reported to be a good model for studying the interaction of toxic molecules with DNA and determining potential genotoxic effects [66]. Results are reported in Table 1. For PCB-77, there are previous theoretical results [44] that are in agreement with those reported in this investigation.
According to the binding energies, all interactions are energetically favorable; products are more stable than reactants. The binding energies of the three PCBs are similar. PCB-77 was reported as the most toxic, but the results of this investigation indicate that the binding energy is similar to the others PCBs. This suggests that the high toxicity is not due to the direct interaction with DNA basis. For PBDEs, the best electron acceptor also presents the highest binding energy. These results do not explain why PBDE-28 is more toxic than PDBE-209. Apparently, better electron acceptors will interact stronger than a good electron donor.
In any case, hydrogen bonds are formed between biomolecules and PCBs or PBDEs, and the binding energies of all systems indicate that these molecules can interact directly with the nitrogenous bases of DNA. This suggests a possible explanation for the highly carcinogenic properties of these chemical compounds. More studies are necessary to analyze other reaction mechanisms that can provide more explanations about the toxicity, but as a consequence of this research, it can be said that these persistent organic pollutants are dangerous due to the oxidative stress they can produce and because they can interact directly with the nitrogenous bases of DNA. Furthermore, it is possible to conclude that those persistent organic pollutants that have more halogen atoms that replace hydrogen are potentially more toxic.
To investigate the reaction of quinone-type derivatives, we also investigate its interaction with guanine and deoxyadenosine. The reaction with GSH is also analyzed because it was reported that quinones interact with GSH with a Michael-type addition. Michael-type addition is represented in Scheme 1. Results are reported in Table 2.
The binding energies of quinone-type derivatives indicate that the interaction is energetically possible, the strongest being with GSH. With GSH, the binding energy is larger for PCB-77-O than for PCB-49-O, and therefore, the first is potentially more dangerous than the second. PCB-77-O is also the best electron acceptor. This corroborates our hypothesis that the best electron acceptors will interact stronger than the best electron donors. These results suggest that quinone-type derivatives are potentially more toxic than PCBs and PBDEs, and this is in complete agreement with previous results reported until now.
To analyze the electron transfer capacity of the complexes, Fig. 7 reports the corresponding DAM. All complexes have similar values to PCBs and PBDEs, with the best electron acceptors being those formed with quinone-type derivatives.
The electro-donating and electro-accepting powers are global chemical reactivity descriptors. To have more information about the reactivity of these persistent organic pollutants, it is important to analyze the reactivity at the local level. Local reactivity parameters are useful to differentiate the reactivity of atoms that form molecules. The Fukui function is one of the most used local reactivity parameters. It is associated with the response of the electron density to a change in the number of electrons, with the external potential remaining constant. Condensed Fukui functions are calculated according to the equations presented previously.
Two systems were analyzed: PCB-77 and PBDE-49. The corresponding quinone derivatives were also investigated. The results are presented in Table 3. The condensed Fukui functions indicate that the electrophilic attack will occur on choro or bromine atoms, while the neutral or radical attack will occur on oxygen atoms of the quinone-type derivatives. This is important and may be useful in explaining previous experimental results indicating greater toxicity of quinone-type derivatives than of PCBs or PBDEs. Quinone-type derivatives can react with free radicals. The consequences of this will have to be investigated in the future.
Conclusions
The electron transfer properties of PCBs and PBDEs show that the number of halogens is directly related to the ability to accept electrons. The more chlorine or bromine atoms there are, the more capacity the molecules will have to accept electrons. Molecules that accept electrons are oxidants, since they accept electrons that other molecules lose. Therefore, molecules with more hydrogens substituted by halogens are expected to be more toxic due to the greater oxidative stress they can produce. Quinone-type derivatives are the best electron acceptors among all the compounds studied, and therefore, they are potentially the most toxic. Quinone-type derivatives increase oxidative stress by accepting electrons.
Melatonin is an antioxidant that was reported as potential antidote for PBDE-47 poisoning. The results reported here reveal that melatonin prevents the oxidation of nitrogen bases that PCBs, PBDEs, and quinone-type derivatives may produce, by donating electrons. It is oxidized first since it is a better electron donor. Oxidation is the loss of electrons; melatonin donates electrons and with that it is oxidized. These results explain the ability of melatonin to prevent oxidation and DNA damage.
PCBs, PBDEs, and quinone-type derivatives are toxic because of their electron donor-accepting properties that are related to oxidative stress and because they could bind directly to the nitrogenous bases of DNA. Quinone-type derivatives interact stronger than PCBs and PBDEs with DNA nitrogen bases, and they also interact strong with GSH, in agreement with the experiments. This increases the potential toxicity of quinone-type derivatives. The condensed Fukui functions indicate that the electrophilic attack will occur on choro or bromine atoms, while the neutral or radical attack will occur on oxygen atoms of the quinone-type derivatives. The importance of this research is that it provides information that can be used together with other experiments and calculations, to analyze possible mechanisms that explain the toxicity of these compounds.
Data availability
No datasets were generated or analyzed during the current study.
References
Lammel G, Ghim YS, Grados A, Gao H, Hühnerfuss H (2007) Levels of persistent organic pollutants in air in China and over the Yellow Sea. Atmos Environ 41:452–464
Sharkey M, Harrad S, Abdallah MAE, Drage DS, Berresheim H (2020) Phasing-out of legacy brominated flame retardants: the UNEP Stockholm Convention and other legislative action worldwide. Environ Int 144:106041
Morales P, Roscales JL, Muñoz-Arnanz J, Barbosa A, Jiménez B (2022) Evaluation of PCDD/Fs, PCBs and PBDEs in two penguin species from Antarctic. Chemosphere 286:131871
Kimbrough RD (1995) Polychlorinated biphenyls (PCBs) and human health: an update. Crit Rev Toxicol 25:133–163
Xin ML, Yang JW, Li Y (2017) The mechanism for enhanced oxidation degradation of dioxin-like PCBs (PCB-77) in the atmosphere by the solvation effect. Chem Cent J 11:61
Alaee M, Arias P, Sjodin A, Bergman A (2003) An overview of commercially used brominated flame retardants, their applications, their use patterns in different countries/regions and possible modes of release. Environ Int 29:683–689
Vuong AM, Yolton K, Dietrich KN, Braun JM, Lanphear BP, Chen A (2018) Exposure to polybrominated diphenyl ethers (PBDEs) and child behavior: current findings and future directions. Horm Behav 10:94–104
Annamalai J, Namasivayam V (2015) Endocrine disrupting chemicals in the atmosphere: their effects on humans and wildlife. Environ Int 76:78–97
Pellacani C, Buschini A, Galati S, Mussi F, Franzoni S, Costa LG (2012) Evaluation of DNA damage induced by 2 polybrominated diphenyl ether flame retardants (BDE-47 and BDE-209) in SK-N-MC cells. Int J Tox 31(4):372–379
Liu Z, Zhu Q, Song E, Song Y (2021) Polybrominated diphenyl ethers quinone exhibits neurotoxicity by inducing DNA damage, cell cycle arrest, apoptosis and p53-driven adaptive response in microglia BV2 cells. Toxicology 457:152807
Xu L, Wang Y, Song E, Song Y (2021) Nucleophilic and redox properties of polybrominated diphenyl ether derived-quinone/hydroquinone metabolites are responsible for their neurotoxicity. J Hazard Mat 420:126697
Ross C (2004) (2004) The public health implications of polychlorinated biphenyls (PCBs) in the environment. Ecotoxicol Environ Safe 59:275–291
He W, He P, Wang A, Xia T, Xu B, Chen X (2008) Effects of PBDE-47 on cytotoxicity and genotoxicity in human neuroblastoma cells in vitro. Mutat Res 649:62–70
He P, He W, Wang A, Xia T, Xu B, Zhang M, Chen X (2008) PBDE-47-induced oxidative stress, DNA damage and apoptosis in primary cultured rat hippocampal neurons. Neurotoxicology 29(1):124–129
Sheikh IA, Khweek AA, Beg MA (2016) Peroxisome Proliferator-activated receptors as potential targets for carcinogenic activity of polychlorinated biphenyls: a computational perspective. Anticancer Res 36:6117–6124
Bavithra S, Selvakumar K, Krishnamoorthy G, Venkataraman P, Arunakaran J (2013) Melatonin attenuates polychlorinated biphenyls induced apoptosis in the neuronal cells of cerebral cortex and cerebellum of adult male rats—in vivo. Environ Toxicol Pharmacol 36(1):152–163
Wang C, Sun Y, Ruan H, Yang J (2021) Toxic effects of 2,4,4′- trichlorobiphenyl (PCB-28 on growth, photosynthesis characteristics and antioxidant defense system of Lemna minor L. Plant Physiol Biochem 166:505–511
Costa LG, Giordano G (2007) Developmental neurotoxicity of polybrominated diphenyl ether (PBDE) flame retardants. Neurotoxicology 28(6):1047–1067
Branchi I, Alleva E, Costa LG (2002) Effects of perinatal exposure to a polybrominated diphenyl ether (PBDE 99) on mouse neurobehavioural development. Neurotoxicology 23(3):375–384
Eriksson P, Viberg H, Jakobsson E, Orn U, Fredriksson A (2002) A brominated flame retardant, 2,2`,4,4`,5-pentabromodiphenyl ether: uptake, retention, and induction of neurobehavioral alterations in mice during a critical phase of neonatal brain development. Toxicol Sci 67(1):98–103
Costa LG, Giordano G (2011) Is decabromodiphenyl ether (BDE-209) a developmental neurotoxicant? Neurotoxicology 32(1):9–24
Ellis-Hutchings RG, Cherr GN, Hanna LA, Keen CL (2006) Polybrominated diphenyl ether (PBDE)-induced alterations in vitamin A and thyroid hormone concentrations in the rat during lactation and early postnatal development. Toxicol Appl Pharmacol 215(2):135–145
Kuriyama SN, Wanner A, Fidalgo-Neto AA, Talsness CE, Koerner W, Chahoud I (2007) Developmental exposure to low-dose PBDE-99: tissue distribution and thyroid hormone levels. Toxicology 242(1–3):80–90
Madia F, Giordano G, Fattori V, Vitalone A, Brnachi I, Capone F, Costa LG (2004) Differential in vitro neurotoxicity of the flame retardant PBDE-99 and of the PCB Aroclor 1254 in human astrocytoma cells. Toxicol Lett 154(1–2):11–21
Dingemans MML, de Groot A, van Kleef RGDM, Bergman A, van den Berg M, Vijverberg KPM, Westerink RHS (2008) Hydroxylation increases the neurotoxic potential of BDE-47 to affect exocytosis and calcium homeostasis in PC12 cells. Environ Health Perspect 116(5):637–643
Giordano G, Kavanagh TJ, Costa LG (2008) Neurotoxicity of a polybrominated diphenyl ether mixture (DE-71) in mouse neurons and astrocytes is modulated by intracellular glutathione levels. Toxicol Appl Pharmacol 232:161–168
Huang SC, Giordano G, Costa LG (2010) Comparative cytotoxicity and intracellular accumulation of five polybrominated diphenyl ether congeners in mouse cerebellar granule neurons. Toxicol Sci 114(1):124–132
Merlani PB (ed) (2010) Flame retardants: functions, properties and safety. Nova Science Pub Inc UK. ISBN-13: 978–1607415015
He W, He P, Wang A, Xia T, Xu B, Chen X (2008) Effects of PBDE-47 on cytotoxicity and genotoxicity in human neuroblastoma cells in vitro. Mutat Res 649(1–2):62–70
Mammana SB, del Abraham EC, Camargo AB, Vázquez Á, Altamirano JC (2020) Enzymatic digestion coupled to surfactant-assisted dispersive liquid-liquid microextraction: a mild approach for determining polybrominated diphenyl ethers in human hair sample. Chem Selec 5:2179–2184
Ji K, Choi K, Giesy JP, Musarrat J, Takeda S (2011) Genotoxicity of several polybrominated diphenyl ethers (PBDEs) and hydroxylated PBDEs, and their mechanisms of toxicity. Environ Sci Technol 45(11):5003–5008
Helleday T, Tuominen K, Bergman A, Jenssen D (1999) Brominated flame retardants induce intragenic recombination in mammalian cells. Mutat Res 439:137–147
An J, Li A, Zhong Y, Wang Y, Zhen K, Zhang X, Wang Y, Wu M, Yu Z, Sheng G, Fu J, Huang Y (2011) The cytotoxic effects of synthetic 6-hydroxylated and 6-methoxylated polybrominated diphenyl ether 47 (BDE47). Environ Toxicol 26(6):591–599
Jensen IJ, Eilertsen KE, Otnæs CHA, Mæhre HK, Elvevoll EO (2020) An update on the content of fatty acids, dioxins, PCBs and heavy metals in farmed, escaped and wild Atlantic salmon (Salmo salar L.) in Norway. Food 9(12):1901
Riva C, Binelli A, Cogni D, Provini A (2007) Evaluation of DNA damage induced by decabromodiphenyl ether (BDE-209) in hemocytes of Dreissena polymorpha using the comet and micronucleus assays. Environ Mol Mutagen 48:735–743
Xua L, Wanga Y, Songa E, Song Y (2021) Nucleophilic and redox properties of polybrominated diphenyl ether derived-quinone/hydroquinone metabolites are responsible for their neurotoxicity. J Hazard Mat 420:126697
Bolton JL, Dunlap T (2017) Formation and biological targets of quinones: cytotoxic versus cytoprotective effects. Chem Res Toxicol 30:13–37
Song Y, Wagner BA, Witmer JR, Lehmler HJ, Buettner GR (2009) Nonenzymatic displacement of chlorine and formation of free radicals upon the reaction of glutathione with PCB quinones. Proc Natl Acad Sci USA 106:9725–9730
Bolton JL, Trush MA, Penning TM, Dryhurst G, Monks TJ (2000) Role of quinones in toxicology. Chem Res Toxicol 13:135–160
Jerez S, Motas M, Benzal J, Diaz J, Vidal V, D’Amico V, Barbosa A (2013) Distribution of metals and trace elements in adult and juvenile penguins from the Antarctic Peninsula area. Environ Sci Pollut Res 20:3300–3311
Fragão J, Bessa F, Otero V, Barbosa A, Sobral P, Waluda CM, Guímaro HR, Xavier JC (2021) Microplastics and other anthropogenic particles in Antarctica: using penguins as biological samplers. Sci Total Env 788:147698
Corsolini S, Borghesi N, Schiamone A, Focardi S (2007) Polybrominated diphenyl ethers, polychlorinated dibenzo-dioxins-furans, and-biphenyls in three species of Antarctic penguins. Environ Sci Pollut Res Int 14:421–429
Montone RC, Taniguchi S, Colabuono FI, Martins CC, Cipro CVZ, Barroso HS, Weber RR (2016) Persistent organic pollutants and polycyclic aromatic hydrocarbons in penguins of the genus Pygoscelis in Admiralty Bay- an Antarctic specially managed area. Mar Pollut Bull 106:377–382
Abtouche S, Very T, Monari A, Brahimi M, Assfeld X (2013) Insight on the interaction of polychlorobiphenyl with nucleic acid–base. J Mol Model 19:581–588
Wells PG, McCallum GP, Chen CS, Henderson JT, Lee CJJ, Perstin J, Preston TJ, Wiley MJ, Wong AW (2009) Oxidative stress in developmental origins of disease: teratogenesis, neurodevelopmental deficits, and cancer. Toxicol Sci 108:4–18
Wells PG, McCallum GP, Lam KCH, Henderson JT, Ondovcik SL (2010) Oxidative DNA damage and repair in teratogenesis and neurodevelopmental deficits. Embryo Today: Rev 90:103–109
Hayashi M (2009) Genetic pathways to glioblastomas. Neuropathology 25:1–8
Frisch MJ, Trucks GW, Schlegel HB et al (2016) Gaussian16, Wallingford, CT. Gaussian16 (Revision A.03)
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Account 120:215–241
Petersson GA, Bennett A, Tensfeldt TG, Al-Laham MA, Shirley WA, Mantzaris J (1988) A complete basis set model chemistry. I. The total energies of closed-shell atoms and hydrides of the first-row elements. J Chem Phys 89:2193–2218
Petersson GA, Al-Laham MA (1994) A complete basis set model chemistry. II. Open-shell systems and the total energies of the first-row atoms. J Chem Phys 94:6081–6090
Geerlings P, Chamorro E, Chattaraj PK, De Proft F, Gázquez JL, Liu S, Morell C, Toro-Labbé A, Vela A, Ayers P (2020) Theor Chem Acc 139:36
Gázquez JL, Cedillo A (2007) Vela A (2007) Electrodonating and electroaccepting powers. J Phys Chem A 111:1966–1970
Gázquez JL (2008) Perspectives on the density functional theory of chemical reactivity. J Mex Chem Soc 52:3–10
Shubin L (2022) Conceptual density functional theory: towards a new chemical. Wiley-VCH GmbH. https://doi.org/10.1002/9783527829941
Pearson RG (1968) Hard and soft acids and bases, HSAB, part I Fundamental principles. J Chem Educ 45:581–587
Morell C, Grand A, Toro-Labbé A (2005) New dual descriptor for chemical reactivity. J Phys Chem A 109:205–212
Gutiérrez-Oliva S, Herrera B, Toro-Labbé A (2018) An extension of the Marcus equation: the Marcus potential energy function. J Mol Model 24:104
Martínez A, Rodríguez-Gironés MA, Barbosa A, Costas M (2008) Donator acceptor map for carotenoids, melatonin and vitamins. J Phys Chem A 112:9037–9042
Alfaro RAD, Gómez-Sandoval Z, Mammino L (2014) Evaluation of the antiradical activity of hyperjovinol-a utilizing donor-acceptor maps. J Mol Model 20:2337
Goode-Romero G, Winnberg U, Dominguez L, Ibarra IA, Vargas R, Winnberg E, Martínez A (2020) New information of dopaminergic agents based on quantum chemistry calculations. Sci Rep 10:21581
Galano A, Martínez A (2012) Capsaicin, a tasty free radical scavenger: mechanism of action and kinetics. J Phys Chem B 116(3):1200–1208
Becker PM (2016) Antireduction: an ancient strategy fit for future. Biosci Rep 36(4):e00367
Romero Y, Martínez A (2015) Antiradical capacity of ommochromes. J Molecular Modeling 21:220
Martínez A, López-Rull I, Fargallo JA (2023) To prevent oxidative stress, what about protoporphyrin IX, biliverdin, and bilirubin? Antioxidants 12:1662
Klaunig J, Kamendulis LM, Hocevar BA (2010) Oxidative stress and oxidative damage in carcinogenesis. Toxicol Pathol 38:96–109
Sánchez-Márquez J (2019) New advances in conceptual-DFT. An alternative way to calculate the Fukui function and dual descriptor. J Molec Mod 25:123
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies Some procedures with reduced errors. Mol Phys 19(4):553–566
Luana P, Zhang H, Chenb X, Zhub Y, Hua G, Caib J, Zhangb Z (2022) Melatonin relieves 2,2,4,4-tetrabromodiphenyl ether (BDE-47)-induced apoptosis and mitochondrial dysfunction through the AMPK-Sirt1-PGC-1α axis in fish kidney cells (CIK) Ecotox & Envir. Safety 232:113276
de Mass E, Benzal J, Merino S, Valera F, Palacios MJ, Cuervo JJ, Barbosa A (2015) Erythrocytic abnormalities in three Antarctic penguin species along the Antarctic Peninsula: biomonitoring of genomic damage. Polar Biol 38:1067–1074
Acknowledgements
AM acknowledges support from the Universidad Nacional Autónoma de México (UNAM) and DGAPA through Programa de Apoyo para la Superación del Personal Académico de la UNAM (PASPA) and thanks to LANCAD-UNAM-DGTIC-141 for computer facilities. This is dedicated to Andrés Barbosa, an excellent researcher who died during my sabbatical visit.
Funding
AM would like to thank the Universidad Nacional Autónoma de México for its financial support and DGAPA through Programa de Apoyo para la Superación del Personal Académico de la UNAM (PASPA).
Author information
Authors and Affiliations
Contributions
AM: conceptualization, calculations, validation, visualization, writing and editing draft.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors provided consent to publish.
Competing interests
The authors declare no competing interests.
Additional information
Supplementary Information
All data are available on request.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This research began during a sabbatical leave at the National Museum of Natural Sciences, CSIC, Spain.
This is in honor of Alejandro Toro-Labbé, on the occasion of his 70th birthday.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martínez, A. Toxicity of persistent organic pollutants: a theoretical study. J Mol Model 30, 97 (2024). https://doi.org/10.1007/s00894-024-05890-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-024-05890-8