Abstract
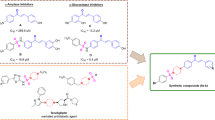
In this work the diastereoisomers (2S) and (2R)-naringenin-6-C-β-D-glucopyroside, isolated for the first time from Clitoria guianensis, were studied using the density functional theory. The frontier molecular orbitals and structural properties showed that the diastereoisomers exhibit the same energy gap 166.61 kcal mol−1 and structural properties different, where in the S diastereoisomer, the bond length between the chiral carbon and the phenolic group is greater (difference of 0.0126 Å). The HPLC data showed that the retention time of the S-diastereoisomer (16.7 min) is shorter than that of R, suggesting that the S compound is more polar than R. The HPLC results corroborates with the molecular electrostatic potential which showed that in the S configuration, the electronegative density was more intense overall, particularly in the glucose molecule. The reactivity indices showed that the diastereoisomers are good electrophiles and reactive species. Finally, the absolute configuration of the diastereoisomers were determined using electronic circular dichroism (ECD) spectroscopy and the theoretical spectra were similar to the experimental.
Methods
All calculations of Density Functional Theory (DFT) and Time-Dependent Density Functional Theory (TDDFT) were performed using the program Gaussian 09 and the structures of the diastereoisomers were generated and analyzed using the GaussView program. The optimization and vibrational frequency calculations were performed using the functional CAM-B3LYP and 6–311 + + G(2d,2p) basis set. Conformational searches were performed for R configuration, by molecular mechanics using the MM + , MMFF, and OPLS05 force fields; the entire molecular mechanics simulation was performed using the Maestro/MacroModel software. The calculations for the simulations of the ECD spectra were performed for the eight lowest energy conformers obtained in the geometric optimization step, and the TDDFT at the CAM-B3LYP/6–311 + + G(2d,2p) theory level used. The effects of methanol and chloroform were calculated using the SMD implicit solvent model.








Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Durigan G (2018) Plantas pequenas do cerrado: biodiversidade negligenciada. Governo do Estado de São Paulo, Secretaria do Meio Ambiente, Instituto Florestal, São Paulo
Fantz PR (1995) Taxonomic notes on new varieties of species of clitoria (leguminisae - phaseoleae - clitoriinae). SIDA, contributions to botany 16:721–730. Published By: The Botanical Research Institute of Texas, Inc
BG, Mehla J, Gupta P (2018) Clitoria-ternatea-linn-a-herb-with-potential-pharmacological-activitiesfuture-prospects-as-therapeutic-herbal-medicine. J Pharma Rep 3:141
Kshetrimayum B (2017) Medicinal plants and its therapeutic uses. OMICS International
Cunha C, Siebeneichler S, Nascimento I, Holzbach J (2020) A new isoflavone and other constituents from roots of clitoria guianensis. J Braz Chem Soc. https://doi.org/10.21577/0103-5053.20200061
Wallace BA, Janes RW (2009) Modern techniques for circular dichroism and synchrotron radiation circular dichroism spectroscopy. Amsterdam: Ios Press Bv,. 232 p. https://www.iospress.com/catalog/books/modern-techniques-for-circular-dichroism-and-synchrotron-radiation-circular-dichroism. Accessed 15 Jun 2022
Fasman GD (1996) Circular dichroism and the conformational analysis of biomolecules. Springer US, Boston
Berova N, Polavarapu PL, Nakanishi K, Woody RW (2012) Frontmatter. In: Comprehensive Chiroptical Spectroscopy. Wiley, Hoboken
Batista A, Angrisani B, Lima ME, da Silva S, Schettini V, Chagas H, dos Santos F Jr, Batista J Jr, Valverde A (2021) Absolute configuration reassignment of natural products: an overview of the last decade. J Braz Chem Soc. https://doi.org/10.21577/0103-5053.20210079
Macedo A, Martorano L, de Albuquerque AC et al (2020) Absolute configuration of (−)-Cubebin, a classical lignan with pharmacological potential, defined by means of chiroptical spectroscopy. J Braz Chem Soc. https://doi.org/10.21577/0103-5053.20200103
Monteiro J, Passero LFD, Jesus JA, Laurenti MD, Lago JHG, Soares MG, Batista ANL, Batista JM, Sartorelli P (2022) Absolute configuration and antileishmanial activity of (–)-Cyclocolorenone isolated from duguetia lanceolata (Annonaceae). Curr Top Med Chem 22:1626–1633
Jimenez DEQ, Barreiro JC, dos Santos Jr FM, de Vasconcellos SP, Porto ALM, Batista JM Jr (2019) Enantioselective ene-reduction of E-2-cyano-3-(furan-2-yl) acrylamide by marine and terrestrial fungi and absolute configuration of (R)-2-cyano-3-(furan-2-yl) propanamide determined by calculations of electronic circular dichroism (ECD) spectra. Chirality 31:534–542. https://doi.org/10.1002/chir.23078
de Freitas Milagre CD, do Amaral BS, Junior JMB, Vilela AFL, Cardoso CL, Milagre HMS (2022) Levetiracetam analogs: chemoenzymatic synthesis, absolute configuration assignment and evaluation of cholinesterase inhibitory activities. Eclética Química 47:17–35. https://doi.org/10.26850/1678-4618eqj.v47.2.2022.p17-35
Budzianowski J, Skrzypczakowa L (1978) 6-C-Glucosylnaringenin from Tulipa gesneriana. Phytochemistry 17:2044. https://doi.org/10.1016/S0031-9422(00)88769-5
Frisch J, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian09, Revision D.01. Gaussian, Inc., Wallingford
Dennington R, Keith T, Millam J (2009) Gauss View, Version 5. Semichem Inc., Shawnee Mission
Parr RG, Donnelly RA, Levy M, Palke WE (1978) Electronegativity: The density functional viewpoint. J Chem Phys 68:3801–3807. https://doi.org/10.1063/1.436185
Duarte HA (2001) Índices de reatividade química a partir da teoria do funcional de densidade: formalismo e perspectivas. Quím Nova 24:501–508. https://doi.org/10.1590/S0100-4042200100040001
Koopmans T (1934) Über die Zuordnung von Wellenfunktionen und Eigenwerten zu den Einzelnen Elektronen Eines Atoms. Physica 1:104–113. https://doi.org/10.1016/S0031-8914(34)90011-2
Schrödinger release (2021) 2021–4: MacroModel, Schrödinger, LLC, New York
Batista Junior JM (2012) Dicroísmo circular vibracional e eletrônico na determinação da configuração absoluta de benzopiranos de Peperomia obtusifolia e Piper gaudichaudianum (Piperaceae): implicações biológicas e biossintéticas. Thesis. Instituto de Química, Universidade Estadual Paulista – UNESP
Marenich AV, Cramer CJ, Truhlar DG (2009) Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B 113:6378–6396. https://doi.org/10.1021/jp810292n
Stein T, Kronik L, Baer R (2009) Reliable prediction of charge transfer excitations in molecular complexes using time-dependent density functional theory. J Am Chem Soc 131:2818–2820. https://doi.org/10.1021/ja8087482
Brédas J-L (2017) Organic electronics: does a plot of the HOMO–LUMO wave functions provide useful information? Chem Mater 29:477–478. https://doi.org/10.1021/acs.chemmater.6b04947
Souza A, Boza I, Ximenes V et al (2019) ELUCIDAÇÃO DA QUIRALIDADE INDUZIDA NA MOLÉCULA DANSILGLICINA NA COMPLEXAÇÃO COM A PROTEÍNA ALBUMINA DO SORO HUMANO (HSA). Quím Nova. https://doi.org/10.21577/0100-4042.20170341
Pereira DH, Ducati LC, Rittner R, Custodio R (2014) A study of the rotational barriers for some organic compounds using the G3 and G3CEP theories. J Mol Model 20:2199. https://doi.org/10.1007/s00894-014-2199-3
Devi D, Arasu MMA, Girija AM (2022) Spectroscopic (FTIR, UV-Vis, NMR) investigations, DFT predictions of global reactivity descriptors and efficient corrosion inhibitors of N-carbobenzoxy-L-ValineSuccinimidyl Ester. J Indian Chem Soc 99:100400. https://doi.org/10.1016/j.jics.2022.100400
Kaviani S, Izadyar M, Housaindokht MR (2016) Solvent and spin state effects on molecular structure, IR spectra, binding energies and quantum chemical reactivity indices of deferiprone–ferric complex: DFT study. Polyhedron 117:623–627. https://doi.org/10.1016/j.poly.2016.06.041
Domingo LR, Pérez P (2011) The nucleophilicity N index in organic chemistry. Org Biomol Chem 9:7168–7175. https://doi.org/10.1039/C1OB05856H
Costa RA, da Silva JN, Oliveira VG, Anselmo LM, Araújo MM, Oliveira KMT, de Nunomura R, CS, (2021) New insights into structural, electronic, reactivity, spectroscopic and pharmacological properties of Bergenin: Experimental, DFT calculations, MD and docking simulations. J Mol Liq 330:115625. https://doi.org/10.1016/j.molliq.2021.115625
Bhattacharya A, Naskar JP, Saha P, Ganguly R, Saha B, Choudhury ST, Chowdhury S (2016) A new oxorhenium(V) complex with benzothiazole derived ligand: Relative stability and global chemical reactivity indices. Inorg Chim Acta 447:168–175. https://doi.org/10.1016/j.ica.2016.04.002
Santos Oliveira I, Marrote Manzano C, Hideki Nakahata D et al (2022) Antibacterial and antifungal activities in vitro of a novel silver(I) complex with sulfadoxine-salicylaldehyde Schiff base. Polyhedron 225:116073. https://doi.org/10.1016/j.poly.2022.116073
Raghi KR, Sherin DR, Saumya MJ, Arun PS, Sobha VN, Manojkumar TK (2018) Computational study of molecular electrostatic potential, docking and dynamics simulations of gallic acid derivatives as ABL inhibitors. Comput Biol Chem 74:239–246. https://doi.org/10.1016/j.compbiolchem.2018.04.001001
Reis DT, de Aguiar Filho SQ, Grotto CGL, Bihain MFR, Pereira DH (2020) Carboxymethylcellulose and cellulose xanthate matrices as potential adsorbent material for potentially toxic Cr3+, Cu2+ and Cd2+metal ions: a theoretical study. Theor Chem Acc 139:96. https://doi.org/10.1007/s00214-020-02610-2
Funding
This study was supported by CAPES (Coordination of Improvement of Higher Education Personnel, Brazil, Funding Code 001 CAPES) and the PROPESQ/Federal University of Tocantins (Edital para tradução de artigos científicos da Universidade Federal do Tocantins, PROPESQ/UFT). Douglas H. Pereira acknowledge the Center for Computational Engineering and Sciences (financial support from FAPESP—Fundação de Amparo à Pesquisa, Grant 2013/08293–7, and Grant 2017/11485–6), the National Center for High Performance Processing (Centro Nacional de Processamento de Alto Desempenho – CENAPAD) in São Paulo for their computational resources and the FAPT (Fundação de Amparo à Pesquisa—Governo do Tocantins) for productivity grant. To Evam Stoll and Christina Moke of the Schrödinger Platform Science and Technology team, Inc. by the license provided.
Author information
Authors and Affiliations
Contributions
A.B.C. and N.N.C. performed the simulations and analyzed the data. C.LC., J.C.H. and I.R.N. performed the experimental analyzes and analyzed the data. I.R.N. supervised the experiments. D.H.P. supervised the simulations. A.B.C, N.N.C. and C.L.C. wrote the first draft of the manuscript. J.C.H., I.R.N. and D.H.P. provided critical feedback and revised the draft the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflicts of interest/competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This paper belongs to the Topical Collection IX Symposium on Electronic Structure and Molecular Dynamics – IX SeedMol
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cruz, Á.B., Ciribelli, N.N., Cunha, C.L. et al. Theoretical and experimental study of the diastereoisomers (2S) and (2R)-naringenin-6-C-β-D-glucopyranoside obtained from Clitoria guianensis. J Mol Model 29, 77 (2023). https://doi.org/10.1007/s00894-023-05482-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-023-05482-y