Abstract
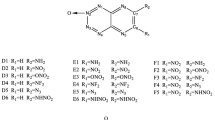
Nitrogen-rich heterocycles and their nitro derivatives are one of the important classes of energetic materials. In the present study, the computational methods have been applied to determine the thermodynamic and detonation properties of nitro derivatives of BN indole molecule. Structure optimization and electronic energy of the designed molecules are determined using the density functional theory. The gas-phase heat of formation of the species concerned is determined by the atomization method. Wave function analysis-surface analysis suite (WFA-SAS) has been applied to determine the condensed phase heat of formation and crystal density of designed molecules. Bond dissociation energy (BDE) is determined to identify the trigger bond. The energy gap between highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) has been calculated to predict the stability of the molecule. Impact sensitivity and detonation properties of designed species are calculated. The calculated parameters show that among all the designed molecules, the molecule A6 (1,2,3,5,6,7-Hexanitrobnindole) has the properties to be considered as a high density energetic molecule.



Similar content being viewed by others
References
Eaton PE, Gilardi RL, Zhang M-X (2000) Polynitrocubanes: advanced high-density, high - energy materials. Adv Mater 12:1143–1148
Gutowski KE, Rogers RD, Dixon DA (2006) Accurate thermochemical properties for energetic materials applications. I Heats of formation of nitrogen-containing heterocycles and energetic precursor molecules from electronic structure theory. J Phys Chem A 110:11890–11897
Qiu L, Xiao HM, Gong XD, Ju XH, Zhu W (2006) Theoretical studies on the structures, thermodynamic properties, detonation properties and pyrolysis mechanism of spironitramines. J Phys Chem A 110:3797–3807
Wang G, Gong XD, Liu Y, Du HC, Xu XJ, Xiao HM (2010) A theoretical investigation on the structures, densities, detonation properties and pyrolysis mechanism of the nitro derivatives of toluenes. J Hazard Mater 177:703–710
Zhang JG, Niu XQ, Zhang SW, Zhang TL, Huang HS, Zhou ZN (2011) Novel potential high-nitrogen-content energetic compounds: theoretical study of diazido-tetrazole (CN10). Comput Theor Chem 964:291–297
Singh HJ, Mukherjee U, Saini R (2012) Computational studies on nitro derivatives of 1-hydroxy-1,2,4-triazole. J Energ Mater 30:265–281
Ghule VD, Srinivas D, Sarangapani R, Jadhav PM, Tiwari SP (2012) Molecular design of aminopolynitroazole-based high-energy materials. J Mol Model 18:3013–3020
Zhang JY, Du HC, Wang F, Gong XD, Huang YS (2011) DFT studies on a high energy density cage compound, 4-trinitroethyl-2,6,8,10,12-pentanitrohezaazaisowurtzitane. J Phys Chem A 115:6617–6621
Xu XJ, Zhu WH, Xiao HM (2008) Theoretical predictions on the structures and properties for polynitrohexaazaadamantanes (PNHAAs) as potential high energy density compounds (HEDCs). J Mol Struct (THEOCHEM) 853:1–6
Fau S, Bartlett RJ (2001) Possible products of the end-on addition of N3− to N5+ and their stability. J Phys Chem A 105(16):4096–4106
Smiglak M, Metlen A, Rogers RD (2007) The second evolution of ionic liquids: from solvents and separations to advanced materials-energetic examples from the ionic liquid cookbook. Acc Chem Res 40:1182–1192
Wang GX, Gong XD, Liu Y, Du HC, Xu XJ, Xiao HM (2011) Looking for high energy density compounds applicable for propellant among the derivatives of DPO with –N3, –NO2, –ONO2 and –NNO2 groups. J Comput Chem 32:943–952
Pagoria PF, Lee GS, Mitchell AR, Schmidt RD (2002) A review of energetic materials synthesis. Thermochim Acta 384:187–204
Kamlet MJ, Adolph HG (1979) The relationship of impact sensitivity with structure of organic high explosives. II Polynitroaromatic explosives. Propellants Explos Pyrotech 4:30–34
Murray JS, Lane P, Politzer P (1995) Relationship between impact sensitivities and molecular surface electrostatic potentials of nitroaromatic and nitroheterocyclic molecules. Mol Phys 85:1–8
Murray JS, Lane P, Politzer P (1998) Effects of strongly electron-attracting components on the molecular surface electrostatic potentials. Application to predicting the impact sensitivities of energetic molecules. Mol Phys 93:187–194
Rice BM, Hare JJ (2002) A quantum mechanical investigation of the relation between the impact sensitivity and charge distribution in energetic molecules. J Phys Chem A 106:1770–1783
Wu W-J, Chi W-J, Li Q-S, Ji J-N, Li Z-S (2016) Strategy of improving the stability and detonation performance for energetic material by introducing the boron atoms. J Phys Org Chem 30:e3699
Wu W-J, Chi W-J, Li Q-S, Li Z-S (2017) Strategy for designing stable and powerful nitrogen-rich high-energy materials by introducing boron atoms. J Mol Model 23:191
Abbey ER, Zakharov LN, S-Yuan L (2011) Boron in disguise: the parent “fused” BN-indole. J Am Chem Soc 133:11508–11511
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Revision A.02. Gaussian, Inc, Wallingford
Chen ZX, Xiao JM, Xiao HM, Chiu YN (1999) Studies on heats of formation for tetrazole derivatives with density functional theory B3LYP method. J Phys Chem A 103:8062–8066
Chen PC, Chieh YC, Tzeng SC (2003) Density functional calculations of the heats of formation for various aromatic nitro compounds. J Mol Struct THEOCHEM 634:215–224
Li M, Xu H, Wu F (2014) Computational studies on the energetic properties of polynitroxanthines. J Mol Model 20:2204–2211
Duan XM, Song GL, Li ZH, Wang XJ, Chen GH, Fan KN (2004) Accurate prediction of heat of formation by combining Hartree-Fock/density functional theory calculation with linear regression correction approach. J Chem Phys 121:7086–7095
Shu Y, Li H, Gao S, Xiong Y (2013) Theoretical studies on densities, stability and detonation properties of 2D polymeric complexes Cu (DAT)2Cl2 and its new analogues Zn (DAT)2Cl2. J Mol Model 19:1583–1590
Singh HJ, Upadhyay MK (2013) Nitro derivatives of 1,3,5-triazepine as potential high energy materials. J Energ Mater 31:301–313
Nicolaides A, Rauk A, Glukhovtsev MN, Radom L (1996) Heats of formation from G2, G2(MP2), and G2(MP2,SVP) total energies. J Phys Chem 100:17460–17464
Wagman DD, Evans WH, Parker VB, Schumm RH, Halows I, Bailey SM, Churney KL, Nuttall RN (1982) The NBS tables of chemical thermodynamic properties: selected values for inorganic and C1 and C2 organic substances in SI units. J Phys Chem Ref Data 11(Suppl. 2) (https://srd.nist.gov/JPCRD/jpcrdS2Vol11.pdf)
Byrd EFC, Rice BM (2006) Improved prediction of heats of formation of energetic materials using quantum mechanical calculations. J Phys Chem A 110:1005–1013
Politzer P, Murray JS, Grice ME, DeSalvo M, Miller E (1997) Calculation of heats of sublimation and solid phase heats of formation. Mol Phys 91:923
Politzer P, Lane P, Concha MC (2003) In: Politzer P, Murray JS (eds) Energetic materials: part I, decomposition, crystal and molecular properties. Amsterdam, Elsevier Chapter 9
Politzer P, Martinez J, Murray JS, Concha MC, Toro-Labbe A (2009) An electrostatic interaction correction for improved crystal density prediction. J Mol Phys 107:2095–2101
Kamlet MJ, Jacobs SJ (1968) Chemistry of detonations. I A Simple Method for Calculating Detonation Properties of CHNO Explosives. J Chem Phys 48:23–35
Sorin B, Laurence EF, Kurt RG, Howard WM, I-Feng WK, Souers PC, Vitello PA (2012) Lawrence Livermore National Laboratory
Bulat FA, Toro-Labbé A, Brinck T, Murray JS, Politzer P (2010) Quantitative analysis of molecular surfaces: areas, volumes, electrostatic potentials and average local ionization energies. J Mol Model 16:1679–1691
Blanksby SJ, Ellison GB (2003) Bond dissociation energies of organic molecules. Acc Chem Res 36:255–263
Pospišil M, Vávra P, Concha MC, Murray JS, Politzer P (2010) A possible crystal volume factor in the impact sensitivities of some energetic compounds. J Mol Model 16:895–901
Frisch A, Nielsen AB, Holder AJ (2000) Gauss-view user’s manual. Gaussian Inc, Wallingford
Singh HJ, Gupta S, Sengupta SK (2014) Computational studies on nitramino derivatives of 1-amino-1,2-azaboriridine as high energetic material. RSC Adv 4:40534–40541
Politzer P, Murray JS (2016) High performance, low sensitivity: conflicting or compatible? Propellants, explosives. Pyrotechnics. 41:414–425
Islam MJ, Kumer A, Sarker N, Paul S, Zannat A (2019) The prediction and theoretical study for chemical reactivity, thermophysical and biological activity of morpholinium nitrate and nitrite ionic liquid crystals: a DFT Study. Adv J Chem Sect A 2:316–326
Kuklja MM (2001) Thermal decomposition of solid cyclotrimethylene trinitramine. J Phys Chem B 105:10159–10162
Song XS, Cheng XL, Yang XD, He B (2006) Relationship between the bond dissociation energies and impact sensitivities of some nitro explosives. Propellants Explos Pyrotech 31:306–310
Talawar MB, Sivabalan R, Mukundan T, Muthurajan H, Sikder AK, Gandhe BR, Rao AS (2009) Environmentally compatible next generation green energetic materials (GEMs) J. Hazard Mater 161:589
Acknowledgments
The authors would like to thank the Head of the Department of Chemistry, DDU Gorakhpur University, for providing the necessary computational facilities.
Funding
One of the authors S.G. is thankful to DST-SERB, New Delhi, for financial support under its National Post-Doctoral Fellowship (NPDF) Scheme No. PDF/2016/003586.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gupta, S., Singh, H.J. Computational studies on nitro derivatives of BN indole as high energetic material. J Mol Model 26, 83 (2020). https://doi.org/10.1007/s00894-020-4337-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-4337-4