Abstract
The reactions of the CH2XO2 (X=F, Cl) with chlorine radical have been firstly investigated utilizing the BMC-CCSD//B3LYP method. The comprehensive calculations indicate that the association-elimination and SN2 displacement reaction mechanisms existed on the singlet potential energy surface (PES), and H-abstraction and SN2 displacement reaction mechanism existed on the triplet PES for the CH2XO2 (X=F, Cl) + Cl reactions. On the triplet PES, the dominant reactions are production of P3X (CHXO2 (X=F, Cl) + HCl) by direct H-abstraction. On the singlet PES, three energy-rich adducts, IM1X (CH2XOOCl (X=F, Cl)), IM2X (CH2XOClO (X=F, Cl)), and IM3X (CH2(OX)OCl (X=F, Cl)), are produced. RRKM-computed reveals that IM1X (CH2XOOCl (X=F, Cl)) produced by collisional stabilization occupied the reaction T ≤ 500 and 400 K, respectively, while P1X (CHXO (X=F, Cl) + HOCl) are forecasted to be the dominant products at high temperatures. The atmospheric lifetime of CH2FO2 and CH2ClO2 in Cl is around 1.18 and 2.50 weeks, respectively. Time-dependent density functional theory (TDDFT) computations imply that IM1X (CH2XOOCl (X=F, Cl)) will photolyze under the sunlight. The current results could guide us to well understand the mechanism of the CH2XO2 (X=F, Cl) + Cl reactions and may be helpful to understand Cl-combustion chemistry.

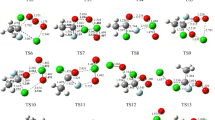
Predicted rate constant of the dominant pathways and the total rate constants at 760 Torr, N2 in the temperature region of 200-3000 K for the CH2XO2 (X=F, Cl) + Cl reactions







Similar content being viewed by others
References
Houghton JT, Ding Y, Griggs DJ Climate change 2001: Thescientific basis. UK Cambridge University Press, Cambridge
Brasseur GP, Orlando JJ, Tyndall GS (eds) (1999) Atomspheric chemistry and global change. Oxford University Press, New York
Tynadall GS, Cox RA, Granier C, Lesclaux R, Moortgat GK, Pilling MJ, Ravishankara AR, Wallington TJ (2001) Atmospheric chemistry of small organic peroxy radicals. J Geophys Res 106:157
Nishida S, Takahashi K, Matsumi Y, Chiappero M, Arguello G, Wallington TJ, Hurley MD, Ball JC (2004) CF3ONO2 yield in the gas phase reaction of CF3O2 radicals with NO. Chem Phys Lett 388:242–247
Sehested J, Nielsen OJ, Wallington TJ (1993) Absolute rate constants for the reaction of NO with a series of peroxy radicals in the gas phase at 295 K. Chem Phys Lett 213:457–464
Vaughan S, Canosa-Mas CE, Pfrang C, Shallcross DE, Watson L, Wayne RP (2006) Kinetic studies of reactions of the nitrate radical (NO3) with peroxy radicals (RO2): an indirect source of OH at night? Phys Chem Chem Phys 8:3749–3760
Wallington TJ, Hurley MD, Schneider WF, Sehested J, Nielsen OJ (1994) Mechanistic study of the gas-phase reaction of CH2FO2 radicals with HO2. Chem Phys Lett 218:34–42
Johnson D, Price DW, Marston G (2004) Correlation-type structure activity relationships for the kinetics of gas-phase RO2 self-reactions and reaction with HO2. Atmos Environ 38:1447–1458
Wallington TJ, Hurley MD, Schneider WF (1996) Atmospheric chemistry of CH3Cl: mechanistic study of the reaction of CH2ClO2 radicals with HO2. Chem Phys Lett 251:164–173
Catoire V, Lesclaux R, Lightfoot PD, Rayez MT (1994) Kinetic study of the reactions of CH2ClO2 with itself and with HO2 and theoretical study of the reactions of CH2ClO, between 251 and 600 K. J Phys Chem 98:2889–2898
Wei WM, Zheng RH (2007) Theoretical study on the reaction mechanism of CH2ClO2 with HO2. J Mol Struct THEOCHEM 812:1–11
Hou H, Deng LZ, Li JC, Wang BS (2005) A systematic computational study of the reactions of HO2 with RO2: the HO2+CH2ClO2, CHCl2O2, and CCl3O2 reactions. J Phys Chem A 109:9299–9309
Shallcross DE, Raventos-Duran MT, Bardwell MW, Bacak A, Solman Z, Percival CJ (2005) A semi-empirical correlation for the rate coefficients for cross- and self-reactions of peroxy radicals in the gas-phase. Atmos Environ 39:763–771
Villenave E, Lesclaux R (1996) Kinetics of the cross reactions of CH3O2 and C2H5O2 radicals with selected peroxy radicals. J Phys Chem 100:14372–14382
Li HW, Tang YZ, Wang RS (2013) The theoretical study on the mechanisms and pathways of the atmospheric CF3O2 + X (X=F,Cl) reactions. Comput Theor Chem 1013:102–108
Frisch MJ, Trucks GW, Schlegel HB, Gill PWM, Johnson BG, Robb MA, Cheeseman JR, Keith TA, Petersson GA, Pople JA (2010) Gaussian 09. Gaussian, Inc., Wallingford
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648
Lee C, Yang W, Par RG (1988) Development of the Colle-Salvetti correlation energy formula into a functional of the electron density. Phys Rev B 37:785
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650–654
Clark T, Chandrasekhar J, Spitznagel GW, Schleyer PVR (1983) Efficient diffuse function-augmented basis sets for anion calculations. III. The 3-21+G basis set for first-row elements, Li–F. J Comput Chem 14:294–301
Gonzalez C, Schlegel HB (1989) An improved algorithm for reaction path following. J Chem Phys 90:2154–2161
Gonzalez C, Schlegel HB (1990) Reaction path following in mass-weighted internal coordinates. J Phys Chem 94:5523
Lynch BJ, Zhao Y, Truhlar DG (2005) The 6-31B(d) basis set and the BMC-QCISD and BMC-CCSD multicoefficient correlation methods. J Phys Chem A 109:1643
Zhao J, Zhang RY (2008) A theoretical investigation of nitrooxyalkyl peroxy radicals from NO3-initiated oxidation of isoprene. Atmos Environ 42:5849–5858
Tang YZ, Nielsen CJ (2012) A systematic theoretical study of imines formation from the atmospheric reactions of RnNH2-n with O2 and NO2 (R=CH3 and CH3CH2; n=1 and 2). Atmos Environ 55:185–189
Tang YZ, Sun HF, Sun JY, Zhang YJ, Wang RS (2014) Theoretical study on mechanisms and pathways of the CF3O2 + ClO reaction. Atmos Environ 92:367–375
Tang YZ, Sun HF, Liu JH, Zhang DL, Wang RS, Dai JH (2013) Computational study on the mechanisms and reaction pathways of the CX3O2 + Br (X = F and Cl) reactions. J Fluor Chem 153:130–136
NIST Computational chemistry comparison and benchmark database. http://srdata.nist.gov/cccbdb/
Seinfeld JH, Pandis SN (1998) Atmoshperic chemistry and physics. Wiley, New York
Brown SS, Stutz J (2012) Nighttime radical observations and chemistry. Chem Soc Rev 41:6405–6447
Chen L, Uchimaru T, Kutsuna S, Tokuhashi K, Sekiya A, Okamoto H (2009) Kinetics of gas-phase reactions of CH3OCH2CF3, CH3OCH3, CH3OCH2CH3, CH3CH2OCH2CH3, and CHF2CF2OCH2CF3 with NO3 radicals at 298 K. Int J Chem Kinet 41:490–497
Csontos J, Rolok Z, Das S, Kallay M (2010) High-accuracy thermochemistry of atmosherically important fluorinated and chlorinated methane derivatives. J Phys Chem A 114:13093–13103
Sander R (1999) Max-Planck institute of chemistry-air chemistry department, version 3. http://www.mpch-mainz.mpg.de/wsander/res/henry.html
Blanco MB, Bejan I, Barnes I, Wiesen P, Teruel MA (2009) The Cl-initiated oxidation of CH3C(O)OCH=CH2, CH3C(O)OCH2CH=CH2, and CH2=CHC(O)O(CH2)3CH3 in the troposphere. Environ Sci Pollut Res 16:641–648
Chang CT, Liu TH, Jeng FT (2004) Atmospheric concentrations of the Cl atom, ClO radical, and HO radical in the coastal marine boundary layer. Environ Res 94:67–74
Funding
This work was supported by the Natural Science Foundations of China (Nos. 21707062 and 41775119), Scientific Research Starting Foundation of Mianyang Normal University (No. QD2016A007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 60 kb)
Rights and permissions
About this article
Cite this article
Zhang, Y., Tang, Y., Sun, J. et al. Theoretical investigations on mechanisms and kinetics of CH2XO2 (X=F, Cl) with Cl reaction in the atmosphere. J Mol Model 26, 139 (2020). https://doi.org/10.1007/s00894-020-4318-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-4318-7