Abstract
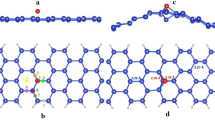
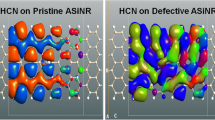
Density functional theory based first-principles investigation study is done on armchair silicene nanoribbons (ASiNRs) for adsorption of uric acid molecule. Pristine and defect-induced variants of ASiNR are considered, and the electronic and transport properties are calculated with the adsorption. The pristine ASiNR with zero band gap is engineered with defect to create a band gap, and a significant change in the band structure of defective ASiNR after the adsorption is observed. The adsorption energy of the defective complex is calculated as − 9.21 eV which is more compared to that of the pristine counterpart, whose adsorption energy comes out to be 7.76 eV. The study shows that introduction of defect reduced the sensitivity of ASiNR toward uric acid molecule.






Similar content being viewed by others
References
Aghaei SM, Monshi MM, Calizo I (2016). RSC 6:94417
Akbari E, Buntat Z, Afroozeh A, Pourmand SE, Farhang Y, Sanaati P (2016). RSC 6:81647
Alesheikh S, Shahtahmassebi N, Roknabadi MR, Shahri RP (2018). Phys Lett A 382:595
Balendhran S, Walia S, Nili H, Sriram S (2014). Willey 11:640
Chen L, Feng B, Wua K (2013). Appl Phys Lett 102:081602
Dávila ME, Marele A, Padova PD, Montero I, Hennies F, Pietzsch A, Shariati MN, Gómez-Rodríguez JM, Lay GL (2012). Nanotechnology 23:385703
Feng J, Liu Y, Wang H, Zhao J, Cai Q, Wang X (2014). Comput Mater Sci 87:218
Guzmán-Verr GG, Voon LC (2007). PRB:76
Han D, Han T, Shan C, Ivaska A, Niua L (2010). Electroanalysis 22:2001
Hu W, Xia N, Wu X, Liab Z, Yang J (2014). Phys Chem Chem Phys 16(15):6957
Liu C, Feng W, Yao Y (2011). Phys. Rev. Lett. 107:076802. https://doi.org/10.1103/physrevlett.107.076802
Osborn T, Farajian A (2014). Nano Res 7(7):945
Quantum Wise, Copenhagen, Denmark: Atomistix Toolkit version 2015.0, 2015 Available from http://www.quantumwise.com. Accessed Jan-June 2019
Singh P, Randhawa DKK, Tarun, Chaudhary BC, Walia GK, Kaur N (2020). J Mol Model. https://doi.org/10.1007/s00894-019-4243-9
Takeda K, Shiraishi K (1994). PRB. 50:14916
Tsai W, Huang C, Chang T, Lin H, Jeng H-T, Bansil A (2013). NCBI 4(1):1500
Walia GK, Randhawa DKK (2014). Su Sci 670:33
Walia GK, Randhawa DKK (2018). Struct Chem 29:1893
Walia GK, Randhawa DKK (2018). J Mol:24(4)
Walia GK, Randhawa DKK. Struct.Chem. 29(1):257
Walia GK, Randhawa DKK. J Mol 24:242
Wang X, Li H, Wang (2012). Phys Chem Chem Phys 14(9):3031
Wella SA, Syaputra M, TDK W, Suprijadi (2016). AIP Conf Proc 1719:030039
Zaminpayma E, Nayebi P (2016). Phys E 84:555
Zhang Y, Chen Y, Zhou K, Liu C, Zeng J, Li H, Peng Z (2009). IOP Sci 20(18):185504
Zheng B, Zhang C (2012). Nanoscale Res Lett 7:422
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tarun, T., Randhawa, D.K.K., Singh, P. et al. Analysis of uric acid adsorption on armchair silicene nanoribbons: a DFT study. J Mol Model 26, 63 (2020). https://doi.org/10.1007/s00894-020-4313-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-020-4313-z