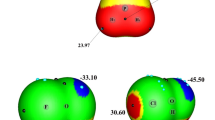
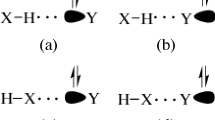Abstract
Quantum chemical calculations have been per-formed for the complexes of formamidine (FA) and hypohalous acid (HOX, X = F, Cl, Br, I) to study their structures, properties, and competition of hydrogen bonds with halogen bonds. Two types of complexes are formed mainly through a hydrogen bond and a halogen bond, respectively, and the cyclic structure is more stable. For the F, Cl, and Br complexes, the hydrogen-bonded one is more stable than the halogen-bonded one, while the halogen-bonded structure is favorable for the I complexes. The associated H-O and X-O bonds are elongated and exhibit a red shift, whereas the distant ones are contracted and display a blue shift. The strength of hydrogen and halogen bonds is affected by F and Li substitutents and it was found that the latter tends to smooth differences in the strength of both types of interactions. The structures, properties, and interaction nature in these complexes have been understood with natural bond orbital (NBO) and atoms in molecules (AIM) theories.



Similar content being viewed by others
References
Scheiner S (1997) Hydrogen bonding: a theoretical perspecitive. Oxford University Press, New York
Jeffrey GA (1997) An introduction to hydrogen bonding. Oxford University Press, New York
Czyznikowska Z (2009) J Mol Struct: THEOCHEM 895:161–167
Scheiner S (2013) Int J Quantum Chem 113:1609–1620
Gilday LC, Lang T, Caballero A, Costa PJ, Felix V, Beer PD (2013) Angew Chem, Int Ed 52:4356–4360
Jentzsch AV, Matile S (2013) J Am Chem Soc 135:5302–5303
Khavasi HR, Tehrani AA (2013) Inorg Chem 52:2891–2905
Ormond-Prout JE, Smart P, Brammer L (2012) Cryst Growth Des 12:205–216
El-Sheshtawy HS, Bassil BS, Assaf KI, Kortz U, Nau WM (2012) J Am Chem Soc 134:19935–19941
Meazza L, Foster JA, Fucke K, Metrangolo P, Resnati G, Steed JW (2013) Nat Chem 5:42–47
Politzer P, Murray JS, Clark T (2010) Phys Chem Chem Phys 12:7748–7757
Del Bene JE, Alkorta I, Elguero J (2010) J Phys Chem A 114:12958–12962
Palusiak M (2010) J Mol Struct: THEOCHEM 945:89–92
Zou JW, Jiang YJ, Guo M, Hu GX, Zhang B, Liu HC, Yu QS (2005) Chem Eur J 11:740–751
Tian WK, Miao Q, Li QZ, Li WZ, Cheng JB (2013) Comput Theor Chem 1012:41–46
Clark T, Hennemann M, Murray JS, Politzer P (2007) J Mol Model 13:291–296
Stone AJ (2013) J Am Chem Soc 135:7005–7009
Tomura M (2009) Chem Phys 359:126–131
Raghavendra B, Arunan E (2007) J Phys Chem A 111:9699–9706
Chudzinski MG, McClary CA, Taylor MS (2011) J Am Chem Soc 133:10559–10567
Lipkowski P, Grabowski SJ, Leszczynski J (2006) J Phys Chem A 110:10296–10302
Li QZ, Wang YL, Liu ZB, Li WZ, Cheng JB, Gong BA, Sun JZ (2009) Chem Phys Lett 469:48–51
Metrangolo P, Murray JS, Pilati T, Politzer P, Resnati G, Terraneo G (2011) CrystEngComm 13:6593–6596
Bouchmella K, Boury B, Dutremez SG, van der Lee A (2007) Chem Eur J 13:6130–6138
Aakeroy CB, Fasulo M, Schultheiss N, Desper J, Moore C (2007) J Am Chem Soc 129:13772–13773
Francisco JS, Sander SP (1993) J Chem Phys 99:6219–6220
Molina MJ, Rowland FS (1974) Nature 249:810–812
Rowland FS, Molina MJ (1975) Rev Geophys Space Phys 13:1–35
Thomas E (1979) Infect Immun 23:522–531
Roos D, Winterbourn CC (2002) Science 296:669–671
Yang YC, Lu HH, Wang WT, Liau I (2011) Anal Chem 83:8267–8272
Cheng XH, Jia HZ, Long T, Feng J, Qin JG, Li Z (2011) Chem Commun 47:11978–11980
Solimannejad M, Alkorta I (2008) Chem Phys Lett 454:201–206
Yuan K, Liu YZ, Zhu YC, Zhang J, Zhang JY (2009) Acta Chin Sin 67:499–506
Yuan K, Liu YZ, Lue LL, Ma WC (2008) Acta Chin Sin 24:1257–1263
Yuan K, Liu YZ, Ma WC, Tang HA, Zhu YC, Zhang J (2009) Chi J Chem 27:900–906
Li ZF, Wang XY, Tang HA, Zhu YC, Li HY (2009) Chem J Chin Univ 30:92–95
Li ZF, Li HY, Liu YZ, Shi XN, Tang HA (2009) Chin Chem Bull 54:3014–3022
Alkorta I, Blanco F, Solimannejad M, Elguero J (2008) J Phys Chem A 112:10856–10863
Li Q, Xu X, Liu T, Jing B, Li W, Cheng J, Gong B, Sun JC (2010) Phys Chem Chem Phys 12:6837–6843
Li QZ, Jing B, Li R, Liu ZB, Li WZ, Luan F, Cheng JB, Gong BA, Sun JZ (2011) Phys Chem Chem Phys 13:2266–2271
Li QZ, Zhao JL, Jing B, Li R, Li WZ, Cheng JB (2011) J Comput Chem 32:2432–2440
Blanco F, Alkorta I, Solimannejad M, Elguero J (2009) J Phys Chem A 113:3237–3244
Zhao Q, Feng D, Sun Y, Hao J, Cai Z (2011) J Mol Model 17:1935–1939
Zabaradsti A, Kakanejadifard A, Ghasemian M (2012) Comput Theor Chem 989:1–6
Zhang ZF, Shen J, Jin NZ, Chen LP, Yang ZY (2012) Comput Theor Chem 999:48–54
Panek JJ, Berski S (2008) Chem Phys Lett 467:41–45
Roohi H, Nowroozi A, Eshghi F (2010) Int J Quantum Chem 110:1489–1499
Greenhill JV, Lue P (1993) Prog Med Chem 30:203–326, and references cited herein
Hollingworth RM (1976) Environ Health Persp 14:57–69
Lim JH, Lee EK, Kim Y (1997) J Phys Chem A 101:2233–2239
Kim Y, Lim S, Kim Y (1999) J Phys Chem A 103:6632–6637
Walewski L, Smaga A, Lesyng B, Sadlej J (2012) J Phys Chem A 116:10412–10419
Li P, Bu YX, Ai HQ, Yan SH, Han KL (2004) J Phys Chem B 108:16976–16982
Bell RL, Truong TN (1994) J Chem Phys 101:10442–10451
Siegbahn PEM, BlombergM RA, Crabtree RH (1997) Theor Chem Acc 97:289–300
Boys SF, Bernardi F (1970) Mol Phys 19:553–558
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian09, Revision A.02. Gaussian Inc, Wallingford
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford, UK
Bulat FA, Toro-Labbé A, Brinck T, Murray JS, Politzer P (2010) J Mol Model 16:1679–1691
Murray JS, Politzer P (2013) ChemPhysChem 14:278–294
Politzer P, Murray JS, Clark T (2013) Phys Chem Chem Phys 15:11178–11189
Berski S, Silvi B, Latajka Z, Leszczynski J (1999) J Chem Phys 111:2542–2555
Politzer P, Riley KE, Bulat FA, Murray JS (2012) Comp Theor Chem 998:2–8
Koch U, Popelier PLA (1995) J Phys Chem A 99:9747–9754
Lipkowski P, Grabowski SJ, Robinson TL, Leszczynski J (2004) J Phys Chem A 108:10865–10872
Arnold WD, Oldfield E (2000) J Am Chem Soc 122:12835–12841
Espinosa E, Molins E, Lecomte C (1998) Chem Phys Lett 285:170–173
Acknowledgments
This work was supported by the National Natural Science Foundation of China (20973149), the Outstanding Youth Natural Science Foundation of Shandong Province (JQ201006), and the Program for New Century Excellent Talents in University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
An, X., Zhuo, H., Wang, Y. et al. Competition between hydrogen bonds and halogen bonds in complexes of formamidine and hypohalous acids. J Mol Model 19, 4529–4535 (2013). https://doi.org/10.1007/s00894-013-1969-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-013-1969-7