Abstract
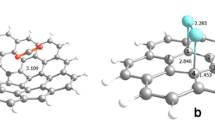
Density functional theory (DFT) calculations performed at the PBE/DZP level using the DFT-D2 method were utilized to investigate the adsorption of phenol on pristine activated carbon (AC) and on activated carbon functionalized with OH, CHO, or COOH groups. Over the pristine AC, the phenol molecule undergoes weak physical adsorption due to van der Waals interactions between the aromatic part of the phenol and the basal planes of the AC. Among the three functional groups used to functionalize the AC, the carboxylic group was found to interact most strongly with the hydroxyl group of phenol. These results suggest that functionalized AC-COOH has great potential for use in environmental applications as an adsorbent of phenol molecules in aqueous phases.





Similar content being viewed by others
References
Fierro V, Torne-Fernandez V, Montane D, Celzard A (2008) Microporous Mesoporous Mater 111:276
Aksu Z, Yener J (2001) Waste Manage 21:695
Park KH, Balathanigaimani MS, Shim WG, Lee JW, Moon H (2010) Microporous Mesoporous Mater 127:1
Dursun G, Cicek H, Dursun A (2005) J Hazard Mater B125:175
Goncharuk VV, Kucheruk DD, Kochkodan VM, Badekha VP (2002) Desalination 143:45
Kojima T, Nishijima K, Matsukata M (1995) J Membr Sci 102:43
Kujawski W, Warszawski A, Ratajczak W, Porebski T, Capala W, Ostrowska I (2004) Desalination 163:287
Rengaraj S, Moon SH, Sivabalan R, Arabindoo B, Murugesan V (2002) Waste Manage 22:543
Bansal RC, Donnet JB, Stoeckli F (1988) Active carbon. Dekker, New York
Boehm HP (1966) Advances in catalysis, vol 16. Academic, New York
Puri BR (1970) In: Walker PJ (ed) Chemistry and physics of carbon, vol 6. Dekker, New York
Marsh H, Heintz EA, Rodriguez-Reinoso F (eds) (1997) Introduction to carbon technologies. University of Alicante, San Vicente del Raspeig
Avgul NN, Kiselev AV (1970) In: Walker PJ (ed) Chemistry and physics of carbon, vol 6. Dekker, New York
Adib F, Bagreev A, Bandosz TJ (1999) J Colloid Interface Sci 216:360
Adib F, Bagreev A, Bandosz TJ (2000) Langmuir 16:1980
Bandosz TJ, Jagiello J, Schwarz J (1993) Langmuir 9:2518
Salame II, Bandosz TJ (2003) J Colloid Interface Sci 264:307
Leng CC, Pinto NG (1997) Carbon 35:1375
Tessmer TH, Vidic RD, Uranowski LJ (1997) Environ Sci Technol 31:1872
Franklin RE (1951) Proc R Soc A 209:196
Harris PJF, Burian A, Duber S (2000) Phil Mag Lett 80:381
Harris PJF (2005) Crit Rev Solid State Mater Sci 30:235
Zetterstrom P, Urbonaite S, Lindberg F, Delaplane RG, Leis J, Svensson G (2005) J Phys Condens Matter 17:3509
Hawelek L, Koloczek J, Brodka A, Dore JC, Honkimaeki V, Burian A (2007) Phil Mag 87:4973
Harris PJF (2008) J Phys Condens Matter 20:362201
Padak B, Wilcox J (2009) Carbon 47(12):2855
Chen N, Yang RT (1998) Carbon 36:1061
Chen N, Yang RT (1998) J Chem Phys A 102:6348
Lamoen D, Persson BNJ (1998) J Chem Phys 108:3332
Zhu ZH, Lu GQ (2004) Langmuir 20:10751
Janiak C, Hoffmann RR, Sjovall P, Kasemo B (1993) Langmuir 9:3427
Pliego JR, Resende SM, Humeres E (2005) J Chem Phys 314:127
Thomson KT, Gubbins KE (2000) Langmuir 16:5761
Terzyk AP, Furmaniak S, Gauden PA, Harris PJF, Włoch J, Kowalczyk P (2007) J Phys Condens Matter 19:406208
Terzyk AP, Furmaniak S, Harris PJF, Gauden PA, Włoch J, Kowalczyk P, Rychlicki G (2007) Phys Chem Chem Phys 9:59195
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Klamt A, Schüürmann G (1993) J Chem Soc Perkin Trans 2 5:799–805
Wu X, Vargas MC, Nayak S, Lotrich V, Scoles G (2001) J Chem Phys 115:8748
Grimme S (2006) J Comput Chem 27:1787
Zhao X, Gao B, Cai QH, Wang XG, Wang XZ (2011) Theor Chem Accounts 129:85
Peyghan AA, Baei MT, Moghimi M, Hashemian S (2012) Comput Theor Chem 997:63
Rokhina ER, Lahtinen M, Makarova K, Jegatheesan V, Virkutyte J (2012) Bioresour Technol 113:127–131
Moreno-Castilla C, Ferro-Garcia MA, Joly JP, Bautista-Toledo I, Carrasco-Marin F, Rivera-Utrilla J (1995) Langmuir 11:4386–4392
Díaz E, Ordóñez S, Vega A (2007) J Colloid Interface Sci 305:7–16
Michalska D et al (2001) J Phys Chem A 105:8734–8739
Acknowledgments
This work was supported by the Ministry of Training and Education under project number B2011-17- 03. The authors appreciate the financial support from grants B2011-17-03.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cam, L.M., Van Khu, L. & Ha, N.N. Theoretical study on the adsorption of phenol on activated carbon using density functional theory. J Mol Model 19, 4395–4402 (2013). https://doi.org/10.1007/s00894-013-1950-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-013-1950-5