Abstract
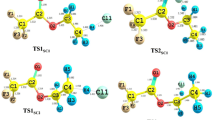
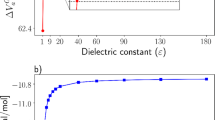
We investigated CO2 absorption in aqueous alkanolamine solutions using density functional theory with dielectric continuum solvation models (SMD/IEF-PCM and COSMO-RS). We varied the alkyl chain length (m = 2, 3, 4) and the alcohol chain length (n = 2, 3, 4) in the alkanolamine structures, H(CH2) m NH(CH2) n OH. Using the SMD/IEF-PCM/B3LYP/6-311++G(d,p) and COSMO-RS/BP/TZVP levels of theory, our calculations predict that the product of CO2 absorption (carbamate or bicarbonate) is strongly affected by the alcohol length but does not differ significantly by varying the alkyl chain length. This prediction was confirmed experimentally by 13C-NMR. The observed sensitivity to the alcohol chain length can be attributed to hydrogen bonding effects. The intramolecular hydrogen bonds of HN · · · HO, NH2 + · · · OH, and NCOO− · · · HO induce ring structure formation in neutral alkanolamines, protonated alkanolamines, and carbamate anions, respectively. The results from our studies demonstrate that intramolecular hydrogen bonds play a key role in CO2 absorption reactions in aqueous alkanolamine solutions.







Similar content being viewed by others
References
Rochelle GT (2009) Amine scrubbing for CO2 capture. Science 325:1652–1654
Rochelle G, Chen E, Freeman S, Wagener D, Xu Q, Voice A (2011) Aqueous piperazine as the new standard for CO2 capture technology. Chem Eng J 171:725–733
Maiti A, Bourcier WL, Aines RD (2011) Atomistic modeling of CO2 capture in primary and tertiary amines—heat of absorption and density changes. Chem Phys Lett 509:25–28
Robinson K, McCluskey A, Attalla M (2011) An FTIR spectroscopic study on the effect of molecular structural variations on the CO2 absorption characteristics of heterocyclic amines. ChemPhysChem 12:1088–1099
Goto K, Okabe H, Chowdhury F, Shimizu S, Fujioka Y, Onoda M (2011) Development of novel absorbents for CO2 capture from blast furnace gas. Int J Greenhouse Gas Control 5:1214–1219
Yamada H, Matsuzaki Y, Okabe H, Shimizu S, Fujioka Y (2011) Quantum chemical analysis of carbon dioxide absorption into aqueous solutions of moderately hindered amines. Energy Procedia 4:133–139
Yamada H, Matsuzaki Y, Higashii T, Kazama S (2011) Density functional theory study on carbon dioxide absorption into aqueous solutions of 2-amino-2-methyl-1-propanol using a continuum solvation model. J Phys Chm A 115:3079–3086
Yamada H, Shimizu S, Okabe H, Matsuzaki Y, Chowdhury F, Fujioka Y (2010) Prediction of the basicity of aqueous amine solutions and the species distribution in the amine−H2O−CO2 system using the COSMO-RS method. Ind Eng Chem Res 49:2449–2455
McCann N, Phan D, Wang X, Conway W, Burns R, Attalla M, Puxty G, Maeder M (2009) Kinetics and mechanism of carbamate formation from CO2(aq), carbonate species, and monoethanolamine in aqueous solution. J Phys Chem A 113:5022–5029
da Silva EF, Svendsen HF (2007) Computational chemistry study of reactions, equilibrium and kinetics of chemical CO2 absorption. Int J Greenhouse Gas Control 1:151–157
Vaidya PD, Kenig EY (2007) CO2—alkanolamine reaction kinetics: a review of recent studies. Chem Eng Technol 30:1467–1474
Chakraborty AK, Astarita G, Bischoff KB (1986) CO2 absorption in aqueoussolutions of hinderedamines. Chem Eng Sci 41:997–1003
Puxty G, Rowland R, Allport A, Yang Q, Bown M, Burns R, Maeder M, Attalla M (2009) Carbon dioxide postcombustion capture: A novel screening study of the carbon dioxide absorption performance of 76 amines. Environ Sci Technol 43:6427–6433
Jezierska-Mazzarello A, Szatyłowicz H, Krygowski TM (2012) Interference of H-bonding and substituent effects in nitro- and hydroxy-substituted salicylaldehydes. J Mol Model 18:127–135
Zawada A, Kaczmarek-Kędziera A, Bartkowiak W (2012) On the potential application of DFT methods in predicting the interaction-induced electric properties of molecular complexes. J Mol Model 18:3073–3086
Nagaraju M, Sastry GN (2011) Effect of alkyl substitution on H-bond strength of substituted amide-alcohol complexes. J Mol Model 17:1801–1816
Oliveira BG, Araújo RCMU, Carvalho AB, Ramos MN (2009) A chemometrical study of intermolecular properties of hydrogen-bonded complexes formed by C2H4O · · · HX and C2H5N · · · HX with X = F, CN, NC, and CCH. J Mol Model 15:421–432
Delchev VB, Mikosch H (2006) DFT study of the gas phase proton transfer in guanine assisted by water, methanol, and hydrogen peroxide. J Mol Model 12:229–236
Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105:2999–3093
Marenich AV, Cramer CJ, Truhlar DG (2009) Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B 113:6378–6396
Klamt A (2011) The COSMO and COSMO-RS solvation models. WIREs Comput Mol Sci 1:699–709
Eckert F, Klamt A (2006) Accurate prediction of basicity in aqueous solution with COSMO-RS. J Comput Chem 27:11–19
Eckert F, Klamt A (2004) Fast solvent screening via quantum chemistry: COSMO-RS approach. AICHE J 48:369–385
Klamt A, Jonas V, Burger T, Lohrenz JCW (1998) Refinement and parameterization of COSMO-RS. J Phys Chem A 102:5074–5085
Francl MM (1985) Polarization corrections to electrostatic potentials. J Phys Chem 89:428–433
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam NJ, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, Rev A1. Gaussian Inc, Wallingford
da Silva EF, Svendsen HF (2003) Prediction of the pK a values of amines using ab initio methods and free-energy perturbations. Ind Eng Chem Res 42:4414–4421
Böttinger W, Maiwald M, Hasse H (2008) Online NMR spectroscopic study of speciesdistribution in MEA−H2O−CO2 and DEA−H2O−CO2. Fluid Phase Equilibria 263:131–143
Jakobsen JP, Krane J, Svendsen HF (2005) Liquid-phase composition determination in CO2−H2O−alkanolamine systems: An NMR study. Ind Eng Chem Res 44:9894–9903
Hori K, Yamaguchi T, Uezu K, Sumimoto M (2011) A free-energy perturbation method based on Monte Carlo simulations using quantum mechanical calculations (QM/MC/FEP method): Application to highly solvent-dependent reactions. J Comput Chem 32:778–786
Acknowledgments
This work was financially supported by the COURSE 50 project founded by the New Energy and Industrial Technology Development Organization, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamada, H., Matsuzaki, Y., Chowdhury, F. et al. Computational investigation of carbon dioxide absorption in alkanolamine solutions. J Mol Model 19, 4147–4153 (2013). https://doi.org/10.1007/s00894-012-1749-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1749-9