Abstract
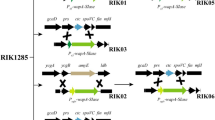
The Thermus thermophilus TH125 α-galactosidase gene, agaT, and flanking sequences were cloned in Escherichia coli and sequenced as well as flanking sequences of the previously cloned agaT from Thermus brockianus ITI360. Different structures of putative α-galactosidase operons in the two Thermus strains were revealed. Downstream of and overlapping with the α-galactosidase genes of both strains, a gene was identified that is similar to the galactose-1-phosphate uridylyltransferase gene (galT) of E. coli and Streptomyces lividans. Upstream of the agaT of T. brockianus ITI360, four open reading frames were observed. The deduced translation products displayed similarity to components of bacterial binding protein-dependent transport systems and a β-galactosidase. No galactoside utilization genes were identified upstream of agaT in T. thermophilus TH125. The inactivation of the α-galactosidase genes of both strains by insertional mutagenesis led to an inability to use melibiose or galactose as a single carbohydrate source. An attempt was made to isolate a gene encoding the enzyme responsible for para-nitrophenyl-(pNP-) β-galactoside hydrolyzing activity in T. thermophilus TH125. A gene designated bglT was cloned and expressed in E. coli. The inactivation of the bglT gene led to 55% reduction of the pNP-β-galactoside hydrolyzing activity in the mutant strain in comparison to the wild type.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: April 28, 1999 / Accepted: September 9, 1999
Rights and permissions
About this article
Cite this article
Fridjonsson, O., Watzlawick, H. & Mattes, R. The structure of the α-galactosidase gene loci in Thermus brockianus ITI360 and Thermus thermophilus TH125. Extremophiles 4, 23–33 (2000). https://doi.org/10.1007/s007920050004
Issue Date:
DOI: https://doi.org/10.1007/s007920050004