Abstract
The background of this study is to examine the associations of individual and combined early morning patterns (i.e., active commuting to school, physical activity before school, having breakfast and good sleep) with white matter microstructure (WMM) and, whether the associated white mater microstructure outcomes were related to mental health outcomes in children with overweight or obesity. 103 children with overweight or obesity (10.0 ± 1.1 years old, 42 girls) from the ActiveBrains project participated in this cross-sectional study. Early morning patterns and mental health indicators (i.e., self-esteem, optimism, positive and negative affect, stress, depression and anxiety) were self-reported by the children using validated questionnaires. WMM was assessed by magnetic resonance imaging using diffusion tensor imaging. When examined independently, early morning patterns were not related with WMM (all P > 0.05). However, the combination of early morning patterns was related with WMM (P < 0.05). Specifically, physically active early morning patterns (i.e., active commuting to school and physical activity before school) were associated with global fractional anisotropy (FA) (β = 0.298, P = 0.013) and global radial diffusivity (RD) (β = − 0.272, P = 0.021), as well as with tract-specific FA (β = 0.314, P = 0.004) and RD (β = − 0.234, P = 0.032) in the superior longitudinal fasciculus (SLF). Furthermore, combined physically active early morning pattern-associated global (i.e., FA and RD) and tract-specific (i.e., FA and RD in the SLF) WMM indicators were positively associated with happiness (β absolute value range from 0.252 to 0.298, all P < 0.05). A combination of physically active early morning patterns may positively relate to white matter microstructure in children with overweight or obesity, and, in turn, happiness.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Childhood is a critical period for neurodevelopment [1]. In particular, the brain undergoes significant changes in white matter structure [2]. The structure of white matter is responsible for providing fast and efficient transmission of information between brain areas into structural networks to support cognition and mental health [3, 4]. Indeed, white matter microstructure was related to general and specific psychopathology symptoms (e.g., internalizing and externalizing problems) in children [5]. Emerging studies have considered white matter as one brain feature susceptible for modification by lifestyle behaviors (e.g., physical activity, sleep or diet) during childhood [6,7,8,9], which in turn may be related to childhood mental health outcomes [5].
Specifically, we have previously shown that physical activity was related to enhanced white matter microstructure in the present sample of children with overweight and obesity [7]. In addition, we observed in a population-based study conducted in normal-weight children that total physical activity (i.e., outdoor playing and sport participation) was associated with improved white matter microstructure indicators [8]. Importantly, new paradigms based on chronotype suggest that physically active early morning behaviors may confer additional health benefits, such as dementia risk [10]. Likewise, irrespective of total physical activity, morning physical activity has been associated with better cardiometabolic health outcomes [11, 12] and lower risks of incident cardiovascular diseases [13]; this highlights the potential importance of chronoactivity mainly in physical physiological health, which might be extended to brain health. However, the previous studies have not examined the association of early active morning patterns with white matter microstructure. The only study examining active commuting, included both active commuting to (during mornings) and from (during afternoon) school, and found no associations with white matter indicators [8]. In addition, other early morning factors, such as having breakfast or good sleep, may also influence white matter microstructure [6, 9, 14]. For example, sleep disturbances were negatively associated with white matter microstructure in preadolescents [9]. In addition, having bread (compared with rice) for breakfast was associated with greater white matter volume in children [14].
Beyond the influence of individual behaviors on white matter, an approach that combines multiple behaviors, has been broadly recommended based on the multifactorial etiology of chronic diseases and behavioral outcomes [15]; in turn, considering the cumulative, or combined, effects of each behavior may have a larger impact on health outcomes than any single factor [16]. As far as we know, there has not been a previously published study that has examined the association between individual and combined early morning patterns on white matter microstructure in children with overweight or obesity. Specifically, obesity is associated with alterations in white matter properties as well as development of psychiatric disorders as compared to normal-weight individuals [17,18,19]. For example, obese children have shown worse white matter microstructure in the cerebellar peduncles and higher risk of both anxiety and depression than their normal-weight peers [20, 21]. Therefore, there is a clear need for studies that examine different early morning patterns and their associations with white matter microstructure in overweight/obese children, as well as their coupled influence on mental health.
The aim of the present study was to examine the associations of individual and combined early morning patterns (i.e., active commuting to school, physical activity before school, having breakfast and good sleep) with white matter microstructure and, whether the associated white matter microstructure outcomes were related to mental health in children with overweight or obesity.
Methods
Participants
The present cross-sectional study was developed within the Active Brains Project framework (http://profith.ugr.es/). A detailed description of the study design and methods has been published elsewhere [22]. A total of 110 children with overweight and obesity (8–11 years old) from Granada (Spain) were recruited and assessed from 2014 to 2016. Children were categorized as overweight and obesity grade I/II/III according to age- and sex-specific BMI cut points [23, 24]. Of these, 103 (42 girls) were included in the present analyses. The ActiveBrains study was approved by the Review Committee for Research Involving Human Subjects at the University of Granada and was registered in ClinicalTrials.gov (Identifier: NCT02295072).
Early morning patterns
Early morning patterns included both physically active behaviors (i.e., physical activity before school and active commuting to school) and non- physically active behaviors (i.e., having breakfast and good sleep). Physical activity before school and active commuting to school were assessed by the Youth Activity Profile-Spain (YAP-S) questionnaire. The YAP-S is a cross-translated and adapted version of the original YAP (available at: http://profith.ugr.es/yap?lang=en). The original YAP was calibrated through a series of studies by Saint Maurice et al. [25, 26]. The YAP-S questionnaire showed adequate reliability for activity at school and out-of-school [weighted kappa coefficient –K– = 0.66–0.76; the intraclass correlation coefficient (ICC) = 0.83–0.86] in children [27]. Participants were asked on “how many days” during the last week: (i) they performed physical activity before school (from 6.00 a.m. to 9.00 a.m.) for at least 10 min (not including walk or bike to school) and (ii) they walked or cycled to school; answer ranging from 0 to 5 days. Each answer was classified into two categories as never (0 days) vs. some days (1 to 5 days).
Having breakfast was evaluated by an item from the KIDMED Questionnaire [28]. This questionnaire consists of 16 yes/no questions regarding the Mediterranean diet, and includes an item for skipping breakfast. This answer was inverted as not having breakfast vs. having breakfast. Although, a classical validation study has not been performed due to the structure of the KIDMED questionnaire; there is evidence that higher scoring of the KIDMED index is associated with expected patterns of food and nutrient intakes indicating good dietary quality, demonstrating its construct validity [29, 30], and has been extensively used in youth Spanish population [31,32,33].
Good sleep was defined as combining quantity and quality of sleep. Sleep quantity was evaluated by the Paediatric Sleep Questionnaire (PSQ) and sleep quality was evaluated by the Child Health Utility 9D Questionnaire (CHU9D). The PSQ was completed by the parents according to their child’s usual sleep habits and includes two items on the normal time to go to bed and the time of day for the child to get out of bed during a weekday. The Spanish version of the PSQ showed good reliability for usual sleep habits (K > 0.50) [34]. Sleep duration was calculated and recoded as meeting the sleep recommendation for children by the National Sleep Foundation (9–11 h) vs. not meeting the sleep recommendation [35]. The CHU9D consists of 9 items to assess the child’s functioning across several domains including sleep problems. Participants rated their response using a 5-point Likert type scale, from 1 = “no sleep problems” to 5 = “almost couldn’t sleep”; answers were categorized as no sleep problems [1] vs. others [2,3,4,5]. The Cronbach alpha for the CHU9D was 0.781, indicating an acceptable level of internal consistency [36]. The label of good sleep was defined based on two categories: either meeting the sleep recommendation and not experiencing sleep problems vs. others (i.e., not meeting the recommendations or the presence of sleep disturbances).
Magnetic resonance imaging (MRI) procedure
Imaging acquisition
MRI data were collected with a 3.0 Tesla Siemens Magnetom Tim Trio scanner (Medical Solutions, Erlangen, Germany) [7]. Diffusion tensor imaging (DTI) data were acquired with an Echo Planar Imaging (EPI) sequence following the parameters: echo time (TE) = 90 ms, trace of the tensor (TR) = 3300 ms, field of view (FOV) = 230 mm × 230 mm, flip angle = 90, matrix = 128 × 128, slice thickness = 4 mm, number of slices = 25 and voxel resolution = 1.8 × 1.8 × 4 mm3. One volume without diffusion weighting (b = 0 s/mm2) and 30 volumes with diffusion weighting (b = 1000 s/mm2) were collected [7]. We use fractional anisotropy (FA), mean diffusivity (MD), radial diffusivity (RD), and axial diffusivity (AD) as derived DTI metrics. FA shows the degree to which water diffuses preferentially along one axis and increases during child development and to be lower in neurological and psychiatric diseases. MD is a scalar describing the average diffusion in all directions, with higher levels indicating relatively unimpeded diffusion (i.e., negatively correlated with FA). RD measures water diffusion perpendicular to the axonal wall. AD represents the diffusivity of water in the direction parallel to the fiber bundles in anisotropic tissue, such as white matter. High values of FA and AD, and low values of MD and RD are shown as indicators of healthier white matter microstructure during childhood [37].
Imaging preprocessing
Image preprocessing was carried out through the Functional MRI of the Brain Software Library (FSL) [38]. First, we corrected for eddy current-induced distortions and participant movement with the 6.0.1 version of eddy correction (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/eddy). Next, outliers were replaced by non-parametric predictions by the Gaussian Process. To account for rotations applied to the imaging data during motion correction, the resulting transformation matrices were used to rotate the diffusion gradient direction table. Non-brain tissue was removed using the FSL Brain Extraction Tool. Finally, the diffusion tensor was fit, and common scalar maps (i.e., FA, MD, RD and, AD) were estimated.
Probabilistic fiber tractography
Probabilistic fiber tractography was performed using the fully automated FSL plugin, “AutoPtx” (https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/AutoPtx). Diffusion data were processed with the Bayesian Estimation of Diffusion Parameters Obtained using Sampling Techniques (BEDPOSTx), considering two fiber orientations at each voxel [39]. Then, for each participant, the FSL nonlinear registration tool (FNIRT) was used to align the FA map to the FMRIB-58 FA template image. The inverse of the nonlinear warp field was calculated and applied to a series of predetermined target, seed, exclusion, and termination masks produced by the AutoPtx plugin [40].
Probabilistic fiber tracking was carried out with the FSL Probtrackx module using the supplied tract-specific masks (i.e., target, seed, etc.) that were deformed to the native diffusion image space of each subject. The resulting path distributions were normalized to a scale from 0 to 1 using the total number of successful seed-to-target attempts and low-probability voxels likely related to noise were removed as a result of the established threshold.
White matter tract segmentation was accomplished by thresholding the normalized tract density images based on previously established values [cingulate gyrus part of cingulum (CGC): 0.01, corticospinal tract (CST): 0.005, inferior longitudinal fasciculus (ILF): 0.005, superior longitudinal fasciculus (SLF): 0.001, uncinate fasciculus (UNC): 0.01, forceps major (FMA): 0.005, forceps minor (FMI): 0.01] [40]. Next, average FA, MD, RD, and AD values were calculated for each tract. Connectivity distributions were estimated for seven large fiber bundles (i.e., CGC, CST, ILF, SLF, UNC, FMA, and FMI) selected based on previous reports [4, 40]. To assess whether exposures were related to global measures of white matter integrity (i.e., global FA, MD, RD, and AD), selected tracts were combined into a single factor “Global Factor”. The global factor was calculated by averaging all tracts and weighting this average by the size (volume) of the tracts (in the neuroimaging literature, in particular in cortical morphology studies, it is a common practice to ensure that small regions do not contribute equally as larger regions) [8, 9]. Left and right hemispheres were averaged for the global and individual tracts’ DTI metrics.
Image quality assurance
Image quality assurance was performed following the previous procedures used in children [8]. Image quality was classified using a 4-point scale with values 1 = “excellent”, 2 = “minor”, 3 = “moderate”, and 4 = “severe”. Datasets which had insufficient quality (i.e., moderate and severe) for statistical analyses were excluded (n = 2). The sum-of-squares error maps from the tensor estimation were computed and visually inspected for structured noise in FSL. First, the native space FA map registration was examined to ensure that images were all correctly aligned to the template (masks were properly mapped to native space). Second, all tracts were visualized to ensure rigorous path reconstruction. Raw image quality and probabilistic tractography data were visually examined by a rater blind to clinical data, and a second rater checked it in case of ambiguity [4].
Mental health indicators
Mental health indicators were divided into two groups: psychological well-being (i.e., happiness, self-esteem, optimism and positive affect) and psychological ill-being (i.e., stress, depression anxiety and negative affect) [41].
Happiness was measured with the 4-item Subjective Happiness Scale (SHS). The subjects responded to 4 questions with answers ranging from 1 to 7. The score was obtained from the sum of the 3 first items with values ranging from 3 (low happiness) to 21 (high happiness). The Spanish version of SHS showed an adequate internal consistency (Cronbach’s alpha ranged from 0.79 to 0.94), appropriate test–retest reliability (Pearson’s r ranged from 0.55 to 0.90) and convergent validity (Pearson’s r ranged from 0.52 to 0.72) [42].
Self-esteem was evaluated by the Rosenberg Self-Esteem Scale (RSE). The test includes 10 items measuring positive and negative feelings. The final score is a measure of global self-worth. Higher scores indicate higher self-esteem. The RSE is a reliable (Cronbach’s alpha = 0.86) and valid (Pearson’s r = 0.41) self-report to assess self-esteem in children [43].
Dispositional optimism was measured with the Life Orientation Test-Revised (LOT-R). This test contains 10 items from pessimism to optimism, and the final score was calculated by summing the 6 items that assessed optimism. LOT-R is a valid, reliable and useful self-report measure to evaluate optimism in children (Cronbach’s alpha = 0.78 and Pearson’s r ranged for test–retest reliability from 0.56 to 0.79) [44].
Stress was assessed by The Children’s Daily Stress Inventory (CDSI). The CDSI measures the prevalence of stressful events in four areas: school, friends, health and family. The answers are dichotomous with yes/no items. The result of the final sum indicates a high or low level of stress. The inventory has good reliability (Pearson’s r = 0.78) and validity (Cronbach’s alpha = 0.70) in primary school students from Spain [45].
Depression was evaluated by the Children’s Depression Inventory (CDI). It measures symptoms related to dysthymic disorder or depression in children. The 27 items are grouped into five factor areas: interpersonal problems, negative mood, anhedonia, ineffectiveness and negative self-esteem. The final score was calculated summing values from 0 (low depression level) to 54 (high depression level). The Cronbach alpha for this test has reported to be 0.84 for males and 0.87 for females [46].
Childhood trait anxiety was assessed by the State-Trait Anxiety Inventory for Children (STAIC-T). The STAI measures tension, apprehension, worry and nervousness, but is normally used as a global anxiety measure. The inventory has 20 items with scores categorized from 1 to 3 (almost never-often). A high score is indicative of a high trait anxiety level. STAIC-T is widely used, reliable and extensively validated (Cronbach alpha = 0.94) [47].
Positive and negative affect was evaluated by The Positive and Negative Affect Schedule for Children (PANAS-C). The PANAS-C has 20 items with answers ranging from 1 to 3. Positive affect was calculated by summing 10 items, and negative affect was obtained from the sum of 10 items; higher scores reflect higher positive and negative affect. Prior data have shown a Cronbach alpha from 0.87 to 0.90 for the positive affect subscale and 0.87–0.94 for the negative affect subscale [48].
Covariates
Sex, peak height velocity (PHV, year) and parent education university level were included as covariates based on the previous studies from the ActiveBrains project [49,50,51,52]. PHV is a common indicator of maturity (i.e., biological age) in children and adolescents [53]. PHV was obtained from anthropometric variables (weight, height and/or seated height) using Moore’s equations [54]. Parent education, a proxy measure of socioeconomic status, was defined by the highest completed education and divided into 3 groups ranging from 0 (neither), 1 (one of parent) or 2 (both parents). In sensitivity analyses, additional covariates were included [body mass index (BMI), other physical activity, watching TV and Mediterranean Diet Index]. Body weight and height were obtained with participants barefoot and wearing underclothes. Weight was measured with an electronic scale (SECA 861, Hamburg, Germany), and height (cm) with a stadiometer (SECA 225, Hamburg, Germany). Both measurements were performed twice and averages were used. BMI was expressed as kg/m2. Physical activity and watching TV were assessed by the Youth Activity Profile-Spain (YAP-S) questionnaire [27] and Mediterranean Diet Index by the KIDMED questionnaire [28].
Statistical analysis
All analyses were performed using the Statistical Package for Social Sciences (IBM SPSS Statistics for Windows, version 23.0) and the level of significance was set to 0.05. Characteristics of the study sample are presented as means and standard deviations (SD) or percentages, after checking for normality using Kolmogorov–Smirnov test. Multiple linear regression analyses adjusting for basic confounders (i.e., sex, PHV and parental education) are presented as standardized β coefficients (95% confidence interval, P). First, we studied the associations between individual early morning patterns (i.e., active commuting to school, physical activity before school, having breakfast and good sleep) as independent variables and global white matter microstructure indicators (i.e., global FA, global MD, global RD and global AD) as dependent variables. Second, we categorized the data as physically active early morning patterns (i.e., sum of active commuting to school and physical activity before school, and operationalized as 0 physically active patterns, 1 pattern or 2 patterns) and non-physically active early morning patterns (i.e., sum of having breakfast and good sleep, and operationalized as 0–1 non-physically active patterns or 2 patterns, since only 3 participants had 0 patterns), to examine the associations of combined physically active and non-physically active early morning patterns with global white matter microstructure indicators. Third, to determine whether the relationship with white matter microstructure was global or restricted to a particular set of white matter bundles, associations with white FA, MD, RD, and AD within individual tracts were also tested if exposures showed an association with global DTI metrics.
In addition, a number of sensitivity analyses were run: (i) basic significant models were additionally adjusted for BMI, physical activity, watching TV or Mediterranean diet, (ii) non-physically active early morning pattern models were repeated excluding those with 0 patterns, and (iii) including both physically active and non-physically active early morning patterns in the same regression model; that is, when the physically active early morning pattern variable was modelled as the main exposure, the analysis was also adjusted for the non-physically active early morning pattern variable, and when the non-physically active early morning pattern variable was modelled as the main exposure, the analysis was also adjusted for the physically active early morning pattern variable.
Finally, the associations of global white matter microstructure indicators as independent variables with mental health indicators as dependent variables, adjusting for basic confounders (i.e., sex, PHV and parental education), were performed if early morning patterns showed an association with global DTI metrics. Similarly, associations of FA, MD, RD, and AD within individual tracts with mental health indicators were also tested for those mental health indicators associated with global DTI metrics. Main analyses were corrected for multiple comparisons using the Benjamini–Hochberg method based on each DTI metric (30 comparisons for FA, 14 for MD, 30 for RD and 14 for AD) [55].
Results
The descriptive characteristics of the study sample are shown in Table 1. A total of 103 children (42 girls, mean age 10 ± 1.1 years) with mean BMI of 26.7 ± 3.6 kg/m2 were included in this study. The descriptive values for global and tract-specific white matter microstructure indicators are shown in Table S1.
Individual early morning patterns and global white matter microstructure
The associations of individual early morning patterns with global white matter microstructure indicators are shown in Table 2. Overall, no associations were found between individual morning patterns and global white matter microstructure indicators (all P > 0.05).
Combined physically active and non-physically active early morning patterns with global white microstructure
Physically active morning patterns were associated with greater global FA (β = 0.298, P = 0.013) and lesser global RD (β = − 0.272, P = 0.021), but not associated with global MD (P = 0.054) or global AD (P = 0.524). No association was found between non-physically active early morning patterns and global indicators (all P > 0.05) (Table 3). In sensitivity analyses, the association between non-physically active early morning patterns with global white matter microstructure indicators was studied excluding those with 0 patterns (n = 3) and results were similar (Table S2). Results remained similar when basic significant models were additionally adjusted for BMI, physical activity, watching TV, or Mediterranean diet (Table S3). In addition, when we included both physically active and non-physically active early morning patterns in the same regression model, results were virtually the same (data no shown).
Combined physically active early morning patterns with tract-specific white matter microstructure
Associations of combined physically active early morning patterns with tract-specific white matter microstructure indicators are shown in Table 4. Compared to those with 0 physically active early morning patterns, those with 2 physically active early morning patterns had higher FA (β = 0.314, P = 0.004) and lower RD (β = − 0.234, P = 0.032) in the SLF. Results were similar when separately run the associations for left and right SLF FA (left SLF: β = 0.367, P = 0.002; right SLF: β = 0.374, P = 0.002) and RD (left SLF: β =− 0.327, P = 0.006; right SLF: β =− 0.361, P = 0.003) indicators. Results were virtually the same when basic significant models were additionally adjusted for BMI, physical activity, watching TV or Mediterranean diet (Table S4). No other associations were found with white matter metrics (i.e., FA, and RD) in any other tract (P > 0.05).
Combined physically active early morning pattern-associated global and tract-specific white matter indicators with mental health
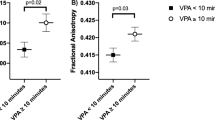
The associations of mental health indicators with that of the combined physically active early morning patterns that were associated with global white matter indicators are shown in Table 5. Global FA was positively associated with happiness (β = 0.252, P = 0.022). In addition, global RD (β = − 0.298, P = 0.008) was negatively associated with happiness. No other associations were found between global white matter microstructure indicators and mental health outcomes (P > 0.05). Finally, FA (β = 0.207, P = 0.045) and RD (β = − 0.256, P = 0.013) in the SLF was related to happiness (Fig. 1).
Associations of tract-specific white matter microstructure indicators with mental health indicators. Panel A Fractional anisotropy (FA); Panel B Radial Diffusivity (RD). β values are standardized regression coefficients. Analyses were adjusted for sex, peak height velocity (year), and parent education university level (neither/one/both). Superior longitudinal fasciculus was selected, since it was the tract previously associated with active morning patterns. *Statistically significant values surpassed multiple comparisons using the Benjamini and Hochberg method
Discussion
Individual early morning patterns or combined non-physically active early morning patterns were not associated with white matter microstructure indicators. However, our findings suggest that a combination of physically active early morning activities, particularly active commuting to school and physical activity before school, is related to global and SLF tract-specific white matter indicators (FA, RD); and in turn, those white matter indicators are related to happiness. Therefore, these novel results suggest synergistic effects of physically active early morning patterns on white matter microstructure, coupled with better happiness in children with overweight or obesity.
Previous studies examined the associations between individual lifestyle behaviors (e.g., physical activity, sleep or diet) and white matter microstructure in children [6,7,8,9]. For example, a larger study in normal-weight children found that total physical activity (i.e., outdoor playing and sport participation) was associated with improved white matter indicators (i.e., higher global FA and lower global MD); however, active commuting, including both active commuting to (during mornings) and from (during afternoon) school, was not associated with white matter [8]. Another study using the present sample of children with overweight or obesity showed that total physical activity was related to greater white matter microstructure (i.e., global FA) [9]. In addition, non-physically active behaviors (having breakfast or good sleep) may also influence white matter [6, 9, 14]. Specifically, breakfast staple type was differentially associated with white matter volume in children [14] and sleep disturbances were related to less white matter microstructural integrity (i.e., lower FA) [9]. In contrast, we found that individual early morning patterns (i.e., active commuting to school, physical activity before school, having breakfast and good sleep) were not associated with white matter microstructure in children with overweight or obesity. These discrepancies between studies may be due to: (i) characteristics of the sample (i.e., normal-weight children vs. children with overweight/obesity), since overweight/obese children may adhere to unhealthier behaviors and show differential white matter development compared with their normal-weight peers [56,57,58], and (ii) methodological differences, since early morning patterns were assessed using different self-reported questionaries across studies. Thus, future studies are needed to understand the individual associations of early morning patterns and white matter microstructure using standardized methodologies and comparing the whole range of body mass index distribution in children.
Importantly, we found that the combination of non-physically active early morning patterns (i.e., having breakfast and good sleep) was not significantly related to white matter indicators; however, the combination of physically active early morning patterns was associated with white matter microstructure. Specifically, compared to those having 0 active patterns, those performing both patterns, active commuting to school and physical activity before school, had greater white matter microstructure. Importantly, these results were independent of other factors not occurring in the morning, such as other physical activity, watching TV, BMI or Mediterranean diet index. Therefore, these results highlight the possible importance of engaging physical activity in the morning, and walking or cycling to school for white matter microstructure in childhood. Although identifying time-dependent biological mechanisms is beyond the scope of this study, we can speculate about several potential reasons underlying the morning activity-white matter associations. Biological processes (e.g., metabolism, hormone production, immunity, or behavior) are orchestrated by circadian clocks, and temporal gating of those processes is essential for maintaining homeostasis. Particularly, timing of physical activity can entrain circadian control of systemic energy homeostasis and behavioral activity [59, 60]. For example, a study in mice found that physical activity timing is a critical factor to amplify the benefits of exercise on metabolic pathways in skeletal muscle and systemic energy homeostasis; specifically decreasing muscle and blood glucose levels only after early morning activity [59]. These physiological adaptations, might in turn, influence brain tissue, by secreted exerkines crossing the blood–brain barrier [61]. Indeed, a study in mice found that kynurenine was increased only after morning physical activity [60]; kynurenine directly targets important neurotransmitter receptors and affects redox processes, and thus likely influences brain physiology [62]. Likewise, ketones such as beta-hydroxybutyrate were remarkably increased by morning physical activity, indicating a greater reliance on fatty acid oxidation and increased buffering against metabolic stress, which may exert neuroprotective effects [63]. Finally, hypothalamic neurotransmitters serotonin, dopamine, and its catabolite homovanillate only increase by morning physical activity [61]. Thus, based on animal models, a time-dependent impact of physical activity on the human brain seems to be biologically plausible, albeit it is important to highlight that exercise physiology and circadian rhythms are highly variable in humans and animals. Future longitudinal studies and clinical trials in humans are needed to confirm the synergistic- and early morning-specific effects of active behaviors on white matter microstructure, as well as its potential underlying mechanisms.
Another interesting finding was that in addition to the associations with global white matter microstructure, there was a tract-specific association between a combination of physically active early morning patterns and white matter microstructure in the SLF. The SLF connects parietal, temporal and occipital regions to the frontal lobe. This tract is involved in supporting different functions including processing of visual spatial information, decision-making and emotional regulation [64,65,66]. In particular, studies in children have associated the SLF with several different processes and functions [5, 67, 68]. For instance, Bruckert et al. [67] found an association between SLF in both hemispheres with later reading in children and Brandes-Aitken et al. [68] observed an association with both visuomotor control and cognitive control. Moreover, a large population-based study of 10-year-old children showed that improved white matter microstructure was associated with lower general psychopathology, which in turn was related to happiness [5]. Interestingly, in the present study, we found that both global and tract-specific (i.e., SLF) FA and RD were related to happiness, but not with any other mental health indicator, suggesting a specific role of white matter of the SLF with happiness during childhood. In this line, while no previous studies in children have shown a direct white matter–happiness association, some recent investigations in adults examined this hypothesis [69, 70]. For example, a study in 417 healthy Japanese adults found that FA of several tracts, including SLF, was related with happiness assessed also by the SHS questionnaire [69]. Additionally, a meta-analysis including 37 studies found disruptions in the SLF as the most common white matter alteration in adults suffering from psychiatric emotional conditions such as bipolar disorder, social anxiety disorder, major depressive disorder, post-traumatic stress disorder and obsessive–compulsive disorder [71]. Hence, the SLF, although classically associated with spatial functions [72], may have an emotional subcomponent. However, future longitudinal studies with larger sample sizes should examine the global and tract-specific influence of white matter microstructure on mental health indicators in children.
Finally, several limitations require discussion. First, the cross-sectional design does not allow inferences about causality to any of the associated factors. Second, the sample with overweight and obesity limits the generalizability of our findings to the entire range of the BMI distribution. Third, the voxel size was a nonisotropic 4-mm-section voxel (1.8 × 1.8 × 4 mm3). Therefore, FA could be underestimated in regions containing crossing fibers (i.e., SLF). On the other hand, the FA measured in regions without crossing fibers (i.e., CST) is not prone to underestimation. Finally, our study relied on self-reported, while validated, measures to assess early morning patterns. On the other hand, the study has several strengths, including the relatively large sample with neuroimaging data, the analyses of both global and tract-specific white matter indicators, and the assessment of a broad mental health battery.
Conclusion
Our results suggest that individual early morning patterns were not associated with white matter microstructure in children with overweight or obesity. In addition, while combined non-physically active early morning patterns may not be related to white matter indicators, the combination of the two physically active early morning patterns (i.e., active commuting and physical activity before school) may positively relate to global and SLF tract-specific white matter indicators (FA, RD), and in turn, happiness. Yet, these results need to be confirmed in randomized controlled trials.
From a public health perspective, implementing an early morning exercise intervention may generate healthier brains and happier children. Educational and health institutions should target before physical activity school programs as well as promote strategies to walk or cycle to school during childhood to improve brain and mental health.
Data availability
The dataset supporting the conclusions of this article is available upon reasonable request, by emailing to the corresponding author.
References
Lenroot RK, Giedd JN (2006) Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neurosci Biobehav Rev 30(6):718–729
Schmithorst VJ, Yuan W (2010) White matter development during adolescence as shown by diffusion MRI. Brain Cogn 72(1):16–25
Schmithorst VJ, Wilke M, Dardzinski BJ, Holland SK (2005) Cognitive functions correlate with white matter architecture in a normal pediatric population: a diffusion tensor MRI study. Hum Brain Mapp 26(2):139–147
Muetzel RL, Blanken LME, van der Ende J, El Marroun H, Shaw P, Sudre G et al (2018) Tracking brain development and dimensional psychiatric symptoms in children: a longitudinal population-based neuroimaging study. Am J Psychiatry 175(1):54–62
Neumann A, Muetzel RL, Lahey BB, Bakermans-Kranenburg MJ, van Ijzendoorn MH, Jaddoe VW et al (2020) White matter microstructure and the general psychopathology factor in children. J Am Acad Child Adolesc Psychiatry. https://doi.org/10.1016/j.jaac.2019.12.006
Taki Y, Kawashima R (2012) Brain development in childhood. Open Neuroimaging J 6:103–110
Rodriguez-Ayllon M, Esteban-Cornejo I, Verdejo-Roman J, Muetzel RL, Migueles JH, Mora-Gonzalez J et al (2020) Physical activity, sedentary behavior, and white matter microstructure in children with overweight or obesity. Med Sci Sports Exerc 52(5):1218–1226
Rodriguez-Ayllon M, Derks IPM, van den Dries MA, Esteban-Cornejo I, Labrecque JA, Yang-Huang J et al (2020) Associations of physical activity and screen time with white matter microstructure in children from the general population. Neuroimage 205:116258
Mulder TA, Kocevska D, Muetzel RL, Koopman-Verhoeff ME, Hillegers MH, White T et al (2019) Childhood sleep disturbances and white matter microstructure in preadolescence. J Child Psychol Psychiatry 60(11):1242–1250
Thapa N, Kim B, Yang JG, Park HJ, Jang M, Son HE et al (2020) The relationship between chronotype, physical activity and the estimated risk of dementia in community-dwelling older adults. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph17103701
Qian J, Walkup MP, Chen SH, Brubaker PH, Bond DS, Richey PA et al (2021) Association of objectively measured timing of physical activity bouts with cardiovascular health in type 2 diabetes. Diabetes Care 44(4):1046–1054
Chomistek AK, Shiroma EJ, Lee IM (2016) The relationship between time of day of physical activity and obesity in older women. J Phys Act Health 13(4):416–418
Albalak G, Stijntjes M, van Bodegom D, Jukema JW, Atsma DE, van Heemst D et al (2022) Setting your clock: associations between timing of objective physical activity and cardiovascular disease risk in the general population. Eur J Prev Cardiol. https://doi.org/10.1093/eurjpc/zwac239
Taki Y, Hashizume H, Sassa Y, Takeuchi H, Asano M, Asano K et al (2010) Breakfast staple types affect brain gray matter volume and cognitive function in healthy children. PLoS ONE 5(12):e15213
Prochaska JO (2008) Multiple Health Behavior Research represents the future of preventive medicine. Prev Med 46(3):281–285
Davis JS, Banfield E, Lee HY, Peng HL, Chang S, Wood AC (2019) Lifestyle behavior patterns and mortality among adults in the NHANES 1988–1994 population: a latent profile analysis. Prev Med 120:131–139
Kullmann S, Schweizer F, Veit R, Fritsche A, Preissl H (2015) Compromised white matter integrity in obesity. Obes Rev 16(4):273–281
van Bloemendaal L, Ijzerman RG, Ten Kulve JS, Barkhof F, Diamant M, Veltman DJ et al (2016) Alterations in white matter volume and integrity in obesity and type 2 diabetes. Metab Brain Dis 31(3):621–629
Koivukangas J, Bjornholm L, Tervonen O, Miettunen J, Nordstrom T, Kiviniemi V et al (2016) Body mass index and brain white matter structure in young adults at risk for psychosis—the Oulu Brain and Mind Study. Psychiatry Res Neuroimaging 254:169–176
Augustijn M, Deconinck FJA, D’Hondt E, Van Acker L, De Guchtenaere A, Lenoir M et al (2018) Reduced motor competence in children with obesity is associated with structural differences in the cerebellar peduncles. Brain Imaging Behav 12(4):1000–1010
Lindberg L, Hagman E, Danielsson P, Marcus C, Persson M (2020) Anxiety and depression in children and adolescents with obesity: a nationwide study in Sweden. BMC Med 18(1):30
Cadenas-Sanchez C, Mora-Gonzalez J, Migueles JH, Martin-Matillas M, Gomez-Vida J, Escolano-Margarit MV et al (2016) An exercise-based randomized controlled trial on brain, cognition, physical health and mental health in overweight/obese children (ActiveBrains project): rationale, design and methods. Contemp Clin Trials 47:315–324
Cole TJ, Lobstein T (2012) Extended international (IOTF) body mass index cut-offs for thinness, overweight and obesity. Pediatr Obes 7(4):284–294
Bervoets L, Massa G (2014) Defining morbid obesity in children based on BMI 40 at age 18 using the extended international (IOTF) cut-offs. Pediatr Obes 9(5):e94–e98
Saint-Maurice PF, Kim Y, Hibbing P, Oh AY, Perna FM, Welk GJ (2017) Calibration and validation of the youth activity profile: the FLASHE Study. Am J Prev Med 52(6):880–887
Saint-Maurice PF, Welk GJ (2015) Validity and calibration of the youth activity profile. PLoS ONE 10(12):e0143949
Segura-Díaz JM, Barranco-Ruiz Y, Saucedo-Araujo RG, Aranda-Balboa MJ, Cadenas-Sanchez C, Migueles JH et al (2021) Feasibility and reliability of the Spanish version of the Youth Activity Profile questionnaire (YAP-Spain) in children and adolescents. J Sports Sci 39(7):801–807
Serra-Majem L, Ribas L, Ngo J, Ortega RM, García A, Pérez-Rodrigo C et al (2004) Food, youth and the Mediterranean diet in Spain. Development of KIDMED, Mediterranean diet quality index in children and adolescents. Public Health Nutr 7(7):931–935
Serra-Majem L, Ribas L, García A, Pérez-Rodrigo C, Aranceta J (2003) Nutrient adequacy and Mediterranean Diet in Spanish school children and adolescents. Eur J Clin Nutr 57(Suppl 1):S35–S39
Lazarou C, Panagiotakos DB, Matalas AL (2009) Level of adherence to the Mediterranean diet among children from Cyprus: the CYKIDS study. Public Health Nutr 12(7):991–1000
Esteban-Cornejo I, Izquierdo-Gomez R, Gomez-Martinez S, Padilla-Moledo C, Castro-Pinero J, Marcos A et al (2016) Adherence to the Mediterranean diet and academic performance in youth: the UP&DOWN study. Eur J Nutr 55(3):1133–1140
Adelantado-Renau M, Beltran-Valls MR, Esteban-Cornejo I, Martínez-Vizcaíno V, Santaliestra-Pasías AM, Moliner-Urdiales D (2019) The influence of adherence to the Mediterranean diet on academic performance is mediated by sleep quality in adolescents. Acta Paediatr (Oslo, Norway : 1992) 108(2):339–346
Tapia-Serrano MA, Esteban-Cornejo I, Rodriguez-Ayllon M, Vaquero-Solís M, Sánchez-Oliva D, Sánchez-Miguel PA (2021) Adherence to the Mediterranean diet and academic performance in adolescents: does BMI status moderate this association? Clin Nutr (Edinburgh, Scotland) 40(6):4465–4472
Tomás Vila M, Miralles Torres A, Soto BB (2007) Spanish version of the Pediatric Sleep Questionnaire (PSQ). A useful instrument in investigation of sleep disturbances in childhood. Reliability analysis. An Pediatr 66(2):121–128
Hirshkowitz M, Whiton K, Albert SM, Alessi C, Bruni O, DonCarlos L et al (2015) National Sleep Foundation’s sleep time duration recommendations: methodology and results summary. Sleep Health 1(1):40–43
Furber G, Segal L (2015) The validity of the Child Health Utility instrument (CHU9D) as a routine outcome measure for use in child and adolescent mental health services. Health Qual Life Outcomes 13:22
Metwalli NS, Benatar M, Nair G, Usher S, Hu X, Carew JD (2010) Utility of axial and radial diffusivity from diffusion tensor MRI as markers of neurodegeneration in amyotrophic lateral sclerosis. Brain Res 1348:156–164
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM (2012) FSL. Neuroimage 62(2):782–790
Behrens TE, Berg HJ, Jbabdi S, Rushworth MF, Woolrich MW (2007) Probabilistic diffusion tractography with multiple fibre orientations: What can we gain? Neuroimage 34(1):144–155
de Groot M, Ikram MA, Akoudad S, Krestin GP, Hofman A, van der Lugt A et al (2015) Tract-specific white matter degeneration in aging: the Rotterdam Study. Alzheimer’s Dement 11(3):321–330
Rodriguez-Ayllon M, Cadenas-Sanchez C, Esteban-Cornejo I, Migueles JH, Mora-Gonzalez J, Henriksson P et al (2018) Physical fitness and psychological health in overweight/obese children: a cross-sectional study from the ActiveBrains project. J Sci Med Sport Sports Med Aust 21(2):179–184
Lyubomirsky S, Lepper HS (1999) A measure of subjective happiness: preliminary reliability and construct validation. Soc Indic Res 46(2):137–155
Gómez-Lugo M, Espada JP, Morales A, Marchal-Bertrand L, Soler F, Vallejo-Medina P (2016) Adaptation, validation, reliability and factorial equivalence of the rosenberg self-esteem scale in colombian and spanish population. Span J Psychol 19:E66
Ey S, Hadley W, Allen DN, Palmer S, Klosky J, Deptula D et al (2005) A new measure of children’s optimism and pessimism: the youth life orientation test. J Child Psychol Psychiatry 46(5):548–558
Trianes Torres MV, Blanca Mena MJ, Fernández Baena FJ, Escobar Espejo M, Maldonado Montero EF, Muñoz Sánchez AM (2009) Assessment of stress in childhood: Children’s Daily Stress Inventory (Inventario Infantil de Estresores Cotidiano, IIEC). Psicothema 21(4):598–603
Knight D, Hensley VR, Waters B (1988) Validation of the children’s depression scale and the children’s depression inventory in a prepubertal sample. J Child Psychol Psychiatry 29(6):853–863
Rodrigo G, Lusiardo M (1989) Spanish version of the revised children’s manifest anxiety scale in uruguay: reliability and concurrent validity. Psychol Rep 65(1):94
Laurent J, Catanzaro SJ, Joiner TE Jr, Rudolph KD, Potter KI, Lambert S et al (1999) A measure of positive and negative affect for children: scale development and preliminary validation. Psychol Assess 11:326–338
Esteban-Cornejo I, Cadenas-Sanchez C, Contreras-Rodriguez O, Verdejo-Roman J, Mora-Gonzalez J, Migueles JH et al (2017) A whole brain volumetric approach in overweight/obese children: examining the association with different physical fitness components and academic performance. The ActiveBrains project. Neuroimage 159:346–354
Esteban-Cornejo I, Mora-Gonzalez J, Cadenas-Sanchez C, Contreras-Rodriguez O, Verdejo-Roman J, Henriksson P et al (2019) Fitness, cortical thickness and surface area in overweight/obese children: the mediating role of body composition and relationship with intelligence. Neuroimage 186:771–781
Esteban-Cornejo I, Rodriguez-Ayllon M, Verdejo-Roman J, Cadenas-Sanchez C, Mora-Gonzalez J, Chaddock-Heyman L et al (2019) Physical fitness, white matter volume and academic performance in children: findings from the ActiveBrains and FITKids2 projects. Front Psychol 10:208
Rodriguez-Ayllon M, Esteban-Cornejo I, Verdejo-Román J, Muetzel RL, Mora-Gonzalez J, Cadenas-Sanchez C et al (2020) Physical fitness and white matter microstructure in children with overweight or obesity: the ActiveBrains project. Sci Rep 10(1):12469
Malina RM, Rogol AD, Cumming SP, Coelho e Silva MJ, Figueiredo AJ (2015) Biological maturation of youth athletes: assessment and implications. Br J Sports Med 49(13):852–859
Moore SA, McKay HA, Macdonald H, Nettlefold L, Baxter-Jones AD, Cameron N et al (2015) Enhancing a somatic maturity prediction model. Med Sci Sports Exerc 47(8):1755–1764
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc Ser B (Methodol) 57(1):289–300
Croce CM, Fisher JO, Coffman DL, Bailey RL, Davey A, Tripicchio GL (2022) Association of weight status with the types of foods consumed at snacking occasions among US adolescents. Obesity (Silver Spring, Md). https://doi.org/10.1093/cdn/nzab055_017
Xiu L, Hagströmer M, Bergqvist-Norén L, Johansson E, Ekbom K, Svensson V et al (2019) Development of sleep patterns in children with obese and normal-weight parents. J Paediatr Child Health 55(7):809–818
Ou X, Andres A, Pivik RT, Cleves MA, Badger TM (2015) Brain gray and white matter differences in healthy normal weight and obese children. J Magn Reson Imaging 42(5):1205–1213
Sato S, Basse AL, Schönke M, Chen S, Samad M, Altıntaş A et al (2019) Time of exercise specifies the impact on muscle metabolic pathways and systemic energy homeostasis. Cell Metab 30(1):92-110.e4
Sato S, Dyar KA, Treebak JT, Jepsen SL, Ehrlich AM, Ashcroft SP et al (2022) Atlas of exercise metabolism reveals time-dependent signatures of metabolic homeostasis. Cell Metab 34(2):329–45.e8
Chow LS, Gerszten RE, Taylor JM, Pedersen BK, van Praag H, Trappe S et al (2022) Exerkines in health, resilience and disease. Nat Rev Endocrinol 18(5):273–289
Schwarcz R, Bruno JP, Muchowski PJ, Wu HQ (2012) Kynurenines in the mammalian brain: when physiology meets pathology. Nat Rev Neurosci 13(7):465–477
Sleiman SF, Henry J, Al-Haddad R, El Hayek L, Abou Haidar E, Stringer T et al (2016) Exercise promotes the expression of brain derived neurotrophic factor (BDNF) through the action of the ketone body beta-hydroxybutyrate. Elife. https://doi.org/10.7554/eLife.15092
Siehl S, King JA, Burgess N, Flor H, Nees F (2018) Structural white matter changes in adults and children with posttraumatic stress disorder: a systematic review and meta-analysis. NeuroImage Clin 19:581–598
Pisner DA, Shumake J, Beevers CG, Schnyer DM (2019) The superior longitudinal fasciculus and its functional triple-network mechanisms in brooding. NeuroImage Clinical 24:101935
Bolhuis K, Muetzel RL, Stringaris A, Hudziak JJ, Jaddoe VWV, Hillegers MHJ et al (2019) Structural brain connectivity in childhood disruptive behavior problems: a multidimensional approach. Biol Psychiat 85(4):336–344
Bruckert L, Borchers LR, Dodson CK, Marchman VA, Travis KE, Ben-Shachar M et al (2019) White matter plasticity in reading-related pathways differs in children born preterm and at term: a longitudinal analysis. Front Hum Neurosci 13:139
Brandes-Aitken A, Anguera JA, Chang YS, Demopoulos C, Owen JP, Gazzaley A et al (2018) White matter microstructure associations of cognitive and visuomotor control in children: a sensory processing perspective. Front Integr Neurosci 12:65
Kokubun K, Nemoto K, Yamakawa Y (2022) Brain conditions mediate the association between aging and happiness. Sci Rep 12(1):4290
Ioannucci S, George N, Friedrich P, Cerliani L, der Thiebaut Schotten M (2020) White matter correlates of hemi-face dominance in happy and sad expression. Brain Struct Funct 225(4):1379–1388
Jenkins LM, Barba A, Campbell M, Lamar M, Shankman SA, Leow AD et al (2016) Shared white matter alterations across emotional disorders: a voxel-based meta-analysis of fractional anisotropy. NeuroImage Clin 12:1022–1034
Parlatini V, Radua J, Dell’Acqua F, Leslie A, Simmons A, Murphy DG et al (2017) Functional segregation and integration within fronto-parietal networks. Neuroimage 146:367–375
Funding
Funding for open access publishing: Universidad de Granada/CBUA. This study was supported by the Spanish Ministry of Economy and Competitiveness (DEP2013-47540, BES-2014-068829, DEP2016-79512-R & PSI2012-3929, DEP2017-91544-EXP, RTI2018-095284-J-100), the Alicia Koplowitz Foundation, and for the Spanish Ministry of Science and Innovation (RYC-2011-09011 & RYC2019-027287-I). This study takes place thanks to the additional funding from the University of Granada, Plan Propio de Investigación 2016, Excellence actions: Units of Excellence; Unit of Excellence on Exercise and Health (UCEES), the Junta de Andalucía, Consejería de Conocimiento, Investigación y Universidades and European Regional Development Fund (ERDF, SOMM17/6107/UGR). Additional funding from the EXERNET Research Network on Exercise and Health in Special Populations (DEP2005-00046/ACTI, the SAMID III network, RETICS, funded by the PN I + D + I 2017-2021 (Spain), ISCIII- Sub-Directorate General for Research Assessment and Promotion and the European Regional Development Fund (ERDF) (Ref. RD16/0022, SOMM17/6107/UGR). Funding for open access charge: Universidad de Granada / CBUA. The present study was submitted to “Premios Nacionales de Investigación en Medicina del Deporte. Fundación Cajastur. Universidad de Oviedo, año 2022.”
Author information
Authors and Affiliations
Contributions
IE-C and IL-J had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design: IE-C and IL-J. Statistical analysis: IE-C, IL-J, MR-A, and JV-R. Interpretation of the data: IE-C, IL-J, and MR-A. Drafting of the manuscript: IE-C and IL-J. Critical revision of the manuscript for important intellectual content: all authors. Administrative, technical, or material support: AC and MR-A. Study supervision: AC, KE, and FBO. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors reported no biomedical financial interests or potential conflicts of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Esteban-Cornejo, I., Lara-Jimenez, I., Rodriguez-Ayllon, M. et al. Early morning physical activity is associated with healthier white matter microstructure and happier children: the ActiveBrains project. Eur Child Adolesc Psychiatry 33, 833–845 (2024). https://doi.org/10.1007/s00787-023-02197-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-023-02197-6