Abstract
Objectives
Periodontitis (PD) can cause systematic inflammation and is associated with various metabolic processes in the body. However, robust serum markers for these relationships are still lacking. This study aims to identify novel circulating inflammation-related proteins associated with PD using targeted proteomics.
Materials and methods
We used population-based, cross-sectional data from 619 participants of the Polish Longitudinal University Study (Bialystok PLUS). Mean pocket probing depth (mPPD) and proportion of bleeding on probing (pBOP) served as exposure variables. Fifty-two inflammation-related proteins were measured using the Olink Target 96 Cardiovascular III and the Olink Target 96 Immune Response panels. Associations between periodontal measures and proteins were tested using covariate-adjusted linear regression models.
Results
At a false discovery rate of < 0.05, we identified associations of mPPD and pBOP with platelet-endothelial cell adhesion molecule-1 (PECAM-1) and tripartite motif–containing protein 21 (TRIM21).
Conclusion
This study revealed novel associations between PD and serum levels of PECAM-1 and TRIM21. Our results suggest that these proteins might be affected by molecular processes that take place in the inflamed periodontium.
Clinical relevance
Novel associations of PECAM-1 and TRIM21 with PD indicate promising serum markers for understanding the disease’s pathophysiological processes and call for further biomedical investigations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Periodontitis (PD), as a complex multifactorial condition, is one of the most common chronic diseases worldwide [1]. Dysbiotic plaque biofilm is the main component initializing the pathological processes. However, extent and progression are largely determined by host-specific features such as an exuberant inflammatory immune response [2]. Previous studies suggest that the relationship between inflammation and PD is in fact mutual with inflammation contributing to periodontal damage and periodontal damage inducing inflammation, a self-sustaining vicious cycle. It is precisely the persistent low-grade inflammation that is suspected to impair overall health [3]. To this date, PD diagnostics focus on assessing the local periodontium, therapies are aimed at managing the biofilm rather than the host’s exacerbating immune system, and the analysis of blood—despite being the most widespread diagnostic procedure in medicine [4]—is rarely used in clinical dental care [5]. This is primarily owed to the lack of appropriate (serum) biomarkers [6].
An appropriate biomarker is a “characteristic that is measured as an indicator of normal biological processes, pathogenic processes, or biological responses to an exposure or intervention” [7]. The concept of using serum proteins to individualize medicine is not new; measurement of C-reactive protein, for instance, has been used to assess inflammation since the protein was discovered nearly 100 years ago [8]. But the potential of available methods has increased dramatically in the last decades [9]. In this regard, “targeted proteomics” describes a rapidly emerging, sensitive, and reliable method to quantify entire protein clusters. The analysis can thus be specifically tailored to a system or tissue under investigation [10]. Recent advances in sequencing technologies provide increasingly powerful tools to identify protein biomarkers, which, in turn, might provide the framework for a variety of future applications: For example, serum biomarkers indicating chronic inflammation in PD could improve diagnostics or help develop and monitor individualized therapies [4].
The ability to generate comprehensive protein profiles at a relatively low cost and the ongoing trend toward individualized, patient-specific medicine drive the increasing adoption of these methods in all medical fields; dental medicine is no exception [11]. So far, however, the application has been limited to studies with small numbers of participants or the investigation of PD in the context of (inflammatory) comorbidities [12,13,14,15]. In contrast, this study examined associations between PD measures and circulating levels of 52 inflammation-related serum proteins in a population-based study aiming to identify indicators of systemic PD effects.
Materials and methods
Study population
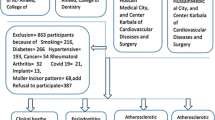
For the Polish Longitudinal University Study (Bialystok PLUS), 3246 individuals aged 20 to 80 were sampled from population registries and invited to participate in a personal interview and medical and dental examinations in 2017–2021 [16]. Proteomic analyses were performed on a randomly selected subsample consisting of 745 participants. After the exclusion of subjects with no dental measurements (n = 59) or missing values in covariates (n = 67), the analytical sample comprised 619 eligible participants. Relevant ethical approval for the Bialystok PLUS study was granted by the Ethics Committee of the Medical University of Bialystok in conformity with the Declaration of Helsinki (Ethics number: (R-i-002/108/2016), and all participants provided written informed consent.
Periodontal examination
Periodontal examination was performed by four calibrated dentists under conditions of an epidemiological inquiry (using artificial light but without saliva ejector or air jet) and included pocket probing depth (PPD), gingival recession, and bleeding on probing (BOP). Measurements were performed randomly on either the left- or right-side quadrants and included all erupted teeth except the third molars. Four sites per tooth were assessed; mesiobuccal, distobuccal, midbuccal, and midlingual or midpalatinal. A UNC15 periodontal probe was used for assessment. From these variables, collected as whole millimeters (or binary recorded concerning BOP), the variable mean PPD (mPPD) and proportion of sites with BOP (pBOP) were calculated for each participant. Furthermore, a binary variable indicating gingival health was calculated, according to the epidemiological definition adopted in the proceedings of the 2017 World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions, defined as < 10% pBOP with PPD ≤ 3 mm [17].
Proteomic analysis (OLINK Footnote 1 )
Quantification of the protein concentrations in serum was performed using the Olink Target 96 Cardiovascular III and Olink Target 96 Immune Response panel. The approach is based on the proximity extension assay. A detailed description of this method can be found in previous studies [18, 19]. Measured protein concentrations are obtained in a relative quantification unit proposed by Olink, the Normalized Protein eXpression (NPX), which is reported on a log2 scale. The increase of one NPX corresponds to a doubling of the protein concentration. Subsequent normalization reduces intra- and inter-assay variability and strengthens the comparability of measurements (both between plates and different studies). Olink provides a classification of measured biomarker proteins based on specific biological processes or disease areas. We combined all proteins of both panels that were assigned to the disease area “inflammatory,” resulting in 74 proteins. The limit of detection (LOD) for each measured protein is further calculated separately for each Olink assay and sample plate. The LOD value is estimated from the negative controls included on every plate, plus three standard deviations, and represents the minimum detectable concentration. The standard deviation is assay-specific and estimated during product validation for every panel. For studies including more than one plate per panel, the maximum observed LOD for each assay is selected as the study LOD. Consequently, all plates included in the study receive the same assay-specific LOD. Proteins for which more than 25% of the samples had values below the LOD were excluded from further analysis. This led to the exclusion of 22 of 74 proteins. For the remaining 52 proteins (a complete list is provided in Supplementary Table 1), values below the LOD were substituted with the respective LOD [19].
Covariates
Socio-demographic data (i.e., age, sex, school education) as well as self-reported diabetes were obtained from a self-administered questionnaire. For biochemical analyses (including HbA1C), peripheral venous fasting blood was collected in the morning on a visit day, and serum samples were stored at − 80 °C until use. BMI was calculated as weight (in kilograms) divided by the square of height (in meters). Smoking was grouped into never, former, and current smokers. Self-reported alcohol intake was inquired as days on which alcohol was consumed combined with the number of beverage-specific milliliters. The quantity information was used to calculate the amount of alcohol consumed in the last 30 days in grams [20]. For representation purposes in Table 1, the gram per day value is shown. Physical activity was assessed using the International Physical Activity Questionnaire—long format (IPAQ-L) [21]. All IPAQ data was processed using the standardized IPAQ scoring protocol and used as a continuous variable expressed in metabolic equivalent of task (MET) minutes per week. All daily walking, moderate, and vigorous activity time variables were truncated to 3 h, allowing a maximum of 21 h of activity in each category per week. The categorical IPAQ variable was only used to report physical activity in Table 1 and was also built using the IPAQ scoring protocol.
Statistical analysis
The associations of pBOP and mPPD with proteins were analyzed using multivariable linear regression models. Each regression model was adjusted for age, sex, smoking, alcohol intake, and physical activity. Continuous covariates were modeled using restricted cubic splines with three knots at fixed quantiles (0.1, 0.5, 0.9) of the distribution [22]. We computed the Benjamini-Hochberg false discovery rate (FDR) on the resulting p-values [23]. In a sensitivity analysis, we repeated the primary assessment (by using the adopted definition of gingival health in a third regression model while adjusting the FDR accordingly) to evaluate the relationship between gingival health and proteins. Furthermore, we tested the robustness of our findings in a surrogate analysis by including diabetes and BMI as potential confounders. Analyses were performed using the rms (6.2-0), stats (4.0.5), and ggplot2 (3.3.5) packages in R version 4.0.5 (The R Foundation for Statistical Computing).
Results
Among the 619 included study participants, the average age was 48 years, 55% were female, and about 1/5 reported current smoking. Periodontal measurements yielded an average of 1.8 mm for the mPPD and a pBOP of 15%. Compared to the full data, the exclusion of individuals with missing values hardly changed the study characteristics with a minimal tendency to a slightly younger and healthier population. The corresponding values (both before and after exclusions) can be found in Table 1.
Figure 1 presents the FDR adjusted results from multiple linear regressions. At a FDR < 0.05, we found that periodontal measures were associated with two proteins. mPPD and pBOP were positively associated with platelet-endothelial cell adhesion molecule-1 (PECAM-1). In contrast, mPPD and pBOP were inversely associated with tripartite motif–containing protein 21 (TRIM21). FDR adjusted p-values for these associations as well as for five additional proteins, that showed associations with p < 0.05 without adjustment, are listed in Supplementary Table 2 and 3. The surrogate analysis (Fig. 2) reveals a concordant relation—since in this case, the event is being healthy—between gingival health and TRIM21 as well as a positive link with interleukin-1 receptor-associated kinase 1 (IRAK1). The FDR adjusted p-values of this analysis can be found in Supplementary Table 4. Supplementary Table 5 provides the full set of regression results, including regression coefficients, 95% confidence intervals, and both adjusted and unadjusted p-values. The surrogate analysis, in which diabetes and BMI were included as additional confounders, mirrored the results of our primary analysis (Supplementary Figure 1).
Volcano plots (results from multiple linear regressions) showing results from multiple linear regressions for proportion of bleeding on probing (pBOP) and mean pocket probing depth (mPPD) as exposures; FDR, false discovery rate; dashed line, threshold for FDR adjusted p-value = 0.05; protein names are shown when unadjusted p-value ≤ 0.05; linear regression models adjusted for age, sex, smoking, alcohol intake, and physical activity; β-coefficients interpretable as change in normalized protein expression (NPX) per unit change in pBOP or mPPD; a list with complete protein names is provided in Supplementary Table 1
Volcano plot of surrogate analysis (results from multiple linear regressions) showing results from multiple linear regressions for gingival health according to the 2017 World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions [17] as exposure; FDR, false discovery rate; dashed line, threshold for FDR adjusted p-value = 0.05; protein names are shown if unadjusted p-value ≤ 0.05; linear regression models adjusted for age, sex, smoking, alcohol intake, and physical activity; β-coefficients interpretable as the change in normalized protein expression (NPX) when categorized as being a case of gingival health; a list with complete protein names is provided in Supplementary Table 1
Discussion
This explorative study associated 52 serum proteins with two distinct clinical markers for PD, pBOP and mPPD, revealing associations with PECAM-1 and TRIM21. The revealed associations were consistent for both exposures. The association with TRIM21 was also reflected in the surrogate analysis of the epidemiological case definition of gingival health.
PECAM-1 and TRIM21 play multiple roles in basic inflammatory and immunological processes throughout the body (see Fig. 3).
An overview of PECAM-1 and TRIM21. Endothelial cells are, in addition to specialized junctional structures (tight junctions and adherens junctions), connected by cell-specific adhesion molecules, including platelet-endothelial cell adhesion molecule-1 (PECAM-1 also known as CD31). PECAM-1 is both a cellular adhesion and signaling receptor. Platelets and leukocytes also exhibit PECAM-1. Among its functions, this protein is involved in the control of inflammatory and immunological processes. Adhesion of leukocytes to inflamed endothelium initiates cascades that subsequently increase endothelial barrier permeability, promote angiogenesis, and enable leukocyte diapedesis [24, 25]. Tripartite motif–containing protein 21 (TRIM21 or also known as Ro52) is a cytosolic antigen receptor found in most tissues. It binds to pathogens such as viruses or bacteria that enter the cell and, in turn, are attached to antibodies. The resulting complex serves to mark the pathogens for degradation by the proteasome. In addition, TRIM21 activates the NF-κB, AP-1, and IRF signaling pathways, leading to increased production of proinflammatory cytokines [26,27,28] (illustration based on [27, 29])
For PECAM-1, we observed a positive association, stating that higher periodontal burden is accompanied by increased serum protein levels. Although the adhesion molecule is well recognized, there is a lack of periodontal research that can be compared with our study. However, a study involving 40 PD patients and 38 healthy controls did reveal a similar relationship in saliva [30]. In contrast to our results, a genetic study tried linking polymorphisms of PECAM-1 with PD by comparing 105 patients and 101 healthy controls with a mean age of 33.3 and 29.1, respectively, but failed to report a significant association [31]. However, for both studies, the limited sample size and, for the latter, the very young average age of the participants must be taken into account. Further evidence corroborating our results is provided by in vitro and animal studies. Two consecutive studies not only demonstrated a link between periodontal infection and PECAM-1 but also described a novel causative local mechanism; Porphyromonas gingivalis—a major pathogen of PD—infects endothelial cells and utilizes gingipain proteases to degrade PECAM-1, resulting in a significant local reduction of this molecule and a concomitant impairment of the vascular barrier [32, 33]. A known host response to endothelial damage, and the binding of endothelial cells by leukocytes in general (which combat the pathogens present), is increased gene expression of several chemokines and adhesion molecules, including PECAM-1 [34,35,36], which presents a possible explanation for the increased serum levels we measured. Elevated serum PECAM-1—contrasting a local reduction—has also been observed in other diseases with a compromised vascular barrier, such as multiple sclerosis [37].
To our knowledge, there are no previous studies investigating a possible linkage between TRIM21 and PD. Nevertheless, TRIM21 is a known autoantibody target (leading to a low serum protein level) in diseases like systemic lupus erythematosus or other rheumatic autoimmune diseases [38], conditions that, in turn, are associated with PD [3, 39]. Additionally, TRIM21 deficiency is associated with an increased proinflammatory cytokine response [40], and animal models show that a defect in the gene region encoding TRIM21 results in a severely impaired immune response, particularly to viruses [38]. Accordingly, TRIM21 deficiency could provide a possible explanation for the high prevalence and copy count of viruses in patients with progressive PD [41]. Future research is essential to examine how this deficit develops. However, it is conceivable that, due to the continuous bacteremia of periodontal pathogens, TRIM21 is degraded to a greater extent in the course of intracellular neutralization, resulting in declined serum levels of this protein (see Fig. 3).
IRAK1, a noteworthy association in the surrogate analysis, is part of a family of serine/threonine kinases essential in the innate immune system. It participates in Toll-like receptor (TLR) and interleukin-1 receptor signaling pathways, which are important in the immune response against pathogens [42]. Imbalances in these pathways have been linked to a variety of illnesses, such as cardiovascular and inflammatory disorders [43]—including periodontal tissue degeneration [44]. Endogenous anti-inflammatory molecules from the IRAK family help regulate the TLR signaling pathway to counter-regulate inflammatory responses. Recent research suggests that the upregulation of IRAK molecules may reduce inflammatory cytokines, but their exact role is still being investigated. However, due to the known drawbacks of using a dichotomized exposure variable, and in light of the fact that the relation could not be established in the analyses of the clinical (metric) characteristics, caution is advised when interpreting this association [45, 46].
Overall, the results of this exploratory research, in combination with previous findings on the biomarkers’ involvement in periodontal and other diseases, provide new insights into the molecular processes surrounding an inflamed periodontium.
Our study has several limitations worth mentioning. First, the study is cross-sectional. That being the case, a reliable temporal separation between exposure and outcome is not possible (risk of reverse causation), and secondly, in observational studies, there is always the risk of systematic bias (e.g., confounding), which may distort the results. We tried to attenuate bias by choosing two correlated but biologically distinct periodontal markers as exposures; pBOP is the hallmark of gingival inflammation and is presumably under greater (and distinct) influence of systemic processes than mPPD, representing the loosening of the periodontal ligament [47]. Thus, the exposures should be at least partially subject to different confounding factors and the consistent associations found for both logically argue in favor of causation. Future research should, however, consider a longitudinal study design. Third, in population data, the estimated prevalence of severe PD is roughly 10%, and the condition is characterized by intermitting episodes of active and passive progression [48]. Hence, the number of participants with a severe and active periodontal burden in our sample is rather small, which might lead to an underestimation of the systemic effects of PD. We, therefore, recommend the evaluation of a more extensive sample and/or a larger number of cases exhibiting acute periodontal lesions. Fourth, participants originated from a single region, which is a potential source of unobserved bias, such as common genetics, and it is unclear whether the results are generalizable. Thus, it is recommended to verify the results in other locations.
Conclusion
In recent decades, it has become widely accepted that PD is a condition that has not only systemic causes but also systemic effects [3]. However, the biological basis of many observed associations is still unclear, and the direction of the cause-effect relationship is highly controversial. The identification of suitable biomarkers is therefore of great importance. In this context, our study highlights two possible and biologically plausible candidates whose closer investigation may provide new insights into the systemic processes surrounding PD. We suggest further investigation to replicate the retrieved associations of PECAM-1 and TRIM21, if possible, in a longitudinal design with a high number of cases of acute and severe PD.
Data Availability
The Medical University of Bialystok, the implementing body of the Bialystok PLUS study under the direction of Professor Karol A. Kamiński, is the legal owner of all used data. Data from the Bialystok PLUS study are available after data application and the signature of a data transfer agreement with the responsible local authorities.
Notes
Olink Proteomics, Uppsala, Sweden
References
Vos T, Lim SS, Abbafati C et al (2020) Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1204–1222. https://doi.org/10.1016/S0140-6736(20)30925-9
Papapanou PN, Sanz M, Buduneli N et al (2018) Periodontitis: consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Clin Periodontol 45(Suppl 20):S162–S170. https://doi.org/10.1111/jcpe.12946
Hajishengallis G (2000) Interconnection of periodontal disease and comorbidities: evidence, mechanisms, and implications. Periodontol 2000 89:9–18. https://doi.org/10.1111/prd.12430
Geyer PE, Holdt LM, Teupser D et al (2017) Revisiting biomarker discovery by plasma proteomics. Mol Syst Biol 13:942. https://doi.org/10.15252/msb.20156297
Sanz M, Herrera D, Kebschull M et al (2020) Treatment of stage I-III periodontitis-the EFP S3 level clinical practice guideline. J Clin Periodontol 47(Suppl 22):4–60. https://doi.org/10.1111/jcpe.13290
Tonetti MS, Greenwell H, Kornman KS (2018) Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J Clin Periodontol 45(Suppl 20):S149–S161. https://doi.org/10.1111/jcpe.12945
FDA-NIH Biomarker Working Group (2016) BEST (Biomarkers, EndpointS, and other Tools) Resource [Internet]. Silver Spring (MD): Food and drug administration (US); FDA-NIH biomarker working group. 2016 Jan 28 [Updated 2021 Jan 25]. Co-published by national institutes of health (US), Bethesda (MD)
Ansar W, Ghosh S (eds) (2016) Biology of C reactive protein in health and disease. Springer India, New Delhi
Collins FS, Varmus H (2015) A new initiative on precision medicine. N Engl J Med 372:793–795. https://doi.org/10.1056/NEJMp1500523
Borràs E, Sabidó E (2017) What is targeted proteomics? A concise revision of targeted acquisition and targeted data analysis in mass spectrometry. Proteomics 17. https://doi.org/10.1002/pmic.201700180
Correa Rojo A, Heylen D, Aerts J et al (2021) Towards building a quantitative proteomics toolbox in precision medicine: a mini-review. Front Physiol 12:723510. https://doi.org/10.3389/fphys.2021.723510
Panezai J, Ghaffar A, Altamash M et al (2022) Periodontal disease augments cardiovascular disease risk biomarkers in rheumatoid arthritis. Biomedicines 10. https://doi.org/10.3390/biomedicines10030714
Panezai J, Altamash M, Engstrӧm P-E et al (2020) Association of glycated proteins with inflammatory proteins and periodontal disease parameters. J Diabetes Res 2020:6450742. https://doi.org/10.1155/2020/6450742
Panezai J, Ghaffar A, Altamash M et al (2018) Periodontal disease influences osteoclastogenic bone markers in subjects with and without rheumatoid arthritis. PLoS One 13:e0197235. https://doi.org/10.1371/journal.pone.0197235
Børsting T, Venkatraman V, Fagerhaug TN et al (2021) Systematic assessment of salivary inflammatory markers and dental caries in children: an exploratory study. Acta Odontol Scand:1–8. https://doi.org/10.1080/00016357.2021.2011400
Ponce-de-Leon M, Linseisen J, Peters A et al (2022) Novel associations between inflammation-related proteins and adiposity: a targeted proteomics approach across four population-based studies. Transl Res 242:93–104. https://doi.org/10.1016/j.trsl.2021.11.004
Chapple ILC, Mealey BL, van Dyke TE et al (2018) Periodontal health and gingival diseases and conditions on an intact and a reduced periodontium: consensus report of workgroup 1 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Clin Periodontol 45(Suppl 20):S68–S77. https://doi.org/10.1111/jcpe.12940
Reckelkamm SL, Kamińska I, Baumeister S-E et al (2023) Optimizing a diagnostic model of periodontitis by using targeted proteomics. J Proteome Res 22:2509–2515. https://doi.org/10.1021/acs.jproteome.3c00230
Assarsson E, Lundberg M, Holmquist G et al (2014) Homogenous 96-plex PEA immunoassay exhibiting high sensitivity, specificity, and excellent scalability. PLoS One 9:e95192. https://doi.org/10.1371/journal.pone.0095192
Baumeister S-E, Finger JD, Gläser S et al (2018) Alcohol consumption and cardiorespiratory fitness in five population-based studies. Eur J Prev Cardiol 25:164–172. https://doi.org/10.1177/2047487317738594
Craig CL, Marshall AL, Sjöström M et al (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35:1381–1395. https://doi.org/10.1249/01.MSS.0000078924.61453.FB
Harrell FE (2015) Regression modeling strategies: with applications to linear models, logistic and ordinal regression, and survival analysis, Second edn. Springer series in statistics, Springer, Cham, Heidelberg, New York
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Privratsky JR, Newman PJ (2014) PECAM-1: regulator of endothelial junctional integrity. Cell Tissue Res 355:607–619. https://doi.org/10.1007/s00441-013-1779-3
Dejana E (2004) Endothelial cell-cell junctions: happy together. Nat Rev Mol Cell Biol 5:261–270. https://doi.org/10.1038/nrm1357
McEwan WA, James LC (2015) TRIM21-dependent intracellular antibody neutralization of virus infection. Prog Mol Biol Transl Sci 129:167–187. https://doi.org/10.1016/bs.pmbts.2014.10.006
Clift D, So C, McEwan WA et al (2018) Acute and rapid degradation of endogenous proteins by Trim-Away. Nat Protoc 13:2149–2175. https://doi.org/10.1038/s41596-018-0028-3
McEwan WA, Tam JCH, Watkinson RE et al (2013) Intracellular antibody-bound pathogens stimulate immune signaling via the Fc receptor TRIM21. Nat Immunol 14:327–336. https://doi.org/10.1038/ni.2548
Duong CN, Vestweber D (2020) Mechanisms ensuring endothelial junction integrity beyond VE-cadherin. Front Physiol 11:519. https://doi.org/10.3389/fphys.2020.00519
Yılmaz Şaştım Ç, Gürsoy M, Könönen E et al (2021) Salivary and serum markers of angiogenesis in periodontitis in relation to smoking. Clin Oral Investig 25:1117–1126. https://doi.org/10.1007/s00784-020-03411-4
Kdkhodazadeh M, Hajilooi M, Houshmand B et al (2012) Evaluation of PECAM-1 gene polymorphism in patients with periodontal disease and healthy individuals. ISRN Dent 2012:751920. https://doi.org/10.5402/2012/751920
Farrugia C, Stafford GP, Potempa J et al (2021) Mechanisms of vascular damage by systemic dissemination of the oral pathogen Porphyromonas gingivalis. FEBS J 288:1479–1495. https://doi.org/10.1111/febs.15486
Widziolek M, Prajsnar TK, Tazzyman S et al (2016) Zebrafish as a new model to study effects of periodontal pathogens on cardiovascular diseases. Sci Rep 6:36023. https://doi.org/10.1038/srep36023
Hajishengallis G (2021) Oral bacteria and leaky endothelial junctions in remote extraoral sites. FEBS J 288:1475–1478. https://doi.org/10.1111/febs.15510
Walter C, Zahlten J, Schmeck B et al (2004) Porphyromonas gingivalis strain-dependent activation of human endothelial cells. Infect Immun 72:5910–5918. https://doi.org/10.1128/IAI.72.10.5910-5918.2004
Takahashi Y, Davey M, Yumoto H et al (2006) Fimbria-dependent activation of pro-inflammatory molecules in Porphyromonas gingivalis infected human aortic endothelial cells. Cell Microbiol 8:738–757. https://doi.org/10.1111/j.1462-5822.2005.00661.x
Wimmer I, Tietz S, Nishihara H et al (2019) PECAM-1 stabilizes blood-brain barrier integrity and favors paracellular T-cell diapedesis across the blood-brain barrier during neuroinflammation. Front Immunol 10:711. https://doi.org/10.3389/fimmu.2019.00711
Jones EL, Laidlaw SM, Dustin LB (2021) TRIM21/Ro52 - roles in innate immunity and autoimmune disease. Front Immunol 12:738473. https://doi.org/10.3389/fimmu.2021.738473
Benli M, Batool F, Stutz C et al (2021) Orofacial manifestations and dental management of systemic lupus erythematosus: a review. Oral Dis 27:151–167. https://doi.org/10.1111/odi.13271
Gómez-Martín D, Galindo-Feria AS, Barrera-Vargas A et al (2017) Ro52/TRIM21-deficient expression and function in different subsets of peripheral blood mononuclear cells is associated with a proinflammatory cytokine response in patients with idiopathic inflammatory myopathies. Clin Exp Immunol 188:154–162. https://doi.org/10.1111/cei.12914
Slots J, Slots H (2019) Periodontal herpesvirus morbidity and treatment. Periodontol 2000 79:210–220. https://doi.org/10.1111/prd.12241
Cai B, Liu Y, Chong Y et al (2021) IRAK1-regulated IFN-γ signaling induces MDSC to facilitate immune evasion in FGFR1-driven hematological malignancies. Mol Cancer 20:165. https://doi.org/10.1186/s12943-021-01460-1
Wu X, Xu M, Liu Z et al (2022) Pharmacological inhibition of IRAK1 and IRAK4 prevents endothelial inflammation and atherosclerosis in ApoE-/- mice. Pharmacol Res 175:106043. https://doi.org/10.1016/j.phrs.2021.106043
Takahashi N, Honda T, Domon H et al (2010) Interleukin-1 receptor-associated kinase-M in gingival epithelial cells attenuates the inflammatory response elicited by Porphyromonas gingivalis. J Periodontal Res 45:512–519. https://doi.org/10.1111/j.1600-0765.2009.01266.x
Altman DG, Royston P (2006) The cost of dichotomising continuous variables. BMJ 332:1080. https://doi.org/10.1136/bmj.332.7549.1080
Austin PC, Brunner LJ (2004) Inflation of the type I error rate when a continuous confounding variable is categorized in logistic regression analyses. Stat Med 23:1159–1178. https://doi.org/10.1002/sim.1687
Lang NP, Joss A, Orsanic T et al (1986) Bleeding on probing. A predictor for the progression of periodontal disease? J Clin Periodontol 13:590–596. https://doi.org/10.1111/j.1600-051x.1986.tb00852.x
Frencken JE, Sharma P, Stenhouse L et al (2017) Global epidemiology of dental caries and severe periodontitis - a comprehensive review. J Clin Periodontol 44(Suppl 18):S94–S105. https://doi.org/10.1111/jcpe.12677
Acknowledgements
The authors thank all examiners, technicians, interviewers, participants, and other contributors who were involved in the implementation of this study.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported by the statutory funds of the Medical University of Bialystok and funds of the Ministry of Science and Higher Education (Poland) within the project “The Excellence Initiative - Research University.”
Author information
Authors and Affiliations
Contributions
Conception, design, and writing: Stefan Lars Reckelkamm, Sebastian-Edgar Baumeister, Michael Nolde, and Karol Adam Kamiński. Analysis and interpretation of data: Stefan Lars Reckelkamm, Inga Kamińska, Sebastian-Edgar Baumeister, Michael Nolde, and Karol Adam Kamiński. Acquisition, processing, and restructuring of data: Stefan Lars Reckelkamm, Inga Kamińska, Mariana Ponce-de-Leon, Ewa Rodakowska, Joanna Baginska, Sebastian-Edgar Baumeister, Michael Nolde, and Karol Adam Kamiński. Revision and approval of the article: Stefan Lars Reckelkamm, Inga Kamińska, Mariana Ponce-de-Leon, Ewa Rodakowska, Joanna Baginska, Benjamin Ehmke, Sebastian-Edgar Baumeister, Michael Nolde, and Karol Adam Kamiński.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 81 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Reckelkamm, S.L., Kamińska, I., Baumeister, SE. et al. Targeted proteomics in a population-based study identifies serum PECAM-1 and TRIM21 as inflammation markers for periodontitis. Clin Oral Invest 28, 59 (2024). https://doi.org/10.1007/s00784-023-05442-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00784-023-05442-z