Abstract
Objectives
Postoperative flap monitoring is essential in oral microvascular reconstruction for timely detection of vascular compromise. This study investigated the use of attached surface probes for the oxygen-2-see (O2C) analysis system (LEA Medizintechnik, Germany) for intraoral flap perfusion monitoring.
Materials and methods
The study included 30 patients who underwent oral reconstruction with a microvascular radial-free forearm flap (RFFF) or anterolateral thigh flap (ALTF) between 2020 and 2022. Flap perfusion was measured with attached (3-mm measurement depth) and unattached surface probes (2- and 8-mm measurement depths) for the O2C analysis system at 0, 12, 24, 36, and 48 h postoperatively. Flap perfusion monitoring with attached surface probes was evaluated for cut-off values for flap blood flow, hemoglobin concentration, and hemoglobin oxygen saturation indicative of vascular compromise and for accuracy and concordance with unattached surface probes.
Results
Three RFFFs were successfully revised, and one ALTF was unsuccessfully revised. The cut-off values indicative of vascular compromise for flap perfusion monitoring with attached surface probes were for RFFF and ALTF: blood flow < 60 arbitrary units (AU) and < 40AU, hemoglobin concentration > 100AU and > 80AU (both > 10% increase), and hemoglobin oxygen saturation < 40% and < 30%. Flap perfusion monitoring with attached surface probes yielded a 97.1% accuracy and a Cohen’s kappa of 0.653 (p < 0.001).
Conclusions
Flap perfusion monitoring with attached surface probes for the O2C analysis system detected vascular compromise accurately and concordantly with unattached surface probes.
Clinical relevance
Attached surface probes for the O2C analysis system are a feasible option for intraoral flap perfusion monitoring.
Similar content being viewed by others
Introduction
Microvascular reconstruction of the head and neck region with free flaps offers functional and esthetic outcomes as well as high overall success rates [1, 2]. However, the use of microvascular free flaps remains challenging due to transient flap failure with the need for flap revision and terminal flap failure with loss of the flap, which is associated with high patient burden and the need for additional surgeries or lasting functional and esthetic compromise [1, 3, 4].
Microvascular free flap perfusion, as a prerequisite for flap viability, initially depends entirely on continuous arterial inflow and venous outflow through a patent microvascular anastomosis, and ischemia of the flap related to vascular compromise is associated with deleterious effects such as microthrombi formation, ultimately leading to irreversible microcirculation failure and subsequently flap failure [5,6,7,8,9]. Given the inverse relationship of the time interval between the onset of vascular compromise and intervention and the flap salvage rate, postoperative flap monitoring for timely detection of vascular compromise is crucial [8, 10, 11]. In this context, the most commonly performed method of clinical monitoring is based on flap color, surface temperature, capillary refill, and pricking tests; however, such monitoring is limited by several constraints, such as the need for clinical experience, lack of objective values, and delay between the onset of vascular compromise and the appearance of clinical changes [8, 12, 13].
Therefore, the oxygen-2-see (O2C) analysis system (LEA Medizintechnik, Germany) was developed as an objective method for flap perfusion monitoring, and cut-off values indicative of vascular compromise have been established for unattached surface probes measuring flap perfusion at tissue depths of 2 mm and 8 mm [7, 9]. However, attached surface probes measuring flap perfusion at a tissue depth of 3 mm to monitor flap perfusion with the O2C analysis system have potential advantages, such as constant probe pressure and consistent location of the measurement area, both of which may affect the measurements; moreover, attached surface probes have the potential to provide the basis for continuous monitoring [14,15,16].
This study aimed to evaluate the use of attached surface probes for monitoring flap perfusion with the O2C analysis system in microvascular head and neck reconstruction for the detection of vascular compromise.
Materials and methods
Study population
This study was approved by the local ethics committee of the Medical Faculty RWTH Aachen University (EK 22–358). All study data were prospectively collected and retrospectively analyzed.
The study population consisted of 30 patients who had undergone reconstruction with a microvascular free flap — i.e., a radial free forearm flap (RFFF) or an anterolateral thigh flap (ALTF) — in the oral cavity between 2020 and 2022 in our Department of Oral and Maxillofacial Surgery after ablative surgery for malignant or nonmalignant disease. Patients with complete data records who were older than 18 years were included.
Data were obtained from clinical records and O2C analysis system measurement records. Surgery duration and flap ischemia duration were calculated as the time interval between the first incision and the last suture or between the separation of the flap pedicle vessel at the donor site and the release of the vascular clamp to initiate flap perfusion at the recipient side after anastomosis. Flap revision was defined in terms of flaps whose anastomosis was surgically revised with a return to the operating room, and flap failure was defined in terms of flaps that were completely lost due to tissue necrosis and subsequently removed.
All surgical procedures were performed under general anesthesia, and all patients were monitored postoperatively in the intensive care unit. All patients received invasive mechanical ventilation and analgosedation at least until the next morning.
Flap perfusion monitoring
Flap perfusion was simultaneously monitored with the O2C analysis system (oxygen-to-see (O2C), LEA Medizintechnik, Giesen, Germany) with an unattached surface probe (type LFX90) as well as with an attached surface probe (type LF2) at 0, 12, 24, 36, and 48 h postoperatively (Fig. 1). The attached surface probe, measuring perfusion at a tissue depth of 3 mm, was fixed with four sutures in the middle of the RFFF and at a distance of 1 cm from the perforator vessel, approximately in the middle of the ALTF. The unattached probe, measuring perfusion at tissue depths of 2 mm and 8 mm, was held in the same position approximately parallel to the attached surface probe for each measurement.
The O2C analysis system for measuring flap tissue perfusion is based on the following two principles: laser light Doppler spectroscopy (830 nm; 30 mW) for the determination of flap blood flow (arbitrary units [AU]) and white light spectroscopy (500–800 nm; 50 W) for the determination of hemoglobin concentration (AU) and hemoglobin oxygen saturation (%) [7, 17]. The probe transmits the laser and white light into the flap tissue and then detects the backscattered light [7, 17]. Blood flow values are calculated as the product of erythrocyte quantity, based on the analysis of the sum of light absorbances, and velocity, based on the analysis of the Doppler shift of light wave frequencies due to the movement of the erythrocytes in the blood vessels [7, 17]. Hemoglobin concentration and hemoglobin oxygen saturation values are calculated as the sum of light absorbances, and the color change of light absorbances in comparison to prerecorded hemoglobin spectra with defined oxygen saturation, respectively [7, 17]. For both surface probes, the measurement time interval was 10 s.
Statistical analysis
Clinical data are expressed as numbers (with percentages) or medians (with interquartile ranges). Measurement data for flap perfusion — i.e., flap blood flow, hemoglobin concentration, and hemoglobin oxygen saturation — obtained with the unattached surface probe were used as a reference standard, with the following cut-off values indicative of vascular compromise according to previous studies: blood flow at 8-mm tissue depth: RFFF < 20AU and ALTF < 15AU; hemoglobin concentration at 8-mm tissue depth: RFFF and ALTF both > 30% increase; hemoglobin oxygen saturation at 8-mm tissue depth: RFFF < 15% and ALTF < 10%; blood flow at 2-mm tissue depth: RFFF < 10AU and ALTF < 5AU; hemoglobin concentration at 2-mm tissue depth: RFFF and ALTF both > 30% increase; and hemoglobin oxygen saturation at 2-mm tissue depth: RFFF < 15% and ALTF < 10% [7, 9]. Cut-off values indicative of vascular compromise for flap perfusion monitoring with attached surface probes were derived based on flap perfusion measurement values from not revised and revised flaps separately for RFFF and ALTF. Sensitivity, specificity, and accuracy for flap perfusion monitoring with attached surface probes and Cohen’s kappa for determining concordance between flap perfusion monitoring with attached surface probes and flap perfusion monitoring with unattached surface probes were calculated according to commonly used definitions [18, 19]. Values of p < 0.05 were considered to be statistically significant. The statistical analysis was performed using SPSS version 28 (SPSS, IBM, New York, USA).
Results
Study population
The study population was composed of 30 patients, 11 men and 19 women (Table 1). Twenty-seven patients underwent reconstruction with a RFFF, and three patients underwent reconstruction with an ALTF. A total of four flaps were revised (3 RFFF primarily due to venous vascular compromise and 1 ALTF primarily due to arterial vascular compromise), and one flap was lost (ALTF).
Flap perfusion measurement values
Flap perfusion measurement values for blood flow, hemoglobin oxygenation, and hemoglobin oxygen saturation for not revised flaps are described and presented separately for RFFF and ALTF at 0, 12, 24, 36, and 48 h postoperatively (Table 2; Fig. 2). In addition, flap perfusion measurement values for blood flow, hemoglobin oxygenation, and hemoglobin oxygen saturation for revised flaps are presented separately for each flap at 0, 12, 24, 36, and 48 h postoperatively (Fig. 3).
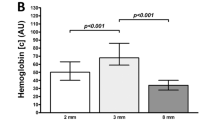
Flap perfusion monitoring of not revised flaps. Data shown as median (with (A) and without (B) interquartile range) separately for RFFF (A) and ALTF (B) for blood flow (1), hemoglobin concentration (2), and hemoglobin oxygen saturation (3) in 2-mm (light grey), 3-mm (middle grey), and 8-mm (dark grey) tissue depths at different measurement timepoints (0 h postoperatively, 12 h postoperatively, 24 h postoperatively, 36 h postoperatively, 48 h postoperatively); AU arbitrary units
Flap perfusion monitoring of revised flaps. Data shown as individual perfusion values for three revised RFFF (A, B, C) primarily due to venous vascular compromise and one revised ALTF (D) primarily due to arterial vascular compromise separately for blood flow (1), hemoglobin concentration (2), and hemoglobin oxygen saturation (3) in 2-mm (dotted line), 3-mm (straight line), and 8-mm (dashed line) tissue depths at different measurement timepoints (0 h postoperatively, 12 h postoperatively, 24 h postoperatively, 36 h postoperatively, 48 h postoperatively); arrow: flap perfusion measurement before flap revision (ALTF revision was not successful); [c] concentration, AU arbitrary units
Determination of cut-off values
Based on the perfusion measurement values for not revised and revised flaps, the following cut-off values indicative of vascular compromise for flap perfusion monitoring with attached surface probes were derived: blood flow RFFF < 60AU and ALTF < 40AU; hemoglobin concentration RFFF > 100AU or > 10% increase and ALTF > 80AU or > 10% increase; and hemoglobin oxygen saturation RFFF < 40% and ALTF < 30% (Table 3).
Evaluation of sensitivity, specificity, accuracy, and concordance
Based on the derived cut-off values indicative of vascular compromise for flap perfusion monitoring with attached surface probes, the following values for sensitivity, specificity, accuracy, and concordance in detecting vascular compromise were obtained: sensitivity, 100.0%; specificity, 97.1%; accuracy, 97.1%; and Cohen’s kappa, 0.653 (p < 0.001).
Discussion
This study evaluated postoperative flap perfusion monitoring with attached surface probes for the O2C analysis system in terms of determining cut-off values indicative of vascular compromise. Based on these cut-off values, sensitivity, specificity, accuracy, and concordance with flap perfusion monitoring using standard unattached surface probes were assessed in terms of detecting vascular compromise [7, 9].
Postoperative flap monitoring is considered crucial in microvascular free flap reconstruction of the head and neck region, as vascular compromise occurs, and timely detection and intervention are prerequisite for flap salvage [8, 10, 11]. In this context, the O2C analysis system was developed as a reliable and objective method for flap monitoring based on flap perfusion measurement, as flap perfusion is a prerequisite for flap viability [7,8,9, 20, 21]. Nonetheless, flap perfusion can be assessed with several other methods, including indocyanine green angiography with the disadvantages of invasiveness and the need for intravenous administration of agents, pin prick test with the disadvantages of invasiveness and subjective interpretation, and basic doppler assessment of vascular flow with the disadvantages of subjective interpretation and difficulty in distinguishing between arterial and venous compromise [7,8,9]. The O2C analysis system is standardly used with unattached surface probes that measure flap perfusion at tissue depths of 2 mm and 8 mm, and the attendant cut-off values indicative of vascular compromise have been previously evaluated and approved [7, 9]. However, attached surface probes that measure flap perfusion only at one tissue depth of 3 mm, but are smaller and lighter, could offer potential advantages for flap perfusion measurement, such as consistent probe pressure and location of the measurement area over the entire postoperative monitoring course — both of which are likely to influence flap perfusion measurement — and could provide the basis for continuous flap perfusion monitoring [14,15,16].
In this study, cut-off values indicative of vascular compromise were determined for flap perfusion monitoring with attached surface probes for the O2C analysis system. Cut-off values indicative of vascular compromise differed between the attached and unattached surface probes, reflecting differences in measurement depth — i.e., 2 mm and 8 mm and 3 mm, respectively — and thus differences in the skin microvasculature depending on the skin layer examined — i.e., the dermis containing the superficial papillary and the deeper reticular plexus and the subcutaneous tissue containing the subcutaneous plexus [22, 23]. Differences in vessel density and diameter between these plexus formations, for example, are likely to affect blood flow and hemoglobin concentration in terms of the relationship between vessel diameter, resistance, and blood flow [22,23,24]. In addition, hemoglobin oxygen saturation has been shown to be dependent on blood flow [7]. When considering the differences in perfusion measurements in this study, it should be noted that in addition to the measurement depth, the measurement area was not identical between the two surface probes [14, 15]. Furthermore, the influence of the intraoral moist environment on the perfusion measurement, which is likely to be more pronounced with attached probes since they cannot be removed for drying, could not be excluded [25].
Notably, in line with cut-off values indicative of vascular compromise for unattached probes for flap perfusion monitoring with the O2C analysis system determined in previous studies and used as the reference standards in this study, cut-off values indicative of vascular compromise for attached probes were higher in terms of blood flow and hemoglobin oxygen saturation in RFFF than in ALTF [7, 9]. The differences with respect to a cut-off value of an increase in hemoglobin concentration greater than 10% for attached surface probes and greater than 30% for unattached surface probes may be related to the tendency for generally higher hemoglobin concentration values with attached surface probes, measuring at a tissue depth of 3 mm, compared with unattached surface probes, measuring at tissue depths of 2 mm and 8 mm [7, 9].
This study found that flap perfusion monitoring with attached surface probes for the O2C analysis system on the basis of all flap perfusion parameters combined — i.e., blood flow, hemoglobin concentration, and hemoglobin oxygen saturation — yielded a sensitivity of 100.0%, a specificity of 97.1%, and an accuracy of 97.1% in detecting vascular compromise, relative to flap perfusion monitoring with attached surface probes for the O2C analysis system serving as the reference standard method. Interestingly, some measurements with attached surface probes showed hemoglobin oxygen saturation values indicative of vascular compromise, whereas this was not the case with unattached surface probes. This resulted in a specificity of 97.1% for hemoglobin oxygen saturation evaluated separately but also possibly reflects the potential of flap perfusion monitoring with attached surface probes in the earlier detection of vascular compromise. In addition, based on a Cohen’s kappa of 0.653, the study found substantial concordance between flap perfusion monitoring with attached and unattached surface probes [18]. However, given the low specificity value, the concordance between attached and unattached surface probes for hemoglobin oxygen saturation evaluated separately was only fair, with a Cohen’s kappa of 0.325 [18].
In general, the study demonstrated that flap perfusion monitoring with attached surface probes for the O2C analysis system was comparable to flap perfusion monitoring with unattached surface probes in terms of technical feasibility and patient safety, as no event of probe detachment or disconnection and no infections or bleeding due to the attachment structures were observed as potential disadvantages of the attached surface probes [26].
Nevertheless, the study has several limitations, such as the small number of patients in the sample, the limited number of ALTFs included in the study, and the fact that only four flaps in the study had vascular compromise and subsequently underwent flap revision. With regard to the limited number of four flaps that required revision, it should be noted that in microvascular reconstruction of the head and neck region, the number of free flaps that require revision is generally low; the percentage of flaps that required revision in this study, 13.3%, was within the range observed in the literature [8, 27,28,29]. However, the determination of cut-off values indicative of vascular compromise and the assessment of values such as sensitivity, specificity, and accuracy in terms of the detection of vascular compromise for attached surface probes can only be considered as approximative values and a first orientation in this context. Furthermore, all revised RFFFs showed primarily venous vascular compromise and therefore likely only secondarily altered values for blood flow; in addition, the only revised ALTF showed primarily arterial vascular compromise and therefore likely only secondarily altered values for hemoglobin concentration [7, 9]. Regarding the limited postoperative monitoring period in this study up to 48 h postoperatively, and the potential occurrence of the requirement for flap revision beyond this period, it should be kept in mind that the risk of vascular compromise in free flaps is highest during the first period of 48 h postoperatively, and the flap salvage rate is lower beyond the period of 48 h postoperatively [11, 30, 31].
This study evaluated for the first time postoperative flap perfusion monitoring with attached surface probes for the O2C analysis system in terms of detecting vascular compromise in microvascular head and neck reconstruction. The study demonstrated that attached surface probes are technically feasible and represent a safe option for patients to be used with the O2C analysis system for postoperative flap perfusion monitoring. Based on distinctive cut-off values, flap perfusion monitoring with attached surface probes showed high accuracy and, more importantly, a substantial concordance with flap perfusion monitoring with unattached surface probes, supporting interchangeability between the two surface probes. Further studies are needed to confirm the cut-off values indicative of vascular compromise for attached surface probes and to validate their accuracy to maintain the clinical utility of the O2C analysis system for flap perfusion monitoring.
Conclusion
This study demonstrated that flap perfusion monitoring with attached surface probes for the O2C analysis system was comparable to the standard use with unattached surface probes in terms of technical feasibility and patient safety. Based on cut-off values indicative of vascular compromise (blood flow: RFFF < 60AU and ALTF < 40AU; hemoglobin concentration: RFFF > 100AU or > 10% increase and ALTF > 80AU or > 10% increase; hemoglobin oxygen saturation: RFFF < 40% and ALTF < 30%), flap perfusion monitoring with attached surface probes showed an accuracy of 97.1% in detecting vascular compromise and may form the basis for continuous flap perfusion monitoring with the O2C analysis system.
Data availability
The data underlying this article is available on reasonable request to the corresponding author.
References
Wong C-H, Wei F-C (2010) Microsurgical free flap in head and neck reconstruction. Head Neck 32:1236–1245. https://doi.org/10.1002/hed.21284
Wang K-Y, Lin Y-S, Chen L-W et al (2020) Risk of free flap failure in head and neck reconstruction: analysis of 21,548 cases from a nationwide database. Ann Plast Surg 84:S3–S6. https://doi.org/10.1097/SAP.0000000000002180
Chang CS, Chu MW, Nelson JA et al (2017) Complications and cost analysis of intraoperative arterial complications in head and neck free flap reconstruction. J Reconstr Microsurg 33:318–327. https://doi.org/10.1055/s-0037-1598618
Abouyared M, Katz AP, Ein L et al (2019) Controversies in free tissue transfer for head and neck cancer: a review of the literature. Head Neck 41:3457–3463. https://doi.org/10.1002/hed.25853
Carroll WR, Esclamado RM (2000) Ischemia/reperfusion injury in microvascular surgery. Head Neck 22:700–713. https://doi.org/10.1002/1097-0347(200010)22:7%3c700:aid-hed10%3e3.0.co;2-h
Siemionow M, Arslan E (2004) Ischemia/reperfusion injury: a review in relation to free tissue transfers. Microsurgery 24:468–475. https://doi.org/10.1002/micr.20060
Hölzle F, Loeffelbein DJ, Nolte D et al (2006) Free flap monitoring using simultaneous non-invasive laser Doppler flowmetry and tissue spectrophotometry. J Craniomaxillofac Surg 34:25–33. https://doi.org/10.1016/j.jcms.2005.07.010
Abdel-Galil K, Mitchell D (2009) Postoperative monitoring of microsurgical free tissue transfers for head and neck reconstruction: a systematic review of current techniques—part I. Non-invasive techniques. Br J Oral Maxillofac Surg 47:351–355. https://doi.org/10.1016/j.bjoms.2008.11.013
Hölzle F, Rau A, Loeffelbein DJ et al (2010) Results of monitoring fasciocutaneous, myocutaneous, osteocutaneous and perforator flaps: 4-year experience with 166 cases. Int J Oral Maxillofac Surg 39:21–28. https://doi.org/10.1016/j.ijom.2009.10.012
Sweeny L, Curry J, Crawley M et al (2020) Factors impacting successful salvage of the failing free flap. Head Neck 42:3568–3579. https://doi.org/10.1002/hed.26427
Shen AY, Lonie S, Lim K et al (2021) Free flap monitoring, salvage, and failure timing: a systematic review. J Reconstr Microsurg 37:300–308. https://doi.org/10.1055/s-0040-1722182
Lohman RF, Langevin C-J, Bozkurt M et al (2013) A prospective analysis of free flap monitoring techniques: physical examination, external Doppler, implantable Doppler, and tissue oximetry. J Reconstr Microsurg 29:51–56. https://doi.org/10.1055/s-0032-1326741
Kwasnicki R, Noakes A, Banhidy N et al (2021) Quantifying the limitations of clinical and technology-based flap monitoring strategies using a systematic recurrent themes analysis. Br J Surg 108:S6. https://doi.org/10.1093/bjs/znab259.699
Tenland T, Salerud EG, Nilsson GE et al (1983) Spatial and temporal variations in human skin blood flow. Int J Microcirc Clin Exp 2:81–90
Abel G, Allen J, Drinnan M (2014) A pilot study of a new spectrophotometry device to measure tissue oxygen saturation. Physiol Meas 35:1769–1780. https://doi.org/10.1088/0967-3334/35/9/1769
Zhang XU, Faber DJ, van Leeuwen TG et al (2020) Effect of probe pressure on skin tissue optical properties measurement using multi-diameter single fiber reflectance spectroscopy. J Phys Photonics 2:34008. https://doi.org/10.1088/2515-7647/ab9071
Beckert S, Witte MB, Königsrainer A et al (2004) The impact of the Micro-Lightguide O2C for the quantification of tissue ischemia in diabetic foot ulcers. Diabetes Care 27:2863–2867. https://doi.org/10.2337/diacare.27.12.2863
McHugh ML (2012) Interrater reliability: the kappa statistic. Biochem Med 22:276–282
Bolboacă SD (2019) Medical diagnostic tests: a review of test anatomy, phases, and statistical treatment of data. Comput Math Methods Med 2019:1891569. https://doi.org/10.1155/2019/1891569
Kamolz L-P, Giovanoli P, Haslik W et al (2002) Continuous free-flap monitoring with tissue-oxygen measurements: three-year experience. J Reconstr Microsurg 18:487–491; discussion 492–493. https://doi.org/10.1055/s-2002-33319
Rahmanian-Schwarz A, Rothenberger J, Amr A et al (2012) A postoperative analysis of perfusion dynamics in deep inferior epigastric perforator flap breast reconstruction: a noninvasive quantitative measurement of flap oxygen saturation and blood flow. Ann Plast Surg 69:535–539. https://doi.org/10.1097/SAP.0b013e31821bd484
Cammarota T, Pinto F, Magliaro A et al (1998) Current uses of diagnostic high-frequency US in dermatology. Eur J Radiol 27(Suppl 2):S215-223. https://doi.org/10.1016/S0720-048X(98)00065-5
Deegan AJ, Wang RK (2019) Microvascular imaging of the skin. Phys Med Biol 64:07TR01. https://doi.org/10.1088/1361-6560/ab03f1
Sirs JA (1991) The flow of human blood through capillary tubes. J Physiol 442:569–583. https://doi.org/10.1113/jphysiol.1991.sp018809
Hohlweg-Majert B, Ristow O, Gust K et al (2012) Impact of radiotherapy on microsurgical reconstruction of the head and neck. J Cancer Res Clin Oncol 138:1799–1811. https://doi.org/10.1007/s00432-012-1263-6
Wu C, Rwei AY, Lee JY et al (2022) A wireless near-infrared spectroscopy device for flap monitoring: proof of concept in a porcine musculocutaneous flap model. J Reconstr Microsurg 38:96–105. https://doi.org/10.1055/s-0041-1732426
Chen K-T, Mardini S, Chuang DC-C et al (2007) Timing of presentation of the first signs of vascular compromise dictates the salvage outcome of free flap transfers. Plast Reconstr Surg 120:187–195. https://doi.org/10.1097/01.prs.0000264077.07779.50
Salgado CJ, Chim H, Schoenoff S et al (2010) Postoperative care and monitoring of the reconstructed head and neck patient. Semin Plast Surg 24:281–287. https://doi.org/10.1055/s-0030-1263069
Hayler R, Low T-HH, Fung K et al (2021) Implantable Doppler ultrasound monitoring in head and neck free flaps: balancing the pros and cons. Laryngoscope 131:E1854–E1859. https://doi.org/10.1002/lary.29247
Devine JC, Potter LA, Magennis P et al (2001) Flap monitoring after head and neck reconstruction: evaluating an observation protocol. J Wound Care 10:525–529. https://doi.org/10.12968/jowc.2001.10.1.26037
Smit JM, Acosta R, Zeebregts CJ et al (2007) Early reintervention of compromised free flaps improves success rate. Microsurgery 27:612–616. https://doi.org/10.1002/micr.20412
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
M.O.: Investigation, Data curation, Formal analysis, Visualization, Writing—Original draft preparation; P.W.: Formal analysis, Writing—Reviewing and Editing; M.H.: Writing—Reviewing and Editing; F.P.: Writing—Reviewing and Editing; A.B.: Writing—Reviewing and Editing; M.K.: Resources, Writing—Reviewing and Editing; F.H.: Resources, Supervision, Writing—Reviewing and Editing; A.M.: Project administration, Conceptualization, Methodology, Supervision, Writing—Reviewing and Editing.
Corresponding author
Ethics declarations
Ethical approval
This retrospective chart review study involving human participants was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the local ethics committee of the Medical Faculty RWTH Aachen University (EK 22–358).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ooms, M., Winnand, P., Heitzer, M. et al. Flap perfusion monitoring with an attached surface probe in microvascular reconstruction of the oral cavity. Clin Oral Invest 27, 5577–5585 (2023). https://doi.org/10.1007/s00784-023-05177-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-023-05177-x