Abstract
Objectives
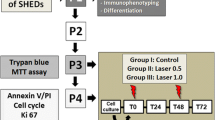
This study was designed to explore the effects of mechanical force on the proliferation, apoptosis, and morphology of stem cells from human exfoliated deciduous tooth pulp (SHEDs).
Materials and methods
Caries-free stranded deciduous teeth were extracted, and SHEDs were isolated through enzymatic digestion. The cultured SHEDs were subjected to different levels of mechanical stimuli (0, 100, 200, and 300 g) for 7 days (30 min/day) using external centrifugal force. Cell proliferation was evaluated with the CCK-8 assay, and the cell cycle and apoptosis were assessed by flow cytometry. The cell morphology was examined using transmission electron microscopy.
Results
Cell proliferation assay showed no differences between the three stimulation groups and the control group in day 1 to day 3. From the 4th day, cell proliferation was significantly lower in the mechanical force groups than in the control group, but no significant difference was observed among the three mechanical force groups. Besides, there was no significant difference in cell apoptosis among the four groups for 7 days. On day 7 after stimulation, the SHEDs were shrunken, with significantly increased isochromosome in the nucleus and an increase in lysosomes.
Conclusions
Mechanical force can inhibit the proliferation and affect morphology of SHEDs, but it has no effect on cell apoptosis.
Clinical relevance
Mechanical force stimulation significantly inhibited cell proliferation of SHEDs. Mechanical force stimulation had no significant effect on cell apoptosis of SHEDs. The morphology and ultrastructure of SHEDs changed after mechanical force stimulation.





Similar content being viewed by others
References
Morsczeck C, Reichert TE (2018) Dental stem cells in tooth regeneration and repair in the future. Expert Opin Biol Ther 18(2):187–196. https://doi.org/10.1080/14712598.2018.1402004
Kim JH, Kim GH, Kim JW, Pyeon HJ, Lee JC, Lee G, Nam H (2016) In vivo angiogenic capacity of stem cells from human exfoliated deciduous teeth with human umbilical vein endothelial cells. Mol Cells 39(11):790–796. https://doi.org/10.14348/molcells.2016.0131
Hata M, Naruse K, Ozawa S, Kobayashi Y, Nakamura N, Kojima N, Omi M, Katanosaka Y, Nishikawa T, Naruse K, Tanaka Y, Matsubara T (2013) Mechanical stretch increases the proliferation while inhibiting the osteogenic differentiation in dental pulp stem cells. Tissue Eng Part A 19(5–6):625–633. https://doi.org/10.1089/ten.tea.2012.0099
Modino SA, Sharpe PT (2005) Tissue engineering of teeth using adult stem cells. Arch Oral Biol 50(2):255–258. https://doi.org/10.1016/j.archoralbio.2005.01.002
Karaoz E, Demircan PC, Saglam O, Aksoy A, Kaymaz F, Duruksu G (2011) Human dental pulp stem cells demonstrate better neural and epithelial stem cell properties than bone marrow-derived mesenchymal stem cells. Histochem Cell Biol 136(4):455–473. https://doi.org/10.1007/s00418-011-0858-3
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A 97(25):13625–13630. https://doi.org/10.1073/pnas.240309797
Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S (2003) SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A 100(10):5807–5812. https://doi.org/10.1073/pnas.0937635100
Mammoto T, Ingber DE (2010) Mechanical control of tissue and organ development. Development 137(9):1407–1420. https://doi.org/10.1242/dev.024166
Hall M, Masella R, Meister M (2001) PDL neuron-associated neurotransmitters in orthodontic tooth movement: identification and proposed mechanism of action. Todays FDA 13(2):24–25
Tsutsui TW (2020) Dental pulp stem cells: advances to applications. Stem Cells Cloning 13:33–42. https://doi.org/10.2147/SCCAA.S166759
Shi S, Bartold PM, Miura M, Seo BM, Robey PG, Gronthos S (2005) The efficacy of mesenchymal stem cells to regenerate and repair dental structures. Orthod Craniofac Res 8(3):191–199. https://doi.org/10.1111/j.1601-6343.2005.00331.x
von Bohl M, Ren Y, Fudalej PS, Kuijpers-Jagtman AM (2012) Pulpal reactions to orthodontic force application in humans: a systematic review. J Endod 38(11):1463–1469. https://doi.org/10.1016/j.joen.2012.07.001
Wise GE, King GJ (2008) Mechanisms of tooth eruption and orthodontic tooth movement. J Dent Res 87(5):414–434. https://doi.org/10.1177/154405910808700509
Yu V, Damek-Poprawa M, Nicoll SB, Akintoye SO (2009) Dynamic hydrostatic pressure promotes differentiation of human dental pulp stem cells. Biochem Biophys Res Commun 386(4):661–665. https://doi.org/10.1016/j.bbrc.2009.06.106
Mu C, Lv T, Wang Z, Ma S, Ma J, Liu J, Yu J, Mu J (2014) Mechanical stress stimulates the osteo/odontoblastic differentiation of human stem cells from apical papilla via erk 1/2 and JNK MAPK pathways. Biomed Res Int 2014:494378. https://doi.org/10.1155/2014/494378
Li J, Jiang L, Liao G, Chen G, Liu Y, Wang J, Zheng Y, Luo S, Zhao Z (2009) Centrifugal forces within usually-used magnitude elicited a transitory and reversible change in proliferation and gene expression of osteoblastic cells UMR-106. Mol Biol Rep 36(2):299–305. https://doi.org/10.1007/s11033-007-9179-y
Huang J, Shen L, Lu Y, Li H, Zhang X, Hu D, Feng T, Song F (2012) Parallel induction of cell proliferation and inhibition of cell differentiation in hepatic progenitor cells by hepatitis B virus X gene. Int J Mol Med 30(4):842–848. https://doi.org/10.3892/ijmm.2012.1060
Nakamura S, Yamada Y, Katagiri W, Sugito T, Ito K, Ueda M (2009) Stem cell proliferation pathways comparison between human exfoliated deciduous teeth and dental pulp stem cells by gene expression profile from promising dental pulp. J Endod 35(11):1536–1542. https://doi.org/10.1016/j.joen.2009.07.024
Heng BC, Zhu S, Xu J, Yuan C, Gong T, Zhang C (2016) Effects of decellularized matrices derived from periodontal ligament stem cells and SHED on the adhesion, proliferation and osteogenic differentiation of human dental pulp stem cells in vitro. Tissue Cell 48(2):133–143. https://doi.org/10.1016/j.tice.2015.12.004
Akpinar G, Kasap M, Aksoy A, Duruksu G, Gacar G, Karaoz E (2014) Phenotypic and proteomic characteristics of human dental pulp derived mesenchymal stem cells from a natal, an exfoliated deciduous, and an impacted third molar tooth. Stem Cells Int 2014:457059. https://doi.org/10.1155/2014/457059
Vishwanath VR, Nadig RR, Nadig R, Prasanna JS, Karthik J, Pai VS (2013) Differentiation of isolated and characterized human dental pulp stem cells and stem cells from human exfoliated deciduous teeth: An in vitro study. J Conserv Dent 16(5):423–428. https://doi.org/10.4103/0972-0707.117509
Jones TD, Naimipour H, Sun S, Cho M, Alapati SB (2015) Mechanical changes in human dental pulp stem cells during early odontogenic differentiation. J Endod 41(1):50–55. https://doi.org/10.1016/j.joen.2014.07.030
Sabbagh J, Ghassibe-Sabbagh M, Fayyad-Kazan M, Al-Nemer F, Fahed JC, Berberi A, Badran B (2020) Differences in osteogenic and odontogenic differentiation potential of DPSCs and SHED. J Dent 101:103413. https://doi.org/10.1016/j.jdent.2020.103413
Frost HM (2004) A 2003 update of bone physiology and Wolff’s Law for clinicians. Angle Orthod 74(1):3–15. https://doi.org/10.1043/0003-3219(2004)074%3c0003:AUOBPA%3e2.0.CO;2
Liu J, Liu T, Zheng Y, Zhao Z, Liu Y, Cheng H, Luo S, Chen Y (2006) Early responses of osteoblast-like cells to different mechanical signals through various signaling pathways. Biochem Biophys Res Commun 348(3):1167–1173. https://doi.org/10.1016/j.bbrc.2006.07.175
Tang L, Lin Z, Li YM (2006) Effects of different magnitudes of mechanical strain on osteoblasts in vitro. Biochem Biophys Res Commun 344(1):122–128. https://doi.org/10.1016/j.bbrc.2006.03.123
Naqvi SM, McNamara LM (2020) Stem cell mechanobiology and the role of biomaterials in governing mechanotransduction and matrix production for tissue regeneration. Front Bioeng Biotechnol 8:597661. https://doi.org/10.3389/fbioe.2020.597661
Kokkinos PA, Zarkadis IK, Kletsas D, Deligianni DD (2009) Effects of physiological mechanical strains on the release of growth factors and the expression of differentiation marker genes in human osteoblasts growing on Ti-6Al-4V. J Biomed Mater Res A 90(2):387–395. https://doi.org/10.1002/jbm.a.32105
Baumert U, Golan I, Redlich M, Roos HA, Muessig DJJoB (2006) Pressure simulation of orthodontic force. 39 (1):S569-S569
Van Schepdael A, Vander Sloten J, Geris L (2013) A mechanobiological model of orthodontic tooth movement. Biomech Model Mechanobiol 12(2):249–265. https://doi.org/10.1007/s10237-012-0396-5
Ueda M, Goto T, Kuroishi KN, Gunjigake KK, Ikeda E, Kataoka S, Nakatomi M, Toyono T, Seta Y, Kawamoto T (2016) Asporin in compressed periodontal ligament cells inhibits bone formation. Arch Oral Biol 62:86–92. https://doi.org/10.1016/j.archoralbio.2015.11.010
Mizuhara M, Kometani-Gunjigake K, Nakao-Kuroishi K, Toyono T, Hitomi S, Morii A, Shiga M, Seta Y, Ono K, Kawamoto T (2020) Vesicular nucleotide transporter mediates adenosine triphosphate release in compressed human periodontal ligament fibroblast cells and participates in tooth movement-induced nociception in rats. Arch Oral Biol 110:104607. https://doi.org/10.1016/j.archoralbio.2019.104607
Woloszyk A, Holsten Dircksen S, Bostanci N, Muller R, Hofmann S, Mitsiadis TA (2014) Influence of the mechanical environment on the engineering of mineralised tissues using human dental pulp stem cells and silk fibroin scaffolds. PLoS ONE 9(10):e111010. https://doi.org/10.1371/journal.pone.0111010
Han Y, Yang Q, Huang Y, Li X, Li WJOD (2020) Mechanical force inhibited hPDLSCs proliferation with the downregulation of MIR31HG via DNA methylation. Oral Dis 27(5):1268–1282. https://doi.org/10.1111/odi.13637
Funding
This work was supported by Medical Scientific Research Foundation of Guangdong Province of China (No. A2019295).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Ethics Committee of Stomatological Hospital of Southern Medical University (EC-CT- [2019] 05).
Consent to participate
Both the children and their guardians agreed to donate human exfoliated deciduous teeth.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Q., Qian, H., Yu, H. et al. Effects of mechanical force on proliferation and apoptosis of stem cells from human exfoliated deciduous teeth. Clin Oral Invest 26, 5205–5213 (2022). https://doi.org/10.1007/s00784-022-04488-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04488-9