Abstract
Objective
The aim of this 12-month mono-centre double-blind randomized placebo-controlled clinical study was to evaluate the efficacy of Lactobacillus reuteri-containing lozenges during the supportive therapy of generalized periodontitis stage III and IV, grade C (GPIII-IVC) patients.
Material and methods
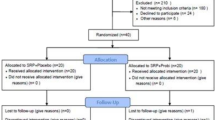
Twenty treated GPIII-IVC patients were randomly divided into 2 groups. The test group received two 3-month-long administrations of L. reuteri (2 lozenges/day after brushing) with a 3-month washout period, while the control one received a placebo. Outcome measures were tooth survival, complications and adverse events, change in probing pockets depth (PPD), change in probing attachment level (PAL), presence of bleeding on probing (BOP) and patient’s evaluation of treatment. Measurements were collected at 3, 6, 9 and 12 months.
Results
At 1 year, no dropout, tooth loss, complications or adverse event were recorded. Mean PPD and mean PAL and percentages of sites with BOP were statistically improved (p < 0.05) compared with baseline in both groups, while more PPD reduction at all time points (p < 0.05) and more PAL gain at 6 months and more BOP reduction at 6 and 9 months were found in the probiotic group (p < 0.05).
Conclusions
Within the limitation of the study, the use of L. reuteri probiotics lozenges improved some clinical outcomes in treated GPIII-IVC patients during maintenance therapy. Studies with a larger number of patients are needed to confirm these data.
Clinical relevance
The use of L. reuteri probiotic lozenges could be considered as an adjunct in the maintenance therapy of GPIII-IVC patients.

Similar content being viewed by others
Notes
Hu-Friedy, Chicago, IL, USA.
References
Tonetti MS, Chapple ILC (2011) on Behalf of Working Group 3 of the Seventh European Workshop on Periodontology. Biological approaches to the development of novel periodontal therapies – consensus of the Seventh European Workshop on Periodontology. J Clin Periodontol 2011 38(Suppl. 11):114–118. https://doi.org/10.1111/j.1600-051X.2010.01675.x
Quirynen M, Vogels R, Pauwels M, Haffajee AD, Socransky SS, Uzel N, van Steenberghe D (2005) Initial subgingival colonization of “pristine” pockets. J Dent Res 83:340–344
Teughels W, Newman MG, Coucke W, Haffajee AD, Van Der Mei HC, Haake SK, Schepers E, Cassiman JJ, Van Eldere J, van Steenberghe D, Quirynen M (2007) Guiding periodontal pocket recolonization: a proof of concept. J Dent Res 86:1078–1082
Teughels W, Loozen G, Quirynen M (2011) Do probiotics offer opportunities to manipulate the periodontal oral microbiota? J Clin Periodontol 38 (Suppl. 1):158–176
Schlundt J. (2001) Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria. Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria (2001) [cited 2013 22 February]; Available from: http://www.who.int/foodsafety/ publications/fs_management/en/probiotics.pdf
Broekaert IJ, Walker WA (2006) Probiotics and chronic disease. J Clin Gastroenterol 40:270–274
Jones SE (2009) Versalovic J (2009) Probiotic Lactobacillus reuteri biofilms produce antimicrobial and anti-inflammatory factors. BMC Microbiol 9:35. https://doi.org/10.1186/1471-2180-9-35
Schaefer L, Auchtung TA, Hermans KE, Whitehead D, Borhan B, Britton RA (2010) The antimicrobial compound reuterin (3-hydroxypropionaldehyde) induces oxidative stress via interaction with thiol groups. Microbiology. 156(Pt 6):1589–1599. https://doi.org/10.1099/mic.0.035642-0 Epub 2010 Feb 11
Vivekananda MR, Vandana KL, Bhat KG (2010) Effect of the probiotic Lactobacilli reuteri (Prodentis) in the management of periodontal disease: a preliminary randomized clinical trial. J Oral Microbiol 2:1–10
Twetman, S., Derawi, B., Keller, M., Ekstrand,K. I. M., Yucel-lindberg, L. A. Y. & Steckse, C. (2009) Short-term effect of chewing gums containing probiotic Lactobacillus reuteri on the levels of inflammatory mediators in gingival crevicular fluid. Acta Odontol Scand 67, 19–24.
Tekce M, Ince G, Gursoy H, Dirikan Ipci S, Cakar G, Kadir T, Yilmaz S (2015) Clinical and microbiological effects of probiotic lozenges in the treatment of chronic periodontitis: a 1-year follow-up study. J Clin Periodontol 2015. https://doi.org/10.1111/jcpe.12387
Martin-Cabezas R, Davideau JL, Tenenbaum H, Huck O (2016) Clinical efficacy of probiotics as an adjunctive therapy to non-surgical periodontal treatment of chronic periodontitis: a systematic review and meta-analysis. J Clin Periodontol 43(6):520–530. https://doi.org/10.1111/jcpe.12545 Epub 2016 May 7
Ikram S, Hassan N, Raffat MA, Mirza S, Akram Z (2018) Systematic review and meta-analysis of double-blind, placebo-controlled, randomized clinical trials using probiotics in chronic periodontitis. J Investig Clin Dent e12338. https://doi.org/10.1111/jicd.12338
Teughels W, Durukan A, Ozcelik O, Pauwels M, Quirynen M, Haytac Mehmet C (2013) Clinical and microbiological effects of Lactobacillus reuteri probiotics in the treatment of chronic periodontitis: a randomized placebo controlledstudy. J Clin Periodontol 40:1025 1035
İnce G, Gürsoy H, İpçi ŞD, Cakar G, Emekli-Alturfan E, Yılmaz S (2015) Clinical and biochemical evaluation of lozenges containing Lactobacillus reuteri as an adjunct to nonsurgical periodontal therapy in chronic periodontitis. J Periodontol 86:746–754
Penala S, Kalakonda B, Pathakota KR et al (2016) Efficacy of local use of probiotics as an adjunct to scaling and root planing in chronic periodontitis and halitosis: a randomized controlled trial. J Res Pharm Pract 2016(5):86
Szkaradkiewicz AK, Stopa J, Karpiński TM (2014) Effect of oral administration involving a probiotic strain of Lactobacillus reuteri on pro-inflammatory cytokine response in patients with chronic periodontitis. Arch Immunol Ther Exp 62:495–500
Vicario M, Santos A, Violant D, Nart J, Giner L (2013) Clinical changes in periodontal subjects with the probiotic Lactobacillus reuteri Prodentis: a preliminary randomized clinical trial. Acta Odontol Scand 71:813–819
Armitage GC (1999) Development of a classification system for periodontal diseases and conditions. Ann Periodontol 4(1):1–6
Caton JG, Armitage G, Berglundh T, Chapple ILC, Jepsen S, Kornman KS, Mealey BL, Papapanou PN, Sanz M, Tonetti MS (2018) A new classification scheme for periodontal and peri-implant diseases and conditions - Introduction and key changes from the 1999 classification. J Periodontol 89(Suppl 1):S1–S8. https://doi.org/10.1002/JPER.18-0157
Claffey N (1995) Egelberg J (1995) Clinical indicators of probing attachment loss following initial periodontal treatment in advanced periodontitis patients. J Clin Periodontol 22:690–696
Lindhe Jan (1989) Examination of patients with periodontal disease. In Lindhe Jan Textbook of Clinical Periodontology. Blackwell Munksgaard, Copenhagen, Denmark, pp 316
Mensi M, Dalè E, Sordillo A (2017) A new method for biofilm removal with erythritol powder guided by disclosing agent. Dental Cadmos 85(2):112–118
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Lawrence Earlbaum Associates, Hillsdale, NJ
Faul F, Erdfelder E, Lang A-G, Buchner A (2007) G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav Res Methods 39:175–191
Liu GF, Lu K, Mogg R, Mallick M, Mehrotra DV (2009) Should baseline be a covariate or dependent variable in analyses of change from baseline in clinical trials? Stat Med 28:2509–2530
Baumer A, Pretzl B, Cosgarea R, Kim TS, Reitmeir P, EickholzP, Dannewitz B. (2011) Tooth loss in aggressive periodontitis after active periodontal therapy: patient-related and tooth-related prognostic factors. J Clin Periodontol 2011:38: 644–651.
Iwasaki K, Maeda K, Hidaka K, Nemoto K, Hirose Y, Deguchi S (2016) (2016) Daily intake of heat-killed Lactobacillus plantarum L-137 decreases the probing depth in patients undergoing supportive periodontal therapy. Oral Health Prev Dent 14(3):207–214. https://doi.org/10.3290/j.ohpd.a36099
Teughels W, Van Essche M, Sliepen I, Quirynen M (2008) Probiotics and oral healthcare. Periodontol 48:111–147
Sacristan JA (2013) Patient-centered medicine and patient-oriented research: Improving health outcomes for individual patients. BMC Med Informat Dec Making 13:6 Salanti, G., Ades, A. E., & Ioannidis, J. P. (2011)
Gwaltney CJ (2010) Patient-reported outcomes (PROs) in dental clinical trials and product development: introduction to scientific and regulatory considerations. J Evidence Based Dent Pract Vol 10,Issue 2, pag.86–90
Smiley CJ, Tracy SL, Abt E, Michalowicz BS, John MT, Gunsolley J, Cobb CM, Rossmann J, Harrel SK, Forrest JL, Hujoel PP, Noraian KW, Greenwell H, Frantsve-Hawley J, Estrich C, Hanson N (2015) Evidence-based clinical practice guideline on the nonsurgical treatment of chronic periodontitis by means of scaling and root planing with or without adjuncts. J Am Dent Assoc 146(7):525–535. https://doi.org/10.1016/j.adaj.2015.01.026
Sgolastra F, Petrucci A, Gatto R, Monaco A (2012) Effectiveness of systemic amoxicillin/metronidazole as an adjunctive therapy to full-mouth scaling and root planing in the treatment of aggressive periodontitis: a systematic review and meta-analysis. J Periodontol 83(6):731–743. https://doi.org/10.1902/jop.2011.110432
Shah MP, Gujjari SK, Chandrasekhar VS (2013) Evaluation of the effect of probiotic (inersan®) alone, combination of probiotic with doxycycline and doxycycline alone on aggressive periodontitis - a clinical and microbiological study. J Clin Diagn Res 7(3):595–600. https://doi.org/10.7860/JCDR/2013/5225.2834
Funding
The study was partially funded with a research grant from NOOS that provided free the probiotic involved in the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. Author MGG has received a research grant from company Noos, who provided to the University free the probiotic used in the study. However, data belonged to the authors and by no means, the company interfered with the conduct of the study
Ethical approval
All procedure involving humans have been approved by the institutional ethics committee of the University of Brescia (protocol n 0059683 NP 1473) and have been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Grusovin, M.G., Bossini, S., Calza, S. et al. Clinical efficacy of Lactobacillus reuteri-containing lozenges in the supportive therapy of generalized periodontitis stage III and IV, grade C: 1-year results of a double-blind randomized placebo-controlled pilot study. Clin Oral Invest 24, 2015–2024 (2020). https://doi.org/10.1007/s00784-019-03065-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03065-x