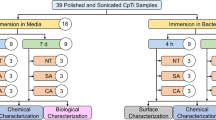
Abstract
Objectives
Biomaterial surfaces are at high risk for initial microbial colonization, persistence, and concomitant infection. The rationale of this study was to assess the initial adhesion on novel implant surfaces of Enterococcus faecalis, Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans upon incubation.
Materials and methods
The tested samples were 3 mol% yttria-stabilized tetragonal zirconia polycrystal (3Y-TZP) samples with nitrogen-doped hydrogenated amorphous carbon (a-C:H:N) coating (A) and 3Y-TZP samples coated with ceria-stabilized zirconia-based (Ce-TZP) composite and a-C:H:N (B). Uncoated 3Y-TZP samples (C) and bovine enamel slabs (BES) served as controls. Once the surface was characterized, the adherent microorganisms were quantified by estimating the colony-forming units (CFUs). Microbial vitality was assessed by live/dead staining, and microbial-biomaterial surface topography was visualized by scanning electron microscopy (SEM).
Results
Overall, A and B presented the lowest CFU values for all microorganisms, while C sheltered significantly less E. faecalis, P. aeruginosa, and C. albicans than BES. Compared to the controls, B demonstrated the lowest vitality values for E. coli (54.12 %) and C. albicans (67.99 %). Interestingly, A (29.24 %) exhibited higher eradication rates for S. aureus than B (13.95 %).
Conclusions
Within the limitations of this study, a-C:H:N-coated 3Y-TZP surfaces tended to harbor less initially adherent microorganisms and selectively interfered with their vitality.
Clinical relevance
This could enable further investigation of the new multi-functional zirconia surfaces to confirm their favorable antimicrobial properties in vivo.






Similar content being viewed by others
References
Wenz HJ, Bartsch J, Wolfart S, Kern M (2008) Osseointegration and clinical success of zirconia dental implants: a systematic review. Int J Prosthodont 21(1):27–36
Kohal RJ, Patzelt SB, Butz F, Sahlin H (2013) One-piece zirconia oral implants: one-year results from a prospective case series. 2. Three-unit fixed dental prosthesis (FDP) reconstruction. J Clin Periodontol 40(5):553–562. doi:10.1111/jcpe.12093
Bellucci D, Sola A, Cannillo V (2014) Bioactive glass/ZrO2 composites for orthopaedic applications. Biomed Mater 9(1):015005. doi:10.1088/1748-6041/9/1/015005
Roy ME, Whiteside LA, Katerberg BJ, Steiger JA (2007) Phase transformation, roughness, and microhardness of artificially aged yttria- and magnesia-stabilized zirconia femoral heads. J Biomed Mater Res A 83(4):1096–1102. doi:10.1002/jbm.a.31438
Kohal RJ, Klaus G, Strub JR (2006) Zirconia-implant-supported all-ceramic crowns withstand long-term load: a pilot investigation. Clin Oral Implants Res 17(5):565–571. doi:10.1111/j.1600-0501.2006.01252.x
Camposilvan E, Marro FG, Mestra A, Anglada M (2015) Enhanced reliability of yttria-stabilized zirconia for dental applications. Acta Biomater 17:36–46. doi:10.1016/j.actbio.2015.01.023
Cotes C, Arata A, Melo RM, Bottino MA, Machado JP, Souza RO (2014) Effects of aging procedures on the topographic surface, structural stability, and mechanical strength of a ZrO-based dental ceramic. Dent Mater. doi:10.1016/j.dental.2014.08.380
Benzaid R, Chevalier J, Saadaoui M, Fantozzi G, Nawa M, Diaz LA, Torrecillas R (2008) Fracture toughness, strength and slow crack growth in a ceria stabilized zirconia-alumina nanocomposite for medical applications. Biomaterials 29(27):3636–3641. doi:10.1016/j.biomaterials.2008.05.021
Chevalier J (2006) What future for zirconia as a biomaterial? Biomaterials 27(4):535–543. doi:10.1016/j.biomaterials.2005.07.034
Chevalier J, Deville S, Munch E, Jullian R, Lair F (2004) Critical effect of cubic phase on aging in 3mol% yttria-stabilized zirconia ceramics for hip replacement prosthesis. Biomaterials 25(24):5539–5545. doi:10.1016/j.biomaterials.2004.01.002
Hallmann L, Mehl A, Ulmer P, Reusser E, Stadler J, Zenobi R, Stawarczyk B, Ozcan M, Hammerle CH (2012) The influence of grain size on low-temperature degradation of dental zirconia. J Biomed Mater Res B Appl Biomater 100(2):447–456. doi:10.1002/jbm.b.31969
Sanon C, Chevalier J, Douillard T, Kohal RJ, Coelho PG, Hjerppe J, Silva NR (2013) Low temperature degradation and reliability of one-piece ceramic oral implants with a porous surface. Dent Mater 29(4):389–397. doi:10.1016/j.dental.2013.01.007
Riool M, de Boer L, Jaspers V, van der Loos CM, van Wamel WJ, Wu G, Kwakman PH, Zaat SA (2014) Staphylococcus epidermidis originating from titanium implants infects surrounding tissue and immune cells. Acta Biomater. doi:10.1016/j.actbio.2014.08.012
Madianos PN, Bobetsis YA, Kinane DF (2005) Generation of inflammatory stimuli: how bacteria set up inflammatory responses in the gingiva. J Clin Periodontol 32(Suppl 6):57–71. doi:10.1111/j.1600-051X.2005.00821.x
Lindhe J, Meyle J (2008) Peri-implant diseases: consensus report of the sixth european workshop on periodontology. J Clin Periodontol 35(8 Suppl):282–285. doi:10.1111/j.1600-051X.2008.01283.x
Charalampakis G, Leonhardt A, Rabe P, Dahlen G (2012) Clinical and microbiological characteristics of peri-implantitis cases: a retrospective multicentre study. Clin Oral Implants Res 23(9):1045–1054. doi:10.1111/j.1600-0501.2011.02258.x
Smeets R, Henningsen A, Jung O, Heiland M, Hammacher C, Stein JM (2014) Definition, etiology, prevention and treatment of peri-implantitis—a review. Head Face Med 10:34. doi:10.1186/1746-160x-10-34
Zhuang LF, Watt RM, Mattheos N, Si MS, Lai HC, Lang NP (2014) Periodontal and peri-implant microbiota in patients with healthy and inflamed periodontal and peri-implant tissues. Clin Oral Implants Res. doi:10.1111/clr.12508
Rams TE, Degener JE, van Winkelhoff AJ (2014) Antibiotic resistance in human peri-implantitis microbiota. Clin Oral Implants Res 25(1):82–90. doi:10.1111/clr.12160
Bremer F, Grade S, Kohorst P, Stiesch M (2011) In vivo biofilm formation on different dental ceramics. Quintessence Int 42(7):565–574
Karygianni LJA, Schienle S, Bernsmann F, Adolfsson E, Kohal RJ, Chevalier J, Hellwig E, Al-Ahmad A (2013) Initial bacterial adhesion on different yttria-stabilized tetragonal zirconia implant surfaces in vitro. Mater 6:5659–5674
Socransky SS, Haffajee AD (2000) Dental biofilms: difficult therapeutic targets. Periodontol 28:12–55
Katsikogianni M, Missirlis YF (2004) Concise review of mechanisms of bacterial adhesion to biomaterials and of techniques used in estimating bacteria-material interactions. Eur Cell Mater 8:37–57
Roy RK, Lee KR (2007) Biomedical applications of diamond-like carbon coatings: a review. J Biomed Mater Res B Appl Biomater 83(1):72–84. doi:10.1002/jbm.b.30768
Dorner-Reisel A, Gartner G, Reisel G, Irmer G (2008) Diamond-like carbon films for polyethylene femoral parts: Raman and FT-IR spectroscopy before and after incubation in simulated body liquid. Anal Bioanal Chem 390(6):1487–1493. doi:10.1007/s00216-007-1744-7
Lappalainen R, Heinonen H, Anttila A, Santavirta S (1998) Some relevant issues related to the use of amorphous diamond coatings for medical applications. Diamond Relat Mater 7:482–485
Almaguer-Flores A, Ximenez-Fyvie LA, Rodil SE (2010) Oral bacterial adhesion on amorphous carbon and titanium films: effect of surface roughness and culture media. J Biomed Mater Res B Appl Biomater 92(1):196–204. doi:10.1002/jbm.b.31506
Hauert R, Glisenti A, Metin S, Goitia J, Kaufman JH, van Loosdrecht PHM, Kellock AJ, Hoffmann P, White RL, Hermsmeier BD (1995) Influence of nitrogen doping on different properties of a-C:H. Thin Solid Films 268:22–29. doi:10.1016/0040-6090(95)06824-4
Tripathi RK, Panwar OS, Srivastava AK, Rawal I, Chockalingam S (2014) Structural, nanomechanical, field emission and ammonia gas sensing properties of nitrogenated amorphous carbon films deposited by filtered anodic jet carbon arc technique. Talanta 125:276–283. doi:10.1016/j.talanta.2014.03.005
Palmero P, Kern F, Sommer F, Lombardi M, Gadow R, Montanaro L (2014) Issues in nanocomposite ceramic engineering: focus on processing and properties of alumina-based composites. J Appl Biomater Funct Mater 12(3):e113–128. doi:10.5301/jabfm.5000185
Tawakoli PN, Al-Ahmad A, Hoth-Hannig W, Hannig M, Hannig C (2013) Comparison of different live/dead stainings for detection and quantification of adherent microorganisms in the initial oral biofilm. Clin Oral Investig 17(3):841–850. doi:10.1007/s00784-012-0792-3
Busscher HJ, Rinastiti M, Siswomihardjo W, van der Mei HC (2010) Biofilm formation on dental restorative and implant materials. J Dent Res 89(7):657–665. doi:10.1177/0022034510368644
Al-Radha AS, Dymock D, Younes C, O’Sullivan D (2012) Surface properties of titanium and zirconia dental implant materials and their effect on bacterial adhesion. J Dent 40(2):146–153. doi:10.1016/j.jdent.2011.12.006
de Oliveira GR, Pozzer L, Cavalieri-Pereira L, de Moraes PH, Olate S, de Albergaria Barbosa JR (2012) Bacterial adhesion and colonization differences between zirconia and titanium implant abutments: an in vivo human study. J Periodontal Implant Sci 42(6):217–223. doi:10.5051/jpis.2012.42.6.217
Al-Ahmad A, Wiedmann-Al-Ahmad M, Fackler A, Follo M, Hellwig E, Bachle M, Hannig C, Han JS, Wolkewitz M, Kohal R (2013) In vivo study of the initial bacterial adhesion on different implant materials. Arch Oral Biol 58(9):1139–1147. doi:10.1016/j.archoralbio.2013.04.011
Nascimento C, Pita MS, Fernandes FH, Pedrazzi V, de Albuquerque Junior RF, Ribeiro RF (2014) Bacterial adhesion on the titanium and zirconia abutment surfaces. Clin Oral Implants Res 25(3):337–343. doi:10.1111/clr.12093
Al-Ahmad A, Wiedmann-Al-Ahmad M, Carvalho C, Lang M, Follo M, Braun G, Wittmer A, Mulhaupt R, Hellwig E (2008) Bacterial and Candida albicans adhesion on rapid prototyping-produced 3D-scaffolds manufactured as bone replacement materials. J Biomed Mater Res A 87(4):933–943. doi:10.1002/jbm.a.31832
Hannig C, Hannig M, Rehmer O, Braun G, Hellwig E, Al-Ahmad A (2007) Fluorescence microscopic visualization and quantification of initial bacterial colonization on enamel in situ. Arch Oral Biol 52(11):1048–1056. doi:10.1016/j.archoralbio.2007.05.006
Karygianni L, Follo M, Hellwig E, Burghardt D, Wolkewitz M, Anderson A, Al-Ahmad A (2012) Microscope-based imaging platform for large-scale analysis of oral biofilms. Appl Environ Microbiol 78(24):8703–8711. doi:10.1128/aem.02416-12
Teles FR, Teles RP, Sachdeo A, Uzel NG, Song XQ, Torresyap G, Singh M, Papas A, Haffajee AD, Socransky SS (2012) Comparison of microbial changes in early redeveloping biofilms on natural teeth and dentures. J Periodontol 83(9):1139–1148. doi:10.1902/jop.2012.110506
Del Carpio-Perochena A, Kishen A, Shrestha A, Bramante CM (2015) Antibacterial properties associated with chitosan nanoparticle treatment on root dentin and 2 types of endodontic sealers. J Endod 41(8):1353–1358. doi:10.1016/j.joen.2015.03.020
Avila ED, Molon RS, Lima B, Lux R, Shi W, Jafelicci Junior M, Spolidorio D, Vergani CE, Mollo Junior F (2015) The impact of physical chemical characteristics of implant abutment surfaces on bacteria adhesion. J Oral Implantol. doi:10.1563/aaid-joi-D-14-00318
Aguayo S, Donos N, Spratt D, Bozec L (2015) Nanoadhesion of Staphylococcus aureus onto titanium implant surfaces. J Dent Res 94(8):1078–1084. doi:10.1177/0022034515591485
Davidson H, Poon M, Saunders R, Shapiro IM, Hickok NJ, Adams CS (2014) Tetracycline tethered to titanium inhibits colonization by gram-negative bacteria. J Biomed Mater Res B Appl Biomater. doi:10.1002/jbm.b.33310
Wood SR, Kirkham J, Marsh PD, Shore RC, Nattress B, Robinson C (2000) Architecture of intact natural human plaque biofilms studied by confocal laser scanning microscopy. J Dent Res 79(1):21–27
Crawford RJ, Webb HK, Truong VK, Hasan J, Ivanova EP (2012) Surface topographical factors influencing bacterial attachment. Adv Colloid Interface Sci 179–182:142–149. doi:10.1016/j.cis.2012.06.015
Gustumhaugen E, Lonn-Stensrud J, Scheie AA, Lyngstadaas SP, Ekfeldt A, Taxt-Lamolle S (2014) Effect of chemical and mechanical debridement techniques on bacterial re-growth on rough titanium surfaces: an in vitro study. Clin Oral Implants Res 25(6):707–713. doi:10.1111/clr.12130
Katsikogianni M, Spiliopoulou I, Dowling DP, Missirlis YF (2006) Adhesion of slime producing Staphylococcus epidermidis strains to PVC and diamond-like carbon/silver/fluorinated coatings. J Mater Sci Mater Med 17(8):679–689. doi:10.1007/s10856-006-9678-8
Kiremitci-Gumusderelioglu M, Pesmen A (1996) Microbial adhesion to ionogenic PHEMA, PU and PP implants. Biomaterials 17(4):443–449
Van der Mei HC, Bos R, Busscher HJ (1998) A reference guide to microbial cell surface hydrophobicity based on contact angles. Colloids Surf B 11:213–221
Teughels W, Van Assche N, Sliepen I, Quirynen M (2006) Effect of material characteristics and/or surface topography on biofilm development. Clin Oral Implants Res 17(Suppl 2):68–81. doi:10.1111/j.1600-0501.2006.01353.x
Bazaka K, Jacob MV, Crawford RJ, Ivanova EP (2012) Efficient surface modification of biomaterial to prevent biofilm formation and the attachment of microorganisms. Appl Microbiol Biotechnol 95(2):299–311. doi:10.1007/s00253-012-4144-7
Ito H, Sasaki H, Saito K, Honma S, Yajima Y, Yoshinari M (2013) Response of osteoblast-like cells to zirconia with different surface topography. Dent Mater J 32(1):122–129
Belibasakis GN, Charalampakis G, Bostanci N, Stadlinger B (2015) Peri-implant infections of oral biofilm etiology. Adv Exp Med Biol 830:69–84. doi:10.1007/978-3-319-11038-7_4
Berglundh T, Gotfredsen K, Zitzmann NU, Lang NP, Lindhe J (2007) Spontaneous progression of ligature induced peri-implantitis at implants with different surface roughness: an experimental study in dogs. Clin Oral Implants Res 18(5):655–661. doi:10.1111/j.1600-0501.2007.01397.x
Lang NP, Berglundh T (2011) Periimplant diseases: where are we now?—consensus of the seventh european workshop on periodontology. J Clin Periodontol 38(Suppl 11):178–181. doi:10.1111/j.1600-051X.2010.01674.x
Salvi GE, Furst MM, Lang NP, Persson GR (2008) One-year bacterial colonization patterns of Staphylococcus aureus and other bacteria at implants and adjacent teeth. Clin Oral Implants Res 19(3):242–248. doi:10.1111/j.1600-0501.2007.01470.x
Ribeiro M, Monteiro FJ, Ferraz MP (2012) Infection of orthopedic implants with emphasis on bacterial adhesion process and techniques used in studying bacterial-material interactions. Biomatter 2(4):176–194. doi:10.4161/biom.22905
Kayaoglu G, Orstavik D (2004) Virulence factors of Enterococcus faecalis: relationship to endodontic disease. Crit Rev Oral Biol Med 15(5):308–320
Burgers R, Hahnel S, Reichert TE, Rosentritt M, Behr M, Gerlach T, Handel G, Gosau M (2010) Adhesion of Candida albicans to various dental implant surfaces and the influence of salivary pellicle proteins. Acta Biomater 6(6):2307–2313. doi:10.1016/j.actbio.2009.11.003
Furst MM, Salvi GE, Lang NP, Persson GR (2007) Bacterial colonization immediately after installation on oral titanium implants. Clin Oral Implants Res 18(4):501–508. doi:10.1111/j.1600-0501.2007.01381.x
Balasubramanian D, Schneper L, Kumari H, Mathee K (2013) A dynamic and intricate regulatory network determines Pseudomonas aeruginosa virulence. Nucleic Acids Res 41(1):1–20. doi:10.1093/nar/gks1039
Khan MS, Vishakante GD (2013) Development and evaluation of porous chitosan nanoparticles for treatment of enterotoxigenic Escherichia coli infection. J Biomed Nanotechnol 9(1):107–114
Wessel SW, Chen Y, Maitra A, van den Heuvel ER, Slomp AM, Busscher HJ, van der Mei HC (2014) Adhesion forces and composition of planktonic and adhering oral microbiomes. J Dent Res 93(1):84–88. doi:10.1177/0022034513511822
Chin MY, Sandham A, de Vries J, van der Mei HC, Busscher HJ (2007) Biofilm formation on surface characterized micro-implants for skeletal anchorage in orthodontics. Biomaterials 28(11):2032–2040. doi:10.1016/j.biomaterials.2006.12.014
Mei L, Ren Y, Busscher HJ, Chen Y, van der Mei HC (2009) Poisson analysis of streptococcal bond-strengthening on saliva-coated enamel. J Dent Res 88(9):841–845. doi:10.1177/0022034509342523
Busscher HJ, van der Mei HC (2012) How do bacteria know they are on a surface and regulate their response to an adhering state? PLoS Pathog 8(1):e1002440. doi:10.1371/journal.ppat.1002440
Kugler R, Bouloussa O, Rondelez F (2005) Evidence of a charge-density threshold for optimum efficiency of biocidal cationic surfaces. Microbiology 151(Pt 5):1341–1348. doi:10.1099/mic.0.27526-0
Murata H, Koepsel RR, Matyjaszewski K, Russell AJ (2007) Permanent, non-leaching antibacterial surface—2: how high density cationic surfaces kill bacterial cells. Biomaterials 28(32):4870–4879. doi:10.1016/j.biomaterials.2007.06.012
Hauser-Gerspach I, Kulik EM, Weiger R, Decker EM, Von Ohle C, Meyer J (2007) Adhesion of Streptococcus sanguinis to dental implant and restorative materials in vitro. Dent Mater J 26(3):361–366
Rimondini L, Cerroni L, Carrassi A, Torricelli P (2002) Bacterial colonization of zirconia ceramic surfaces: an in vitro and in vivo study. Int J Oral Maxillofac Implants 17(6):793–798
Liu C, Zhao Q, Liu Y, Wang S, Abel EW (2008) Reduction of bacterial adhesion on modified DLC coatings. Colloids Surf B Biointerfaces 61(2):182–187. doi:10.1016/j.colsurfb.2007.08.008
Acknowledgments
The authors express their gratitude to Anja Stork and Bettina Spitzmüller for their excellent technical help. Dr. Marie Follo and Dr. Annette Anderson are thanked for their valuable linguistic contribution to this report and support by image processing, respectively. This study was supported by the European Commission (FP7-280741-LONGLIFE Collaborative project).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study protocol was reviewed and approved by the Ethics Committee of the University of Freiburg (Nr. 91/15).
Conflicts of interest
There are no conflicts of interest.
Rights and permissions
About this article
Cite this article
Schienle, S., Al-Ahmad, A., Kohal, R.J. et al. Microbial adhesion on novel yttria-stabilized tetragonal zirconia (Y-TZP) implant surfaces with nitrogen-doped hydrogenated amorphous carbon (a-C:H:N) coatings. Clin Oral Invest 20, 1719–1732 (2016). https://doi.org/10.1007/s00784-015-1655-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-015-1655-5