Abstract
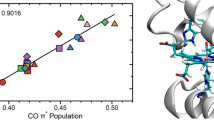
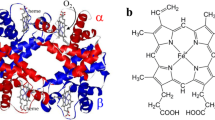
The physiological significance of steric inhibition of CO binding to heme proteins is a fundamentally quantitative issue, since it has meaning only relative to the enhancement of O2 binding by the protein. Previously, difficulties in reconciling structural and energetic data have hindered a quantitative assessment of the energetic cost of distorting the bound CO. Recent progress on both fronts suggests that the energetic cost of CO distortion in myoglobin is quite small. However, distortion of the surrounding protein to accommodate the heme-CO complex may contribute significantly to the free energy of CO binding. Polarized IR measurements on single crystals of MbCO not only yield precise structural information on the CO geometry, but also address the limitations of conventional structural refinement by characterizing conformational disorder in the crystalline state.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received, accepted: 23 May 1997
Rights and permissions
About this article
Cite this article
Sage, J. Myoglobin and CO: structure, energetics, and disorder. JBIC 2, 537–543 (1997). https://doi.org/10.1007/s007750050168
Issue Date:
DOI: https://doi.org/10.1007/s007750050168