Abstract
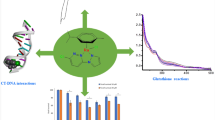
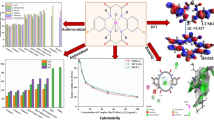
It is now well established that ruthenium complexes are attractive alternatives to platinum-based anticancer agents. Most of the ruthenium compounds currently under investigation contain a single metal center. The synthesis of multinuclear analogues may provide access to novel complexes with enhanced biological activity. In this work, we have synthesized a set of three trinuclear complexes containing organometallic ruthenium fragments—(arene)RuCl—coordinated to a 2,4,6-tris(di-2-pyridylamino)-1,3,5-triazine core [(Arene = benzene (2), p-cymene (1), or hexamethylbenzene (3)]. The interaction of the complexes with DNA was extensively studied using a variety of biophysical probes as well as by molecular docking. The complexes bind strongly to DNA with apparent binding constants ranging from 2.20 to 4.79 × 104 M−1. The binding constants from electronic absorption titrations were an order of magnitude greater. The mode of binding to the nucleic acid was not definitively determined, but the evidence pointed to some kind of non-specific electrostatic interaction. None of the complexes displayed any significant antimicrobial activity against the organisms that were studied and exhibited anticancer activity only at high (> 100 μM) concentration.








Similar content being viewed by others
References
Kostova I (2006) Curr Med Chem 13:1085–1107
Levina A, Mitra A, Lay PA (2009) Metallomics 1:458–470
Bergamo A, Sava G (2011) Dalton Trans 40:7817–7823
Műhlgassner G, Bartel C, Schmid WF, Jakupec MA, Arion VB, Keppler BK (2012) J Inorg Biochem 116:180–187
Bruijnincx PCA, Sadler PJ (2008) Curr Opin Chem Biol 12:197–206
Meier SM, Hanif M, Kandioller W, Keppler BK, Hartinger CG (2012) J Inorg Biochem 108:91–95
Antonarakis ES, Emadi A (2010) Cancer Chemother Pharmacol 66:1–9
Ang WH, Dyson PJ (2006) Eur J Inorg Chem 20:4003–4018
Pitto-Barry A, Barry NPE, Zava O, Deschenaux R, Therrien B (2011) Chem Asian J 6:1595–1603
Mattsson J, Govindaswamy P, Renfrew AK, Dyson PJ, Štěpnička P, Süss-Fink G, Therrien B (2009) Organometallics 28:4350–4357
Therrien B, Furrer J (2014) Adv Chem
Mishra A, Jung H, Park JW, Kim HK, Kim H, Stang PJ, Chi KW (2012) Organometallics 31:3519–3526
Linares F, Procopio EQ, Galindo MA, Romero MA, Navarro JAR, Barea E (2010) CrystEngComm 12:2343–2346
Vajpayee V, Song YH, Yang YJ, Kang SC, Kim H, Kim IS, Wang M, Stang PJ, Chi KW (2011) Organometallics 30:3242–3245
Furrer MA, Schmitt F, Wiederkehr M, Juillerat-Jeanneret L, Therrien B (2012) Dalton Trans 41:7201–7211
Vajpayee V, Yang YJ, Kang SC, Kim H, Kim IS, Wang M, Stang PJ, Chi KW (2011) Chem Commun 47:5184–5186
Schmitt F, Govindaswamy P, Suss-Fink G, Ang WH, Dyson PJ, Juillerat-Jeanneret L, Therrien B (2008) J Med Chem 51:1811–1816
Maeda H, Matsumura Y (1986) Cancer Res 46:6387–6392
Maeda H, Bharate GY, Daruwalla J (2009) Eur J Pharm Biopharm 71:409–419
Allardyce CS, Dyson PJ, Ellis DJ, Salter PA, Scopelliti R (2003) J Organomet Chem 668:35–42
Therrien B, Süss-Fink G, Govindaswamy P, Renfrew AK, Dyson PJ (2008) Angew Chem Int Ed 47:3773–3776
Rao ABP, Umab A, Chiranjeevi T, Bethu MS, Yashwanth B, Venkateswara Rao J, Poluri KM, Kollipara MR (2016) Inorg Chim Acta 353:284–291
Pang J, Tao Y, Freiberg S, Yang X-P, D’Iorioa M, Wang S (2002) J Mater Chem 12:206–212
Reichmann ME, Rice SA, Thomas CA, Doty PJ (1954) J Am Chem Soc 76:3047–3053
Beckford FA, Stott A, Gonzalez-Sarrías A, Seeram NP (2013) Appl Organometal Chem 27:425–434
Beckford FA, Webb KR (2017) Spectrochim Acta Part A 183:158–171
Auzias M, Therrien B, Suss-Fink G, Stepnicka P, Ang WH, Dyson PJ (2008) Inorg Chem 47:578–583
Ang WH, Daldini E, Scolaro C, Scopelliti R, Juillerat-Jeannerat L, Dyson PJ (2006) Inorg Chem 45:9006–9013
Therrien B, Suss-Fink G, Govindaswamy P, Renfrew AK, Dyson PJ (2008) Angew Chem Int Ed Engl 47:3773–3776
Chen HM, Parkinson JA, Morris RE, Sadler PJ (2003) J Am Chem Soc 125:173–186
Wang F, Chen H, Parsons S, Oswald IDH, Davidson JE, Sadler PJ (2003) Chem Eur J 9:5810–5820
Hanif M, Meier SM, Kandioller W, Bytzek A, Hejl M, Hartinger CG, Nazarov AA, Arion VB, Jakupec MA, Dyson PJ, Keppler BK (2011) J Inorg Biochem 105:224–231
Hanif M, Henke H, Meier SM, Martic S, Labib M, Kandioller W, Jakupec A, Arion VB, Kraatz HB, Keppler BK, Hartinger CG (2010) Inorg Chem 49:7953–7963
Hanif A, Nazarov AA, Hartinger CG, Kandioller W, Jakupec MA, Arion VB, Dyson PJ, Keppler BK (2010) Dalton Trans 39:7345–7352
Ambroise A, Maiya BG (2000) Inorg Chem 39:4256–4263
Jiang M, Li YT, Wub ZY, Liu ZQ, Yan CW (2009) J Inorg Biochem 103:833–844
Liu Q, Zhang J, Wang MQ, Zhang DW, Lua QS, Huang Y, Lin HH, Yu XQ (2010) Eur J Med Chem 45:5302–5308
Lacowicz JR (2006) Principles of fluorescence spectroscopy, 3rd edn. Springer, New York
Ghosh KS, Sahoo BK, Jana D, Dasgupta S (2008) J Inorg Biochem 102:1711–1718
Peberdy JP, Malina J, Khalid S, Haman MJ, Rodger A (2007) J Inorg Biochem 101:1937–1945
Friedman AE, Chambron JC, Sauvage JP, Turro NJ, Barton JK (1990) J Am Chem Soc 112:4960–4962
Ni Y, Lin D, Kokot S (2006) Anal Biochem 352:231–242
Pang DW, Abruna HD (1998) Anal Chem 70:3162–3169
Feng Q, Li NQ, Jiang YY (1997) Anal Chim Acta 344:97–104
Carter MT, Bard AJ (1987) J Am Chem Soc 109:7528–7530
Long EC, Barton JK (1990) Acc Chem Res 23:271–273
Carter MT, Rodriguez M, Bard AJ (1989) J Am Chem Soc 111:8901–8911
Aslanoglu M (2006) Anal Sci 22:439–443
Li N, Ma Y, Yang C (2005) Biophys Chem 116:199–205
Ibrahim MS (2001) Anal Chim Acta 443:63–72
Shah A, Qureshi R, Khan AM, Khera RA, Ansari FL (2010) J Braz Chem Soc 21:447–451
Radi A, El Ries MA, Kandil S (2003) Anal Chim Acta 495:61–67
Guin PS, Mandal PC, Das S (2012) ChemPlusChem 77:361–369
Welch TW, Corbett AH, Thorp HH (1995) J Phys Chem 99:11757–11763
Satyanarayana S, Dabrowiak JC, Chaires JB (1992) Biochemistry 31:9319–9324
Pravin N, Kumaravel G, Senthilkumar R, Raman N (2017) Appl Organometal Chem 31:e3739
Chouai A, Wick SE, Turro C, Bacsa J, Dunbar KR, Thummel RP (2005) Inorg Chem 44:5996–6003
Roy M, Santhanagopal R, Chakravarty AR (2009) Dalton Trans 6:1024–1033
Acknowledgements
MBN would like to thank the Department of Natural Sciences at the University of Virginia’s College at Wise, for a Fellowship in the Natural Sciences to carry out this work. The authors would also like to thank CO-ADD (University of Queensland) for the antimicrobial assays along with Eastman Chemicals, Kingsport, TN, and Bruker Biospin for the NMR analyses. AAH would like to thank the National Science Foundation (NSF) for the NSF CAREER Award, as this material is based upon work partially supported by the NSF under CHE-1431172 (Formerly CHE-1151832). AAH would also like to thank Old Dominion University’s Faculty Proposal Preparation Program (FP3) and the University of North Texas Health Science Center, Texas Center for Health Disparities Research Pilot Award, Steps Toward Academic Research (STAR) Fellowship Program (award numbers: R10078-2018-0053 and R10078-2018-0108) for also supporting this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Beckford, F.A., Niece, M.B., Lassiter, B.P. et al. Polynuclear ruthenium organometallic complexes containing a 1,3,5-triazine ligand: synthesis, DNA interaction, and biological activity. J Biol Inorg Chem 23, 1205–1217 (2018). https://doi.org/10.1007/s00775-018-1599-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-018-1599-8