Abstract
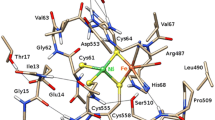
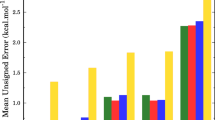
The [NiFe] hydrogenases catalyse the reversible conversion of H2 to protons and electrons. The active site consists of a Fe ion with one carbon monoxide, two cyanide, and two cysteine (Cys) ligands. The latter two bridge to a Ni ion, which has two additional terminal Cys ligands. It has been suggested that one of the Cys residues is protonated during the reaction mechanism. We have used combined quantum mechanical and molecular mechanics (QM/MM) geometry optimisations, large QM calculations with 817 atoms, and QM/MM free energy simulations, using the TPSS and B3LYP methods with basis sets extrapolated to the quadruple zeta level to determine which of the four Cys residues is more favourable to protonate for four putative states in the reaction mechanism, Ni-SIa, Ni-R, Ni-C, and Ni-L. The calculations show that for all states, the terminal Cys-546 residue is most easily protonated by 14–51 kJ/mol, owing to a more favourable hydrogen-bond pattern around this residue in the protein.









Similar content being viewed by others

References
Turner JA (1999) Science 285:687–689
Bockris JOM (2013) Int J Hydrogen Energy 38:2579–2588
Matsumoto T, Kim K, Nakai H, Hibino T, Ogo S (2013) Chem Cat Chem 5:1368–1373
Yang JY, Bullock M, DuBois MR, DuBois DL (2011) MRS Bull 36:39–47
Penner SS (2006) Energy 31:33–43
Cammack R, Frey M, Robson R (2001) Hydrogen as a fuel: learning from nature. Taylor Francis, Beijing
Lubitz W, Ogata H, Ruediger O, Reijerse E (2014) Chem Rev 114:4081–4148
Fontecilla-Camps JC, Volbeda A, Cavazza C, Nicolet Y (2007) Chem Rev 107:4273–4303
Higuchi Y, Ogata H, Miki K, Yasuoka N, Yagi T (1999) Structure 7:549–556
De Lacey AL, Fernandez VM, Rousset M, Cammack R (2007) Chem Rev 107:4304–4330
Lubitz W, Reijerse E, van Gastel M (2007) Chem Rev 107:4331–4365
Vincent KA, Parkin A, Armstrong FA (2007) Chem Rev 107:4366–4413
Tard C, Pickett CJ (2009) Chem Rev 109:2245–2274
Bakhmutov VI (2005) Eur J Inorg Chem 2005:245-255
Fan H-J, Hall MB (2001) J Biol Inorg Chem 6:467–473
Bruschi M, Zampella G, Fantucci P, De Gioia L (2005) Coord Chem Rev 249:1620–1640
Siegbahn PEM, Tye JW, Hall MB (2007) Chem Rev 107:4414–4435
Kampa M, Lubitz W, van Gastel M, Neese F (2012) J Biol Inorg Chem 17:1269–1281
Kraemer T, Kamp M, Lubitz W, van Gastel M, Neese F (2013) Chem Bio Chem 14:1898–1905
Bruschi M, Tiberti M, Guerra A, De Gioia L (2014) J Am Chem Soc 136:1803–1814
Pandelia M-E, Ogata H, Lubitz W (2010) Chem Phys Chem 11:1127–1140
Fichtner C, van Gastel M, Lubitz W (2003) Phys Chem Chem Phys 5:5507–5513
Medina M, Hatchikian EC, Cammack R (1996) Biochim Biophys Acta 1275:227–236
Whitehead JP, Gurbiel RJ, Bagyinka C, Hoffman BM, Maroney MJ (1993) J Am Chem Soc 115:5629–5635
van der Zwaan JW, Albracht SPJ, Fontijn RD, Slater EC (1985) FEBS Lett 179:271–277
Hidalgo R, Ash PA, Healy AJ, Vincent KA (2015) Angewandte Chemie Int Ed 54:7110–7113
Murphy BJ, Hidalgo R, Roessler MM, Evans RM, Ash PA, Myers WK, Vincent KA, Armstrong FA (2015) J Am Chem Soc 137:8484–8489
Pavlov M, Siegbahn PEM, Blomberg MRA, Crabtree RH (1998) J Am Chem Soc 120:548–555
Nilsson Lill SO, Siegbahn PEM (2009) Biochemistry 48:1056–1066
Volbeda A, Charon MH, Piras C, Hatchikian EC, Frey M, Fontecilla-Camps JC (1995) Nature 373:580–587
Matias PM, Soares CM, Saraiva LM, Coelho R, Morais J, Le Gall J, Carrondo MA (2001) J Biol Inorg Chem 6:63–81
Volbeda A, Fontecilla-Camps JC (2005) Coord Chem Rev 249:1609–1619
Galvan IF, Volbeda A, Fontecilla-Camps JC, Field MJ (2008) Proteins 73:95–203
Ogata H, Kramer T, Wang H, Schilter D, Pelmenschikov V, van Gastel M, Neese F, Rauchfuss TB, Gee LB, Scott AD, Yoda Y, Tanaka Y, Lubitz W, Cramer SP (2015) Nat Commun 6:7890
Pardo A, De Lacey AL, Fernandez VM, Fan HJ, Fan YB, Hall MB (2006) J Biol Inorg Chem 11:286–306
Wu H, Hall MB (2008) CR Chim 11:790–804
Amara P, Volbeda A, Fontecilla-Camps JC, Field MJ (1999) J Am Chem Soc 121:4468–4477
Stein M, Lubitz W (2004) J Inorg Biochem 98:862–877
Söderhjelm P, Ryde U (2006) J Mol Struc (Theochem) 770:199–219
Ryde U, Nilsson K (2003) J Mol Struc (Theochem) 632:259–275
Ryde U, Nilsson K (2003) J Am Chem Soc 125:14232–14233
Nilsson K, Ryde U (2004) J Inorg Biochem 98:1539–1546
Rod TH, Ryde U (2005) J Chem Theory Comput 1:1240–1251
Rod TH, Ryde U (2005) Phys Rev Lett 94:138302
Hu L, Eliasson J, Heimdal J, Ryde U (2009) J Phys Chem A 113:11793–11800
Hu L, Söderhjelm P, Ryde U (2011) J Chem Theory Comput 7:761–777
Hu L, Söderhjelm P, Ryde U (2013) J Chem Theory Comput 9:640–649
Sumner S, Söderhjelm P, Ryde U (2013) J Chem Theory Comput 9:4205–4214
Kaukonen M, Söderhjelm P, Heimdal J, Ryde U (2008) J Phys Chem B 112:12537–12548
Kaukonen M, Söderhjelm P, Heimdal J, Ryde U (2008) J Chem Theory Comput 4:985–1001
Delcey MG, Pierloot K, Phung QM, Vancoillie S, Lindh R, Ryde U (2014) Phys Chem Chem Phys 16:7927–7938
Ogata H, Nishikawa K, Lubitz W (2015) Nature 520:571–574
Olsson MHM, Søndergaard CR, Rostkowski M, Jensen JH (2011) J Chem Theory Comput 7:525–537
Gordon JC, Myers JB, Folta T, Shoja V, Heath LS, Onufriev A (2005) Nucl Acids Res 33:W368–W371
Case DA, Berryman JT, Betz RM, Cerutti DS, Cheatham TE III, Darden TA, Duke RE, Giese TJ, Gohlke H, Goetz AW, Homeyer N, Izadi S, Janowski P, Kaus J, Kovalenko A, Lee TS, LeGrand S, Li P, Luchko T, Luo R, Madej B, Merz KM, Monard G, Needham P, Nguyen H, Nguyen HT, Omelyan I, Onufriev A, Roe DR, Roitberg A, Salomon-Ferrer R, Simmerling CL, Smith W, Swails J, Walker RC, Wang J, Wolf RM, Wu X, York DM, Kollman PA (2014) AMBER 14. University of California, San Francisco
TURBOMOLE V65 2013, a development of University of Karlsruhe and Forschungszentrum Karlsruhe GmbH, 1989–2007, TURBOMOLE GmbH, since 2007. Available from http://www.turbomole.com
Tao J, Perdew JP, Staroverov VN, Scuseria GE (2003) Phys Rev Lett 91:146401
Becke AD (1993) J Chem Phys 98:5648–5652
Becke AD (1988) Phys Rev A 38:3098–3100
Lee CT, Yang WT, Parr RG (1988) Phys Rev B 37:785–789
Schäfer A, Horn H, Ahlrichs R (1992) J Chem Phys 97:2571–2577
Weigend F, Ahlrichs R (2005) Phys Chem Chem Phys 7:3297–3305
Rappoport D, Furche F (2010) J Chem Phys 133:134105
Eichkorn K, Treutler O, Öhm H, Häser M, Ahlrichs R (1995) Chem Phys Lett 240:283–290
Eichkorn K, Weigend F, Treutler O, Ahlrichs R (1997) Theor Chem Acc 97:119–124
Ryde U (1996) J Comput-Aided Mol Des 10:153–164
Ryde U, Olsson MHM (2001) Int J Quantum Chem 81:335–347
Reuter N, Dejaegere A, Maigret B, Karplus M (2000) J Phys Chem A 104:1720–1735
Svensson M, Humbel S, Froese RDJ, Matsubara T, Sieber S, Morokuma K (1996) J Phys Chem 100:19357–19363
dftd3 software http://www.thch.uni-bonn.de/tc/index.php?section=downloads&subsection=DFT-D3&lang=english
Maier JA, Martinez C, Kasavajhala K, Wickstrom L, Hauser KE, Simmerling C (2015) J Chem Theory Comput 11:3696–3713
Grimme S, Ehrlich S, Goerigk L (2011) J Comput Chem 32:1456–1465
Luzhkov V, Warshel A (1992) J Comput Chem 13:199–213
Heimdal J, Kaukonen M, Srnec M, Rulisek L, Ryde U (2011) Chem Phys Chem 12:3337–3347
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) J Chem Phys 79:926–935
Kampa M, Pandelia M-E, Lubitz W, van Gastel M, Neese F (2013) J Am Chem Soc 135:3915–3925
Jayapal P, Sundararajan M, Hillier IH, Burton NA (2008) Phys Chem Chem Phys 10:4249–4257
Acknowledgments
This investigation has been supported by grants from the Swedish research council (project 2014-5540), the China Scholarship Council, and COST through Action CM1305 (ECOSTBio). The computations were performed on computer resources provided by the Swedish National Infrastructure for Computing (SNIC) at Lunarc at Lund University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, G., Ryde, U. Protonation states of intermediates in the reaction mechanism of [NiFe] hydrogenase studied by computational methods. J Biol Inorg Chem 21, 383–394 (2016). https://doi.org/10.1007/s00775-016-1348-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-016-1348-9