Abstract
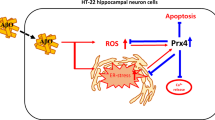
Aggregation and cytotoxicity of Aβ with redox-active metals in neuronal cells have been implicated in the progression of Alzheimer disease. Human metallothionein (MT) 3 is highly expressed in the normal human brain and is downregulated in Alzheimer disease. Zn7MT3 can protect against the neuronal toxicity of Aβ by preventing copper-mediated Aβ aggregation, abolishing the production of reactive oxygen species (ROS) and the related cellular toxicity. In this study, we intended to decipher the roles of single-domain proteins (α/β) and the α–β domain–domain interaction of Zn7MT3 to determine the molecular mechanism for protection against the neuronal cytotoxicity of Aβ1–42 with copper ions. With this in mind, the α and β single-domain proteins, heterozygous β(MT3)–α(MT1), and a linker-truncated mutant ∆31–34 were prepared and characterized. In the presence/absence of various Zn7MT3 proteins, the Aβ1–42–Cu2+-mediated aggregation, the production of ROS, and the cellular toxicity were investigated by transmission electron microscopy, ROS assay by means of a fluorescent probe, and SH-SY5Y cell viability, respectively. The β domain cannot abolish Aβ1–42–Cu2+-induced aggregation, and neither the β domain nor the α domain can quench the production of ROS because of the redox cycling of Aβ–Cu2+. Similarly to wild-type Zn7MT3, the heterozygous β(MT3)–α(MT1) possesses the characteristic of alleviating Aβ1–42 aggregation and oxidative stress to neuronal cells. Therefore, the two domains through the linker Lys-Lys-Ser form a cooperative unit, and each of them is indispensable in conducting its bioactivity. The α domain plays an important role in modulating the stability of the metal–thiolate cluster, and the α–β domain–domain interaction through the linker is critical for its protective role in the brain.



Similar content being viewed by others
References
Khachaturian ZS (1985) Arch Neurol 42:1097–1104
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BL, Beyreuther K (1985) Proc Natl Acad Sci USA 82:4245–4249
Glenner GG, Wong CW (1984) Biochem Biophys Res Commun 120:885–890
Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH, Multhaup G, Beyreuther K, Mullerhill B (1987) Nature 325:733–736
Vigopelfrey C, Lee D, Keim P, Lieberburg I, Schenk DB (1993) J Neurochem 61:1965–1968
Hardy J (1997) Trends Neurosci 20:154–159
Hardy J, Selkoe DJ (2002) Science 297:353–356
Dong J, Atwood CS, Anderson VE, Siedlak SL, Smith MA, Perry G, Carey PR (2003) Biochemistry 42:2768–2773
Bush AI (2003) Trends Neurosci 26:207–214
Smith DG, Cappai R, Barnham KJ (2007) Biochim Biophys Acta Biomembr 1768:1976–1990
Li F, Calingasan NY, Yu FM, Mauck WM, Toidze M, Almeida CG, Takahashi RH, Carlson GA, Beal MF, Lin MT, Gouras GK (2004) J Neurochem 89:1308–1312
Lovell MA, Robertson JD, Teesdale WJ, Campbell JL, Markesbery WR (1998) J Neurol Sci 158:47–52
Smith MA, Harris PLR, Sayre LM, Perry G (1997) Proc Natl Acad Sci USA 94:9866–9868
Bush AI, Pettingell WH, Multhaup G, Paradis MD, Vonsattel JP, Gusella JF, Beyreuther K, Masters CL, Tanzi RE (1994) Science 265:1464–1467
Atwood CS, Moir RD, Huang XD, Scarpa RC, Bacarra NME, Romano DM, Hartshorn MK, Tanzi RE, Bush AI (1998) J Biol Chem 273:12817–12826
Esler WP, Stimson ER, Jennings JM, Ghilardi JR, Mantyh PW, Maggio JE (1996) J Neurochem 66:723–732
Balakrishnan R, Parthasarathy R, Sulkowski E (1998) J Pept Res 51:91–95
Faller P, Hureau C (2009) Dalton Trans 1080–1094
Hureau C, Faller P (2009) Biochimie 91:1212–1217
Halliwell B, Gutteridge JMC (1984) Biochem J 219:1–14
Meloni G, Sonois V, Delaine T, Guilloreau L, Gillet A, Teissie J, Faller P, Vasak M (2008) Nat Chem Biol 4:366–372
Pedersen JT, Hureau C, Hemmingsen L, Heegaard NHH, Ostergaard J, Vasak M, Faller P (2012) Biochemisty 51:1697–1706
Ding Z-C, Teng X-C, Cai B, Wang H, Zheng Q, Wang Y, Zhou G-M, Zhang M-J, Wu H-M, Sun H-Z, Huang Z-X (2006) Biochem Biophys Res Commun 349:674–682
Ding Z-C, Zheng Q, Cai B, Yu W-H, Teng X-C, Wang Y, Zhou G-M, Wu H-M, Sun H-Z, Zhang M-J, Huang Z-X (2007) J Biol Inorg Chem 12:1173–1179
Bao Q, Lu Y, Li W, Sun X, Zhu C, Li P, Huang Z-X, Tan X (2011) J Biol Inorg Chem 16(5):809–816
Palmiter RD, Findley SD, Whitmore TE, Durnam DM (1992) Proc Natl Acad Sci USA 89:6333–6337
Uchida Y, Tomonaga M (1989) Brain Res 481:190–193
Uchida Y, Takio K, Titani K, Ihara Y, Tomonaga M (1991) Neuron 7:337–347
Faller P, Vasak M (1997) Biochemistry 36:13341–13348
Hasler DW, Faller P, Vasak M (1998) Biochemistry 37:14966–14973
Irie Y, Keung WM (2001) Biochem Biophys Res Commun 282:416–420
Irie Y, Keung WM (2003) Brain Res 960:228–234
Schagger H, Vonjagow G (1987) Anal Biochem 166:368–379
Reinke AA, She HY, Gestwicki JE (2009) Bioorg Med Chem Lett 19:4952–4957
Smith DP, Ciccotosto GD, Tew DJ, Fodero-Tavoletti MT (2007) Biochemistry 46:2881–2891
Hou L, Zagorski MGJ (2007) Am Chem Soc 128:9260–9261
Syme CD, Nadal RC, Rigby SE, Viles JHJ (2004) Biol Chem 279:18169–18177
Guilloreau L (2006) J Biol Inorg Chem 11:1024–1038
Ding Z-C, Teng X-C, Zhen Q, Ni F-Y, Cai B, Wang Y, Zhou G-M, Sun H-Z, Tan X-S, Huang Z-X (2009) Biometals 22:817–826
Acknowledgments
This work was supported partly by the National Natural Science Foundation of China (no. 91013001, no. 20771029, and no. 31270869), Shanghai Pujiang Talent Project (08PJ14017), Shanghai Leading Academic Discipline Project (B108), the PhD Program of the Education Ministry of China (20100071110011), and the State Key Laboratory of Medical Neurobiology in the Shanghai Medical College of Fudan University, Shanghai, China.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, Y., Xu, Y., Bao, Q. et al. The molecular mechanism for human metallothionein-3 to protect against the neuronal cytotoxicity of Aβ1–42 with Cu ions. J Biol Inorg Chem 18, 39–47 (2013). https://doi.org/10.1007/s00775-012-0947-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-012-0947-3