Abstract.
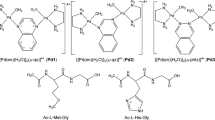
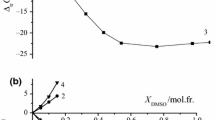
The hydrolysis of glycylglycine (GylGly), glycyl-L-leucine (GlyLeu), L-leucylglycine (LeuGly) and glycyl-DL-serine (GlySer) promoted by a copper(II)-cis,cis-1,3,5-triaminocyclohexane complex [Cu(II)TACH] was investigated at 70 °C and pH 7–10, using HPLC. The observed pseudo-first-order rate constants (k obs) and rate enhancing factors (REF) were as follows: 4.1×10–3 h–1 (REF=23) for GylGly, 1.6×10–3 h–1 (REF=21) for GlyLeu, 5.1×10–3 h–1 (REF=64) for LeuGly and 9.2×10–2 h–1 (REF=47) for GlySer [pH 8.1, dipeptide 2 mM, copper(II) 2 mM and TACH 2 mM]. Based on the pH dependence and dipeptide concentration dependence of the initial rates and speciation of the Cu(II)-TACH-dipeptide system at 25 °C and I=0.1, the reactions proceed via the formation of a ternary complex [Cu(TACH)(dipeptide)]+ as an intermediate followed by OH–-dependent and OH–-independent paths to give amino acid(s). GylGly, GlyLeu and LeuGly preferred the OH–-dependent path, while GlySer preferred the OH–-independent path. The latter can be explained by the intramolecular attack of the amide carbonyl group coordinated with its oxygen atom by the OH group in the serine residue. The X-ray crystal structure of [Cu(TACH)(GlyGly)]BPh4·MeOH confirmed that GlyGly coordinates to copper(II) ion with its terminal amino N and amide O atoms. The crystal structures of [Cu(TACH)(Gly)]BPh4 and [Cu2(TACH)2(OH)2](ClO4)2·NaClO4·H2O are also reported. Electronic supplementary material to this paper can be obtained by using the Springer Link server located at http://dx.doi.org/10.1007/s00775-002-0368-9.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Fujii, Y., Kiss, T., Gajda, T. et al. Copper(II)-cis,cis-1,3,5-triaminocyclohexane complex-promoted hydrolysis of dipeptides: kinetic, speciation and structural studies. J Biol Inorg Chem 7, 843–851 (2002). https://doi.org/10.1007/s00775-002-0368-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00775-002-0368-9